Nonarteritic Anterior Ischemic Optic Neuropathy (NAION)

Optic Neuropathy
1. What is optic neuropathy?
Section titled “1. What is optic neuropathy?”Optic neuropathy is a general term for a group of diseases in which some damage occurs to the optic nerve, leading to decreased visual function. It presents with one or more of the following: decreased visual acuity, visual field defects, color vision abnormalities, and reduced contrast sensitivity 2).
The optic nerve is composed of approximately 1.2 million axons of retinal ganglion cells (RGCs). Within the eye, they bundle together as the optic disc, pass through the optic canal, and reach the optic chiasm. Damage from the optic disc to the anterior part of the optic chiasm is called “anterior visual pathway disorder,” and most clinically significant optic neuropathies correspond to this region 2). Damage beyond the optic chiasm (optic tract, optic radiation, occipital lobe) is termed “posterior visual pathway disorder” and is treated as a visual pathway disorder rather than an optic neuropathy.
The optic nerve is divided into four segments. The intraocular segment (optic disc, about 1 mm) is prone to ischemic and inflammatory optic neuropathies. The intraorbital segment (about 25 mm) is often affected by compressive and inflammatory conditions. The intracanalicular segment (optic canal, about 5–8 mm) is susceptible to traumatic and bony compression. The intracranial segment (about 10 mm) is frequently compressed by pituitary adenomas and craniopharyngiomas. It is also important to distinguish between “anterior optic neuropathy,” where the optic disc can be directly observed, and “retrobulbar optic neuropathy,” where the fundus appears normal. In retrobulbar optic neuropathy, the absence of disc changes makes it easy to overlook as a cause of vision loss.
Blood flow to the optic disc is primarily provided by the short posterior ciliary arteries (SPC arteries). The SPC arteries are terminal branches of the ophthalmic artery and supply blood to the prelaminar, laminar, and retrolaminar regions of the optic disc. Circulatory disturbances in this area are the main pathological basis for NAION and AAION 3). The interior of the optic nerve is protected by the blood-brain barrier, but this protective mechanism breaks down in inflammatory optic neuritis and autoimmune diseases 2).
Common Pathological Pathway and Optic Atrophy
Section titled “Common Pathological Pathway and Optic Atrophy”Regardless of the cause, the final common pathway involves damage to retinal ganglion cells and optic nerve axons, leading to apoptosis and eventually optic atrophy. Optic atrophy is the end-stage of optic neuropathy and is recognized as pallor of the optic disc. Once atrophy is established, recovery of visual function is severely limited 2).
Optic nerve axons are a mixture of myelinated and unmyelinated fibers with diameters of 0.2–10 μm, broadly classified into P cells (responsible for luminance and contrast) and M cells (responsible for motion and contour). In optic neuropathies, P cells, which have high ATP consumption, are more susceptible to damage, which is related to the mechanism of central scotoma in toxic and nutritional optic neuropathies 2).
Overview of Epidemiology
Section titled “Overview of Epidemiology”The annual incidence rates of major optic neuropathies (per 100,000 adults) are summarized. NAION is the most common, with rates of 2.3–10.2 in the United States3). Idiopathic optic neuritis has an incidence of 1.6 in Japanese adults13), and LHON (Leber hereditary optic neuropathy) has approximately 117 new cases per year in Japan (2014 survey), making it a rare disease14). The estimated annual incidence of AAION/GCA is 0.36 per 100,000 in individuals aged 50 and older, and it is more common in Northern European Caucasians (approximately 30 per 100,000 in Norway). Visual complications of GCA occur in 10–30% (up to 70% in some reports), and AAION accounts for 60–90% of these1). The annual incidence of MOGAD is estimated at 1.6–4.8 per 1,000,0006).
Classification of Optic Neuropathies
Section titled “Classification of Optic Neuropathies”Optic neuropathies can be classified from multiple perspectives. The most clinically useful classification is shown below.
Classification by Onset Pattern
- Acute (hours to days): NAION, AAION, traumatic
- Subacute (days to weeks): Idiopathic optic neuritis, LHON, compressive (some)
- Chronic (months to years): ADOA, chronic compressive, radiation-induced (late-onset)
Classification by cause
- Ischemic: NAION, AAION, PION
- Inflammatory (optic neuritis): Idiopathic, MS-related, NMOSD-related, MOGAD-related, infectious
- Compressive: Tumor, cyst, aneurysm, thyroid eye disease
- Toxic: ethambutol, amiodarone, alcohol, etc.
- Nutritional: vitamin B12/B1/folate/copper deficiency
- Hereditary: LHON, ADOA, Wolfram syndrome
- Traumatic: optic canal fracture, direct injury
- Radiation-induced: delayed ischemic degeneration
Classification by location
- Anterior optic neuropathy (with disc changes): NAION, AAION, anterior optic neuritis
- Retrobulbar optic neuropathy (normal disc): toxic, hereditary, MS-related optic neuritis (most cases)
- Chiasmal lesions: pituitary adenoma, craniopharyngioma (bitemporal hemianopia)
Diagnostic Framework for Optic Neuropathy
Section titled “Diagnostic Framework for Optic Neuropathy”The diagnosis of optic neuropathy proceeds in five steps.
Step 1: Confirm optic nerve dysfunction Objectively confirm optic nerve dysfunction using RAPD, visual acuity, color vision, and visual field testing.
Step 2: Localization of the lesion Anterior (with optic disc changes) or retrobulbar (normal optic disc), intraorbital, intracanalicular, or intracranial. Estimate localization based on the visual field defect pattern.
Step 3: Unilateral or bilateral, acute or chronic This allows significant narrowing of the differential diagnosis.
Step 4: Exclusion of dangerous types Prioritize exclusion of GCA (ESR, CRP), compressive (MRI), and infectious (syphilis, fungal) causes.
Step 5: Specific tests Add anti-AQP4 antibody, anti-MOG antibody, mtDNA testing, and nutritional testing depending on the type.
2. Differences from optic neuritis (conceptual clarification)
Section titled “2. Differences from optic neuritis (conceptual clarification)”“Optic neuritis” and “optic neuropathy” are often confused in clinical practice, but they have a relationship of subordinate and superordinate concepts.
Confusing non-inflammatory optic neuropathies (ischemic, compressive, toxic, hereditary) with optic neuritis can lead to erroneous steroid administration. In particular, steroid therapy is contraindicated in invasive aspergillosis 2), and administering steroids in the acute phase of LHON is not only ineffective but also delays definitive diagnosis.
Main clinical features of optic neuritis
Section titled “Main clinical features of optic neuritis”Optic neuritis (inflammatory) has the following features that help differentiate it from non-inflammatory causes.
- Acute onset: progresses over days to 2 weeks, then shows a tendency to recover within 5 weeks.
- Pain on eye movement: present in about 50% of cases. Usually absent in ischemic, compressive, or toxic causes.
- Typical age: 15–45 years (ischemic is usually over 50).
- MRI findings: typical contrast enhancement of the optic nerve (gadolinium-enhanced T1).
- Spontaneous recovery: in idiopathic or MS-related cases, over 90% recover to visual acuity of 0.5 or better after 1 year (ONTT study)4).
- MS conversion risk: 15 years after first episode, 25% without brain MRI lesions and 78% with lesions progress to MS4).
Situations where differentiation is difficult
Section titled “Situations where differentiation is difficult”Below, differentiation between optic neuritis and non-inflammatory optic neuropathy becomes difficult.
- Acute unilateral vision loss in women over 50: NAION vs. AQP4 antibody-positive optic neuritis (NMOSD)
- Acute bilateral vision loss in young adults: LHON vs. simultaneous bilateral optic neuritis vs. toxic optic neuropathy
- Unilateral vision loss with optic disc swelling: NAION vs. anterior optic neuritis vs. optic disc vasculitis
AQP4 antibody-positive optic neuritis is a steroid-resistant refractory disease accounting for about 10% of idiopathic optic neuritis, with a higher age of onset and a female predominance (female-to-male ratio 9:1) compared to typical optic neuritis 5). The AQP4 antibody targets aquaporin-4 channels expressed on astrocytes in the optic nerve, causing complement-dependent cytotoxicity 5). Since treatment strategies differ fundamentally between AQP4 antibody-positive and negative cases, antibody testing should be ordered before steroid pulse therapy. The cell-based assay (CBA) method has higher sensitivity and specificity than ELISA and is recommended 5).
Before performing steroid pulse therapy for optic neuritis, infectious diseases (hepatitis B, syphilis, HIV, fungal infections) must be ruled out 2). High-dose steroid administration in hepatitis B virus carriers can cause fulminant hepatitis. In syphilitic optic neuritis, antibiotics take priority over steroids.
3. Disease types requiring high urgency
Section titled “3. Disease types requiring high urgency”The table below shows the types of optic neuropathy that require particularly urgent management.
| Disease type | Urgency | Consequences if missed |
|---|---|---|
| Arteritic anterior ischemic optic neuropathy (AAION/GCA) | Most urgent | Fellow eye blindness (65% develop within 10 days if untreated) 1) |
| Acute compressive optic neuropathy (tumor, cyst, hematoma) | Urgent | Progressive optic atrophy and irreversible vision loss |
| Nasal optic neuropathy (invasive aspergillosis) | Emergency | Reported mortality rate of 94%2) |
| Traumatic optic neuropathy (optic canal fracture) | Emergency | Early treatment may preserve visual function |
| Posterior ischemic optic neuropathy (PION) | Emergency | Occurs after surgery or massive blood loss; often irreversible |
GCA Emergency Response Flow
Section titled “GCA Emergency Response Flow”Amaurosis fugax appears as a prodromal symptom in approximately 30% of cases of permanent vision loss, occurring on average 8.5 days prior 1). At this point, it is extremely important to suspect GCA and proactively initiate steroids as secondary prevention.
4. Entry Point for Differential Diagnosis
Section titled “4. Entry Point for Differential Diagnosis”Differential diagnosis of optic neuropathy is systematically organized along four axes: acute vs. chronic, unilateral vs. bilateral, presence or absence of pain, and optic disc findings.
Acute vs. Chronic
Section titled “Acute vs. Chronic”Acute (reaches maximum deficit within hours to days)
- NAION: Typically presents with sudden vision loss noticed upon waking
- AAION: Rapid unilateral vision loss, severe (vision <20/200 in >60%) 1)
- Traumatic: Immediately to within hours after trauma
Subacute (days to weeks)
- Idiopathic optic neuritis: Progresses over 2 weeks, then recovery begins
- LHON: Unilateral painless subacute vision loss, becoming bilateral within weeks to months
- Compressive: May progress subacutely due to rapid tumor growth or hemorrhage
Chronic (slow progression over months to years)
- ADOA (autosomal dominant optic atrophy): Bilateral slow progression starting in school age
- Chronic compressive: Due to pituitary adenoma, orbital tumor, etc.
- Toxic/nutritional: Subacute to chronic progression with long-term exposure or malnutrition
One eye or both eyes
Section titled “One eye or both eyes”Diseases that tend to be unilateral
- NAION: Usually unilateral (simultaneous bilateral onset is extremely rare)
- AAION: Starts unilaterally; if untreated, 65% spread to the fellow eye within 10 days1)
- Idiopathic optic neuritis / MS-related: Typically unilateral
- Compressive, traumatic, radiation-induced: Often unilateral
Diseases that tend to be bilateral
- LHON: Initially unilateral, becomes bilateral within weeks to months (eventually nearly 100% bilateral)
- ADOA: Bilateral symmetric onset
- Toxic/nutritional: Principally bilateral (if one eye is completely normal, consider excluding toxic causes) 2)
- NMOSD/AQP4-IgG positive: Severe, bilateral, relapsing. 60–69% have permanent vision loss of 20/200 or worse in at least one eye 5)
- MOGAD: Frequent bilateral onset (extremely rare in MS-related or idiopathic cases) 6)
Presence or absence of pain
Section titled “Presence or absence of pain”| Pain present | No pain (painless) |
|---|---|
| Idiopathic optic neuritis (eye movement pain ~50%)13) | NAION (no eye pain; headache, jaw claudication are GCA systemic symptoms) |
| MS-related optic neuritis | AAION (no eye pain; jaw claudication, headache present) |
| NMOSD-related (periorbital pain) | LHON/ADOA (painless) 14) |
| MOGAD (often with headache) 6) | Toxic/nutritional (no pain with eye movement) |
| Traumatic (pain from injury) | Compressive (usually painless but may have orbital pain) |
Classification by Optic Disc Findings
Section titled “Classification by Optic Disc Findings”5. Examination and Testing
Section titled “5. Examination and Testing”Essential Ophthalmic Evaluation
Section titled “Essential Ophthalmic Evaluation”When optic neuropathy is suspected, perform the following evaluations systematically.
Visual acuity and color vision Corrected visual acuity is fundamental for assessing the degree of impairment and monitoring progression. Color vision (Ishihara, Farnsworth-Munsell 100 hue) is often affected early in optic neuropathy, and color vision abnormalities may precede visual acuity loss even when acuity is relatively preserved. In toxic and nutritional optic neuropathies, desaturation of red perception is an early sign 2). In ADOA (autosomal dominant optic atrophy), acquired tritanopia (blue-yellow color vision defect) is characteristic, showing a tritan axis on the Farnsworth-Munsell 100 hue test 2).
RAPD (relative afferent pupillary defect)
RAPD is assessed using the swinging flashlight test and serves as an objective indicator of optic nerve dysfunction. It is also useful for differentiating from malingering and functional visual disorders 2).
OCT (Optical Coherence Tomography)
Section titled “OCT (Optical Coherence Tomography)”OCT is an essential test for staging and monitoring optic neuropathy.
| Evaluation parameter | Acute phase findings | Chronic phase findings |
|---|---|---|
| pRNFL (peripapillary RNFL thickness) | Thickened in papilledema (caution for false positives) | Thinned in optic atrophy |
| GCL-IPL (ganglion cell + inner plexiform layer thickness) | Little change | Thinned reflecting retinal ganglion cell damage |
| Optic disc shape | Cupping disappears or decreases | Becomes fixed after atrophy |
Pattern of pRNFL thinning and differential diagnosis
The pattern of pRNFL thinning on OCT is useful for differential diagnosis from several weeks to months after onset.
- Severe circumferential thinning: NMOSD (AQP4 antibody-positive type) is the most severe. Thinning is severe all around.
- Temporal thinning (papillomacular bundle): Toxic, nutritional, LHON, ADOA. Corresponds to central scotoma.
- Superior or inferior sectoral thinning: NAION. Corresponds to superior or inferior arcuate defect.
- MOGAD vs. MS-related: pRNFL thinning after MOG-ON is more severe than MS-related 6).
- Differentiation from glaucoma: In glaucoma, optic disc changes (cupping enlargement) precede, and GCL thinning pattern differs.
Visual Field Testing (Humphrey Visual Field)
Section titled “Visual Field Testing (Humphrey Visual Field)”Visual field defect patterns directly relate to anatomical localization of the lesion.
| Visual field defect pattern | Suspected disease | Anatomical correlation |
|---|---|---|
| Central scotoma, cecocentral scotoma | Toxic/nutritional, LHON, ADOA, idiopathic optic neuritis2) | Papillomacular bundle (PMB) damage |
| Horizontal defect (inferotemporal to inferior) | NAION (sectoral defect)3) | Ischemia of upper or lower half of optic disc |
| Bitemporal hemianopia | Optic chiasm compression (pituitary adenoma, craniopharyngioma) | Central crossing fibers of optic chiasm |
| Homonymous hemianopia (same side) | Post-chiasmal lesion (optic tract, optic radiation, occipital lobe) | Central visual pathway |
| Junctional scotoma | Anterior chiasmal lesion, a type of nasal optic neuropathy | Papillomacular bundle + contralateral superotemporal |
| Generalized sensitivity reduction | NMOSD-related / after severe optic neuritis5) | Extensive optic nerve fiber damage |
| Enlarged Mariotte blind spot (normal visual acuity) | Optic disc drusen, optic disc vasculitis, perioptic neuritis | Only around the optic disc |

Blood and Cerebrospinal Fluid Tests
Section titled “Blood and Cerebrospinal Fluid Tests”Recommended tests for initial evaluation of acute optic neuropathy are shown.
| Test Item | Purpose | Clinical Significance |
|---|---|---|
| ESR, CRP, platelets | Exclude GCA (AAION) | Highest priority. CRP has higher specificity than ESR1) |
| Anti-AQP4 antibody (cell-based assay) | Diagnosis of NMOSD | ELISA has lower sensitivity and specificity than CBA5) |
| Anti-MOG antibody (cell-based assay) | Diagnosis of MOGAD | CBA recognizing native MOG molecule is essential6) |
| Syphilis serology (TPHA/RPR) and HIV | Exclusion of infectious optic neuritis | Must be checked before steroids |
| Vitamin B12, B1, folic acid, copper | Evaluation of nutritional optic neuropathy | Especially important after bariatric surgery or on a vegan diet |
| Mitochondrial genes (mt11778, mt14484, mt3460) | Diagnosis of LHON | These three mutations cover over 95% of all cases14) |
| β-D-glucan and fungal culture | Suspected invasive aspergillosis | Tends to be elevated in invasive type2) |
| ACE, lysozyme, chest CT | Rule out sarcoidosis | — |
| ANA, dsDNA, ANCA | Systemic autoimmune diseases (SLE, vasculitis) | — |
Indications for CSF examination Suspected infectious meningitis/encephalitis, diagnostic aid for NMOSD/MOGAD (CSF pleocytosis, elevated IgG index), diagnostic aid for MS (CSF oligoclonal bands). NMOSD may show marked pleocytosis of up to 50 cells/μL or more6).
Imaging tests
Section titled “Imaging tests”| Test | Main evaluation items | Notes |
|---|---|---|
| Orbital MRI (fat-suppressed T2/contrast-enhanced T1) | Optic nerve inflammation, edema, mass, contrast enhancement | Contrast enhancement in optic neuritis. No enhancement in acute LHON14) |
| Head MRI (FLAIR) | Demyelinating lesions (MS, NMOSD), intracranial lesions | Used in McDonald criteria for MS diagnosis |
| CT (bone window) | Optic canal fracture (trauma), sinus lesions, calcification (drusen) | Essential for trauma and nasal diseases |
| Head ultrasound (color Doppler) | Halo sign for GCA diagnosis | Sensitivity 68%, specificity 91% (100% if bilateral positive)1) |
| PET-CT (FDG) | Evaluation of large-vessel involvement in GCA | Sensitivity 92%, specificity 85% (GAPS study)1) |
| Orbital ultrasound | Confirmation of calcification in optic disc drusen | — |
In acute optic neuropathy, orbital MRI is generally required. The priority is to rule out compressive lesions, and treatment strategy differs greatly depending on whether contrast enhancement (inflammatory) or compression/deformation (compressive) is observed in the optic nerve 2). If contrast MRI shows no inflammatory findings in the optic nerve, toxic, hereditary, or nutritional causes should be considered. In the acute phase of LHON, there are no inflammatory findings, and fluorescein angiography shows no leakage from the optic disc, which is an important distinguishing point from inflammatory optic neuritis 14). If MRI is contraindicated, CT is used to evaluate the sinuses, orbit, and optic canal.
Use of Visual Evoked Potentials (VEP)
Section titled “Use of Visual Evoked Potentials (VEP)”Visual evoked potentials (VEP) are useful as a marker for demyelinating optic neuritis. Prolongation of P100 latency (normal within 130 ms) suggests demyelination, and in MS-related and idiopathic optic neuritis, the prolongation often persists even after recovery 13). Reduction in amplitude is prominent in axonal damage (toxic, ischemic). In toxic optic neuropathy, there is no delay in P100 latency, and only amplitude is reduced, which helps differentiate it from demyelinating disease 13). A decrease in critical flicker frequency (CFF) also reflects functional impairment in optic neuropathy, and is notably reduced in NMOSD-related optic neuritis 13).
6. Characteristics by Cause
Section titled “6. Characteristics by Cause”Major optic neuropathies are classified by cause, and the characteristics of each type are summarized.
6-1. Ischemic Optic Neuropathy
Section titled “6-1. Ischemic Optic Neuropathy”Nonarteritic Anterior Ischemic Optic Neuropathy (NAION)
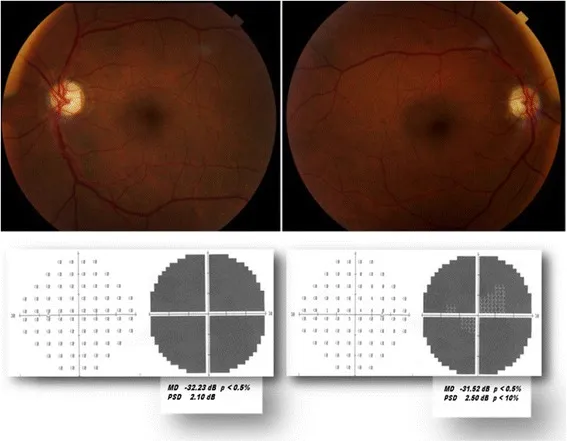
Section titled “Nonarteritic Anterior Ischemic Optic Neuropathy (NAION)”NAION is the most common type of acute optic neuropathy in adults, often occurring in those aged 50 and older 3). Ischemic infarction of the optic disc occurs due to acute circulatory failure of the short posterior ciliary arteries (SPC arteries).
- Epidemiology: Annual incidence in the United States is 2.3–10.2 per 100,000 3). Most cases occur in people aged 50 and older, but incidence in those under 50 is also increasing.
- Risk factors: Hypertension, diabetes, hyperlipidemia, sleep apnea syndrome, and disc at risk (small optic disc with cup-to-disc ratio <0.2). Disc at risk is present in about 80% of fellow eyes 3).
- Symptoms: Painless, acute unilateral vision loss, often noticed upon waking. RAPD positive on the affected side.
- Optic disc findings: Hyperemia and swelling (inferior sectoral pattern). May be accompanied by hemorrhages.
- Visual field: Altitudinal defect (inferior temporal defect is typical).
- Treatment: No established effective treatment in the acute phase. Management of vascular risk factors and treatment of sleep apnea are central to preventing recurrence.
- Recent attention: An observational study in 2024 reported an increased risk of NAION in users of semaglutide (GLP-1 receptor agonist). However, causality has not been established, and the benefits for diabetes and obesity treatment must be weighed when prescribing7).
Arteritic Anterior Ischemic Optic Neuropathy (AAION/GCA)
Section titled “Arteritic Anterior Ischemic Optic Neuropathy (AAION/GCA)”AAION accounts for 5–10% of all anterior ischemic optic neuropathies but is the most urgent optic neuropathy 1). It is caused by vasculitis of the short posterior ciliary arteries supplying the optic nerve head, and over 90% of cases are due to giant cell arteritis (GCA).
- Epidemiology: More common in elderly women aged 50 years or older (especially 75 years or older), with a female-to-male ratio of 3:1 1). The incidence in Japan is 1.47 per 100,000 people, much lower than in Western countries.
- Symptoms: Acute severe unilateral vision loss (vision <20/200 in over 60%, no light perception in over 20%) 1). About 30% experience transient monocular vision loss (amaurosis fugax) beforehand.
- Systemic symptoms: Headache (65–90%), jaw claudication (11–45%), scalp tenderness, fever, and polymyalgia rheumatica-like symptoms.
- Optic disc findings: Chalky pale swelling (pallid swelling) is characteristic.
- Tests: Elevated ESR and CRP (elevated in >80% of cases)1). Temporal artery biopsy (sensitivity and specificity >95%)1).
- Treatment: High-dose steroids (mPSL 500–1,000 mg IV for 3 days, then oral prednisolone taper)1). Tocilizumab (IL-6 receptor antagonist) is effective for steroid sparing (GiACTA trial, RCT evidence)1).
- Prognosis: Visual recovery in the affected eye is rarely expected, but steroid treatment is effective in preventing onset in the fellow eye.
6-2. Inflammatory Optic Neuritis
Section titled “6-2. Inflammatory Optic Neuritis”Idiopathic Optic Neuritis
Section titled “Idiopathic Optic Neuritis”An inflammatory disease of the optic nerve caused by an autoimmune mechanism, accounting for the majority of optic neuritis cases 13). The typical age of onset is 15–45 years, with women comprising approximately 70% of cases. The annual incidence in Japan is 1.6 per 100,000 adults 13).
- Symptoms: Acute unilateral vision loss and eye movement pain (approximately 50%). Progression occurs over several days to 2 weeks, with a tendency for spontaneous recovery within 5 weeks.
- Visual field: Central scotoma or cecocentral scotoma are common.
- MRI: Contrast enhancement (gadolinium) consistent with the optic nerve.
- Prognosis: More than 90% recover to visual acuity of 0.5 or better after 1 year (ONTT study) 4).
- Treatment: Steroid pulse therapy with methylprednisolone 1,000 mg/day for 3 days accelerates recovery. Oral steroid monotherapy is not performed because it increases the recurrence rate (ONTT study) 4).
- Risk of conversion to MS: Cumulative incidence 50% over 15 years after first attack. Without brain MRI lesions: 25%, with lesions: 78%4).
MS-related optic neuritis
Section titled “MS-related optic neuritis”Optic neuritis occurs as the initial symptom in about 30% of MS cases, and up to 75% of MS patients experience at least one episode of optic neuritis during their lifetime8). It is typically unilateral, with pain on eye movement, and often retrobulbar (normal fundus in 65%). Brain MRI showing demyelinating lesions (T2/FLAIR hyperintensity) is key for diagnosis. After diagnosis according to the McDonald criteria (2017 version, revised 2024), disease-modifying therapies (DMTs: interferon beta, natalizumab, fingolimod, anti-CD20 drugs, etc.) are used to prevent relapses.
NMOSD (neuromyelitis optica spectrum disorder)-related optic neuritis
Section titled “NMOSD (neuromyelitis optica spectrum disorder)-related optic neuritis”AQP4-IgG positive. Predominantly affects women (female:male ratio 9:1), middle-aged, and Asian populations. It is more severe, bilateral, and recurrent than MS-related optic neuritis5). Permanent visual impairment of 20/200 or worse in at least one eye occurs in 60–69% of cases5). Orbital MRI shows long segment optic nerve lesions (often involving the chiasm). Treatment includes acute-phase steroid pulse therapy, followed by plasma exchange, and then relapse-prevention immunotherapy (eculizumab, rituximab, satralizumab)5).
MOGAD (MOG antibody-associated disease)-related optic neuritis
Section titled “MOGAD (MOG antibody-associated disease)-related optic neuritis”MOG-IgG positive. Annual incidence is 1.6 to 4.8 per million people 6). Bilateral onset is frequent with marked optic disc swelling and predominant anterior involvement. MRI shows characteristic optic disc swelling, long-segment optic nerve lesions, and optic perineuritis 6). Sex ratio comparison among MS-ON, AQP4-ON, and MOG-ON shows male-to-female ratios of 3:1, 7-9:1, and 1:1, respectively 6). Steroid-responsive but prone to relapse with early dose reduction; careful tapering is necessary 6).
6-3. Compressive Optic Neuropathy
Section titled “6-3. Compressive Optic Neuropathy”Caused by compression of the optic nerve by tumors, aneurysms, hematomas, abscesses, cysts, or enlarged extraocular muscles. Symptoms and visual field defect patterns vary greatly depending on the compression site.
- Orbital apex: Thyroid eye disease (thyroid optic neuropathy complicates 3-8.6% of thyroid eye disease, 70% bilateral), orbital tumors (benign: idiopathic orbital inflammation 20%, pleomorphic adenoma 13%, hemangioma 13%; malignant: malignant lymphoma most common) 9).
- Optic chiasm: Pituitary adenoma (most common in adults, bitemporal hemianopia), craniopharyngioma (most common in children).
- Nasal optic neuropathy (special note): Invasive type of paranasal sinus mycosis (Aspergillus, Mucor) has a reported mortality rate of 94% 2). It may be misdiagnosed as retrobulbar optic neuritis because steroids provide temporary improvement, and unknowingly continuing steroids can be fatal. CT/MRI findings of bone destruction are key to diagnosis, and urgent collaboration with an otolaryngologist is necessary.
The principle of treatment is surgical removal of the causative lesion. The earlier the decompression, the better the recovery of visual function; once optic atrophy is established, recovery is limited.
6-4. Toxic Optic Neuropathy
Section titled “6-4. Toxic Optic Neuropathy”This is a group of diseases in which the anterior visual pathway is damaged by exposure to chemical substances2). It is generally bilateral and painless; if one eye is completely normal, toxic optic neuropathy should be excluded2).
- Causative substances: Tobacco, alcohol, paint thinner (drugs: ethambutol [most famous], amiodarone, linezolid, cisplatin, immunosuppressants, etc.).
- Visual field: Characteristically, a centrocecal scotoma or central scotoma (due to predominant damage to P cells)2).
- Color vision: Dulling of red color perception is an early sign.
- Fundus: Initially normal or mild redness → chronic phase: temporal pallor of the optic disc and loss of papillomacular bundle fibers.
- RAPD: Usually negative due to bilateral symmetry.
- VEP: Decreased amplitude without delay in P100 latency (latency delay is characteristic of demyelinating optic neuritis) 13).
- Ethambutol: Dose-dependent (risk increases above 15 mg/kg). Baseline visual function testing before administration and regular monitoring are essential.
- Treatment: Discontinuation of the causative agent is fundamental. No specific antidote exists.
6-5. Nutritional optic neuropathy
Section titled “6-5. Nutritional optic neuropathy”Bilateral, symmetric, progressive optic neuropathy caused by deficiency of B vitamins (B12, B1, B2, B9) and copper. It lies on the same spectrum as toxic optic neuropathy but is distinguished by nutritional deficiency as the cause. Modern risk factors include post-bariatric surgery (lifelong monitoring required), vegan diet, and alcohol dependence. Symptoms, visual field, and fundus findings are similar to toxic optic neuropathy (central scotoma, painless, temporal pallor of the optic disc). Early nutritional supplementation can lead to recovery, but chronic cases may result in irreversible damage.
6-6. Hereditary Optic Neuropathy
Section titled “6-6. Hereditary Optic Neuropathy”LHON (Leber Hereditary Optic Neuropathy)
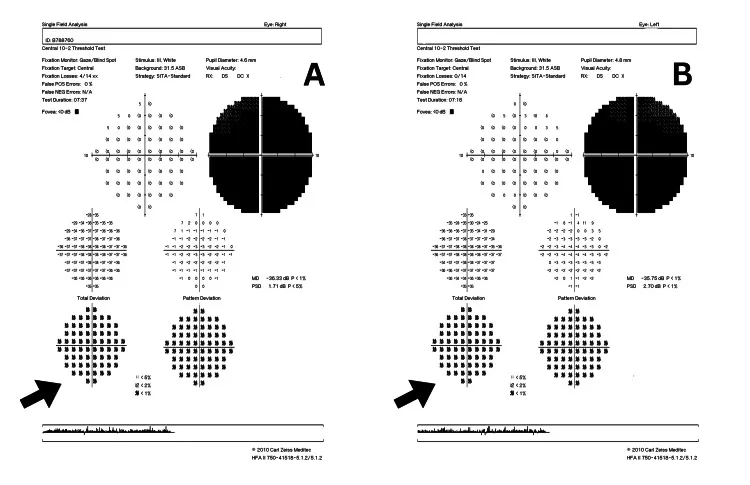
Section titled “LHON (Leber Hereditary Optic Neuropathy)”An acute or subacute optic neuropathy caused by point mutations in mitochondrial DNA (mtDNA) with maternal inheritance 14). Three major mutations (mt3460, mt11778, mt14484) account for over 95% of all cases, with mt11778 accounting for approximately 90% of all cases in Asia 14).
- Epidemiology: Prevalence 1/31,000 to 1/68,000. Penetrance 2.5–17.5%, with most carriers not developing the disease 14). In Japan, approximately 117 new cases per year (2014 survey). Designated as an intractable disease.
- Symptoms: Predominantly affects young males (male-to-female ratio approximately 93:7). Subacute, painless bilateral central scotoma (one eye affected first, then both eyes within weeks to months).
- Acute fundus: Optic disc hyperemia and swelling, with dilation and tortuosity of peripapillary capillaries. Fluorescein angiography shows no leakage from the optic disc (important distinction from inflammatory optic neuritis) 14).
- Pupillary light reflex: Relatively preserved compared to other optic nerve diseases, or only mildly impaired 14).
- Final visual acuity: Around 0.01 or less in mt11778. mt14484 has the highest rate of spontaneous recovery 14).
- Treatment: Idebenone 900 mg/day (approved by EMA). In the LEROS trial, CRR (clinically relevant recovery) was 46.0% (vs placebo) 10). In the Wales cohort study, CRR reached 86% at 27 months 10). Gene therapy has also shown efficacy in phase III trials 11). Smoking cessation guidance is important for preventing onset and progression.
ADOA (Autosomal Dominant Optic Atrophy)
Section titled “ADOA (Autosomal Dominant Optic Atrophy)”The most frequent hereditary optic neuropathy, primarily caused by OPA1 gene mutations. It is discovered during school age as bilateral unexplained visual developmental impairment. Acquired third color vision deficiency (blue-yellow color blindness) is characteristic. OCT shows RNFL thinning predominantly in the temporal to inferior quadrants 2). Currently, there is no effective treatment, and management mainly consists of low vision care and genetic counseling.
6-7. Traumatic Optic Neuropathy
Section titled “6-7. Traumatic Optic Neuropathy”Optic nerve damage occurring after blunt trauma to the head or orbit, often due to indirect force within the optic canal. Acute vision loss occurs immediately after injury. CT bone window confirms optic canal fracture. High-dose steroids or optic canal decompression surgery may be considered, but evidence for both is limited.
6-8. Radiation Optic Neuropathy
Section titled “6-8. Radiation Optic Neuropathy”A delayed ischemic disorder following radiotherapy for head and neck or orbital tumors. Visual function is lost through mechanisms primarily involving endothelial damage. Onset occurs 3 months to 9 years after irradiation, most commonly at 10–20 months, with an average of about 18 months 12). Risk increases with total dose >50 Gy or single fraction >10 Gy. It is usually painless and has a poor prognosis. No established treatment exists; systemic steroids, anticoagulation, and hyperbaric oxygen therapy have limited efficacy 12). MRI shows contrast enhancement corresponding to the optic nerve.
6-9. Other Specific Optic Neuropathies
Section titled “6-9. Other Specific Optic Neuropathies”Posterior Ischemic Optic Neuropathy (PION) Ischemia of the posterior intraorbital or intracanalicular optic nerve. Postoperative PION is typical, occurring after massive hemorrhage, hypotension, spinal surgery, or cardiac surgery. Fundus findings are normal (no optic disc changes), but there is acute vision loss. The pupillary light reflex shows a relative afferent pupillary defect (RAPD). No effective treatment has been established.
Neuroretinitis A syndrome characterized by optic disc edema and a macular star figure. Causes include Bartonella henselae (cat scratch disease), syphilis, and Leber idiopathic stellate neuroretinitis. The star figure results from exudate deposition along the Henle fiber layer in the macula, appearing several weeks after disc edema resolves. It tends to resolve spontaneously, but specific treatment is given for infectious causes.
Papillophlebitis A syndrome occurring in young to middle-aged adults, characterized by unilateral optic disc edema without visual dysfunction and retinal venous dilation and tortuosity. Enlargement of the Mariotte blind spot is observed, but corrected visual acuity is normal. Orbital MRI is normal. Prognosis is good and treatment is not required, but differentiation from papilledema, optic neuritis, and ischemic optic neuropathy is important2).
Immune checkpoint inhibitor-related optic neuropathy Optic neuropathy rarely occurs as an immune-related adverse event (irAE) of cancer immunotherapy (e.g., nivolumab, pembrolizumab). It is treated with steroid pulse therapy, and the continuation of cancer treatment should be discussed with an oncologist.
Summary Table of Optic Neuropathy One-Liners
Section titled “Summary Table of Optic Neuropathy One-Liners”| Disease | Laterality | Pain | Optic disc findings | Visual field | Key points |
|---|---|---|---|---|---|
| NAION | Unilateral | None | Redness and swelling (inferior) | Horizontal sectoranopia | Onset upon waking, disc at risk3) |
| AAION/GCA | Unilateral → bilateral | None (headache, jaw claudication) | Pale swelling | Variable (severe) | Most urgent, elevated ESR/CRP1) |
| Idiopathic optic neuritis | Unilateral | Present (50%) | Swollen or normal | Central scotoma | Young women, risk of MS conversion 13) |
| NMOSD-related | Tends to become bilateral | Yes | Swollen or normal | Variable (severe) | AQP4 antibody positive, refractory5) |
| MOGAD-related | Often bilateral | Present (headache) | Severe swelling | Variable | MOG antibody, perineuritis6) |
| Compressive | Unilateral/Bilateral | None | Normal or atrophic | Variable | MRI required9) |
| Toxic | Both eyes | None | Normal → temporal pallor | Centrocecal scotoma | Ask about causative substance 2) |
| Nutritional | Both eyes | None | Normal → temporal pallor | Central scotoma | B12/copper deficiency |
| LHON | Both eyes (one eye first) | None | Redness/telangiectasia | Central scotoma | Young male, no fluorescein leakage14) |
| ADOA | Both eyes | None | Pale | Central scotoma | School age, blue-yellow color vision deficiency2) |
| Traumatic | Unilateral | Yes (trauma) | Normal | Variable | CT confirms optic canal fracture |
| Radiation-induced | Unilateral/bilateral | None | Normal → atrophy | Various | Average 18 months12) |
7. Treatment Principles
Section titled “7. Treatment Principles”Treatment of optic neuropathy depends on the cause and differs fundamentally by disease type. Common principles are shown below.
Situations Requiring Emergency Treatment
Section titled “Situations Requiring Emergency Treatment”- GCA/AAION: High-dose systemic steroids (start on the same day). Do not wait for test results 1).
- Acute compressive optic neuropathy: Surgical decompression (urgent collaboration with otolaryngology and neurosurgery). In invasive aspergillosis, discontinue steroids and switch to antifungal agents 2).
- Traumatic: Consideration of optic nerve decompression surgery.
Treatment of inflammatory (optic neuritis)
Section titled “Treatment of inflammatory (optic neuritis)”Steroid pulse therapy (mPSL 1,000 mg/day for 3 days) accelerates recovery speed in the acute phase 4). Oral steroids alone are not administered because they increase the risk of relapse in idiopathic optic neuritis (ONTT study) 4). In the acute phase of NMOSD, stepwise treatment is performed: steroid pulse → plasma exchange for insufficient response → relapse prevention immunotherapy 5). MOGAD optic neuritis is steroid-responsive but relapses with early tapering, so careful gradual reduction is necessary 6).
Treatment of ischemic (NAION)
Section titled “Treatment of ischemic (NAION)”No effective treatment has been established for the acute phase. Management of vascular risk factors (hypertension, diabetes, hyperlipidemia, sleep apnea syndrome) is the mainstay, and prevention of onset in the fellow eye is the treatment goal. Regarding semaglutide (GLP-1 receptor agonist), observational studies suggest an association with NAION, so careful explanation and follow-up are desirable in patients with a history or risk 7).
Treatment of hereditary (LHON)
Section titled “Treatment of hereditary (LHON)”Idebenone 900 mg/day is approved by the European Medicines Agency (EMA) and achieved a CRR of 46% in the LEROS trial (placebo-controlled) 10). The Welsh cohort reported a CRR of 86% at 27 months, indicating favorable real-world outcomes 10). Gene therapy (lenadogene nolparvovec) has shown efficacy in a Phase III trial, and further expansion of indications is expected 11). Smoking cessation counseling (smoking is a risk factor for onset) and appropriate low vision care are essential 14).
Treatment of Toxic and Nutritional Optic Neuropathy
Section titled “Treatment of Toxic and Nutritional Optic Neuropathy”The basic approach is discontinuation of the causative substance or supplementation of deficient nutrients. Early intervention can lead to visual function recovery, but chronic cases may result in irreversible damage. Ethambutol optic neuropathy is dose-dependent, so baseline visual function testing before administration and regular monitoring (visual acuity, color vision, visual field) are essential.
Treatment of Compressive Optic Neuropathy
Section titled “Treatment of Compressive Optic Neuropathy”Surgical removal of the causative lesion is the principle. For thyroid optic neuropathy, urgent or elective orbital decompression is performed. Since visual recovery after surgical removal is limited once optic atrophy has progressed, early diagnosis and treatment are important.
Prognosis Overview
Section titled “Prognosis Overview”| Disease type | Visual prognosis | Recovery potential |
|---|---|---|
| Idiopathic optic neuritis | Over 90% achieve 0.5 or better at 1 year4) | Good recovery |
| NAION | 1/3 improve, 1/3 unchanged, 1/3 worsen | Partial recovery |
| AAION/GCA | Recovery of the affected eye is almost never expected | Goal is prevention in the fellow eye1) |
| NMOSD-related | Severe. At least one eye with 20/200 or worse in 60–69%5) | Relapse prevention is important |
| LHON (mt11778) | Final visual acuity often around 0.01 | Spontaneous recovery only in some cases14) |
| LHON (mt14484) | Mutation type with highest spontaneous recovery rate | Recovery cases exist14) |
| ADOA | Slowly progressive, often does not lead to severe blindness | No progression suppression |
| Compressive | Recoverable with early removal | Before atrophy progresses is critical |
| Toxic/nutritional | Recoverable with early cessation/supplementation | Irreversible in chronic cases |
| Radiation-induced | Acute, severe, often irreversible vision loss 12) | No established treatment |
Rapid identification of the underlying cause. If GCA is suspected, start steroids on the same day without waiting for test results to prevent vision loss in the fellow eye 1). For compressive causes, surgical removal of the causative lesion is the only way to preserve visual function, and delay in diagnosis leads to irreversible optic atrophy. In inflammatory cases (optic neuritis), steroid pulse therapy accelerates recovery, but additional treatment strategies vary greatly depending on the subtype. In particular, for anti-AQP4 antibody-positive cases, steroid monotherapy leads to repeated relapses, so early introduction of relapse-preventive immunotherapy is key to preserving visual function 5).
Both conditions present with similar initial symptoms of “acute to subacute painless vision loss,” and are easily confused, especially during the acute phase of LHON when optic disc swelling occurs. The most important distinguishing feature is the presence or absence of fluorescein leakage from the optic disc on fluorescein angiography. Inflammatory optic neuritis shows contrast enhancement and fluorescein leakage, but there is no fluorescein leakage in the acute phase of LHON 14). Additionally, about 50% of optic neuritis cases present with eye movement pain, whereas LHON is painless. A definitive diagnosis can be made by mitochondrial genetic testing (mt11778, mt14484, mt3460), which can be performed as a send-out test 14). Steroid administration is not only ineffective in LHON patients but also delays definitive diagnosis; therefore, it is essential to include LHON in the differential diagnosis for acute bilateral vision loss in young men.
8. Related Articles (Optic Neuropathy)
Section titled “8. Related Articles (Optic Neuropathy)”Arteritic Anterior Ischemic Optic Neuropathy (AAION)
Compressive Optic Neuropathy
Toxic Optic Neuropathy
Nutritional Deficiency Optic Neuropathy
Leber Hereditary Optic Neuropathy (LHON)
Autosomal Dominant Optic Atrophy (ADOA)
Radiation Optic Neuropathy
Optic nerve diseases and tumors to differentiate
Section titled “Optic nerve diseases and tumors to differentiate”Idiopathic optic neuritis
MS-related optic neuritis
Neuromyelitis Optica Spectrum Disorder (NMOSD)
MOG Antibody-Associated Disease (MOGAD)
References
Section titled “References”- Piccus R, Hansen MS, Hamann S, et al. An update on the clinical approach to giant cell arteritis. Eye 2023.
- Biousse V, Newman NJ. Diagnosis and clinical features of common optic neuropathies. Lancet Neurol 2016;15:1355–67.
- Salvetat ML, Pellegrini F, Spadea L, et al. Non-Arteritic Anterior Ischemic Optic Neuropathy (NA-AION): A Comprehensive Overview. Vision 2023;7:72.
- Beck RW, Gal RL. The Optic Neuritis Treatment Trial: a report on the visual function at 15 years. Ophthalmology 2008;115:1079–82.
- Wingerchuk DM, Banwell B, Bennett JL, et al. International consensus diagnostic criteria for neuromyelitis optica spectrum disorders. Neurology 2015;85:177–89.
- Jeyakumar N, Lerch M, Dale RC, Ramanathan S. MOG antibody-associated optic neuritis. Eye 2024.
- Hathaway JT, Shah MP, Hathaway DB, et al. Risk of nonarteritic anterior ischemic optic neuropathy in patients prescribed semaglutide. JAMA Ophthalmol. 2024;142:740-741. PMID: 38958939. doi:10.1001/jamaophthalmol.2024.2514.
- Thompson AJ, Banwell BL, Barkhof F, et al. Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol 2018;17:162–73.
- Biousse V, Newman NJ. Compressive Optic Neuropathies. J Neuroophthalmol 2015;35 Suppl 1:S67–S74.
- Sanders FWB, Votruba M. Outcomes of idebenone therapy for Leber hereditary optic neuropathy in a cohort of patients from Wales. Eye 2025.
- Newman NJ, et al. Hereditary optic neuropathy: advances in treatment. Eye 2024.
- Danesh-Meyer HV. Radiation-induced optic neuropathy. J Clin Neurosci 2008;15:95–100.
- Toosy AT, Mason DF, Miller DH. Optic neuritis. Lancet Neurol 2014;13:83–99.
- Carelli V, Ross-Cisneros FN, Sadun AA. Mitochondrial dysfunction as a cause of optic neuropathies. Prog Retin Eye Res 2004;23:53–89.
