Excitation Light
Blue light with a wavelength of 465–490 nm is irradiated.
SLO uses a 488 nm laser.
An excitation filter cuts off unnecessary wavelengths.

Fluorescein angiography (FA) is a test in which sodium fluorescein, a fluorescent dye, is administered intravenously, and the fundus is photographed with a fundus camera equipped with special filters to dynamically image the blood circulation of the retina and choroid. It is excellent for evaluating retinal circulation dynamics and the state of the blood-retinal barrier, and is widely used for diagnosis and treatment planning of fundus diseases. It is useful not only for retinal vascular diseases but also for differentiating uveitis, choroidal tumors, and optic disc diseases.
In 1961, Harold R. Novotny and David L. Alvis first reported the original method of FA. Subsequently, from 1967, John Donald McIntyre Gass published systematic FA findings in various fundus diseases, leading to rapid expansion of clinical application.
Sodium fluorescein emits yellow-green fluorescence (520–530 nm) when irradiated with blue excitation light (wavelength 465–490 nm). Scanning laser ophthalmoscopes (SLO) use a 488 nm blue laser. Sodium fluorescein is a water-soluble dye with a molecular weight of 376 Da, and its protein binding rate after intravenous administration is approximately 70–80%. The remaining approximately 20–30% is free form and emits fluorescence. Under normal blood-retinal barrier conditions, even the free form does not leak out of the vessels. When the barrier is disrupted, the dye leaks out of the vessels and is observed as characteristic hyperfluorescent findings.
The blood-retinal barrier (BRB) consists of two layers. The inner barrier is the tight junctions of retinal vascular endothelial cells, and the outer barrier is the tight junctions of retinal pigment epithelium (RPE) cells. When the BRB is disrupted, fluorescein leakage occurs, serving as a diagnostic indicator for various retinal diseases.
Excitation Light
Blue light with a wavelength of 465–490 nm is irradiated.
SLO uses a 488 nm laser.
An excitation filter cuts off unnecessary wavelengths.
Fluorescence Emission
It emits yellow-green fluorescence with an emission wavelength of 520–530 nm.
Free form (approximately 20–30%) is the main source of fluorescence.
Protein-bound form (approximately 70–80%) is less likely to fluoresce.
Barrier Filter
Only transmits fluorescence above 520 nm.
Blocks excitation light to sharpen the fluorescence image.
Extravasation is evidence of barrier breakdown.
It was first reported by Novotny and Alvis in 1961. After 1967, Gass systematized its application to various fundus diseases, and it became a standard diagnostic test for fundus examination worldwide.
FA is widely used for visualization of the retinal vasculature. The main indications are listed below.
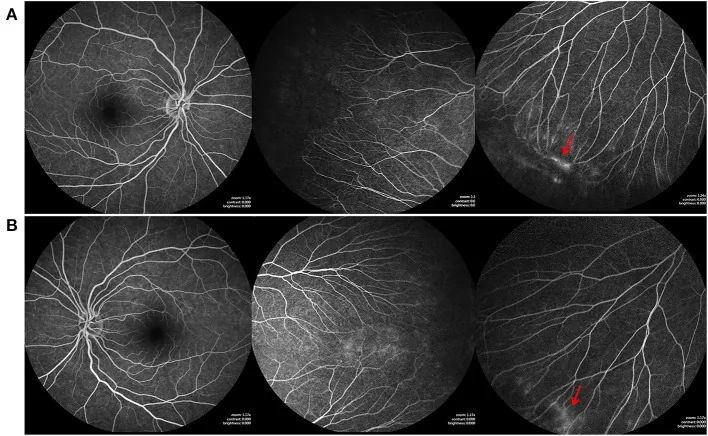
In macular edema following acute retinal necrosis (ARN), FA shows a petaloid leakage pattern, which has been reported to aid in the differential diagnosis of cystoid macular edema (CME) and assessment of treatment efficacy1).
Retinal vascular occlusion may occur during pregnancy, but from the perspective of placental transfer of FA, OCTA is recommended as an alternative examination2).
Before the examination, explain the following and obtain written consent4).
According to the Japanese Ophthalmological Society’s Fundus Angiography Implementation Standards (Revised Edition), the following procedures are performed4).
Patients taking beta-blockers or alpha-blockers have an increased risk of side effects, so this should be identified in advance 4). Skin reaction testing has limited utility, and even a negative result cannot completely rule out serious side effects 4).
Rapidly inject 3–5 mL of 10% fluorescein solution intravenously 4). For children, a dose of 0.1 mg/kg is used as a guide, and for patients with renal impairment, the dose should be half or less of the usual amount 4).
Continuous imaging is performed for about 1 minute immediately after injection, followed by late-phase images at 5 and 10 minutes. Including the time for pupil dilation, the entire procedure takes about 15 to 20 minutes.
FA is observed as multiple phases over time.
FA findings are broadly classified into three categories: hypofluorescence, hyperfluorescence, and vascular morphological abnormalities.
Fluorescence Blockage (Hypofluorescence)
Definition: Hemorrhage, pigmentation, exudates, etc., block background fluorescence.
Features: Well-defined borders, no change in shape over time.
Representative diseases: Subretinal hemorrhage, hard exudates, choroidal nevus.
Filling Defect (Hypofluorescence)
Definition: No or delayed inflow of fluorescent dye due to vascular occlusion.
Features: Capillary non-perfusion areas remain dark throughout the entire course.
Representative diseases: Retinal artery occlusion, avascular areas in diabetic retinopathy.
Fluorescein Leakage (Hyperfluorescence)
Definition: Breakdown of the blood-retinal barrier causes dye to leak out of vessels.
Features: Enlarges over time, borders become indistinct. A petaloid pattern is characteristic of cystoid macular edema 1).
Representative diseases: Macular edema, CNV, retinal vasculitis.
Transmitted Fluorescence (Hyperfluorescence)
Definition: Choroidal fluorescence visible through a defect in the RPE (window defect).
Features: Does not change shape over time, but may show faint staining in the late phase.
Representative diseases: Geographic atrophy, confluent drusen, macular hole, angioid streaks.
Hypofluorescence is further subdivided by cause as follows.
| Type of Hypofluorescence | Cause/Mechanism | Representative Diseases |
|---|---|---|
| Blockage of Fluorescence | Hemorrhage, exudate, or nevus blocks background fluorescence | Subretinal hemorrhage, hard exudates, choroidal nevus |
| Filling Defect: Retinochoroidal Vascular Stenosis | Occlusion or stenosis of large vessels | Internal carotid artery occlusion, Takayasu arteritis |
| Filling defect: retinal vascular occlusion | Arterial/venous occlusion | Retinal artery occlusion (CRAO/BRAO), retinal vein occlusion |
| Filling defect: capillary occlusion | Peripheral circulatory disorder | Diabetic retinopathy (NPDR), Eales disease |
| Filling defect: choroidal circulatory disorder | Choroidal blood flow insufficiency | Vogt-Koyanagi-Harada disease, APMPPE, hypertensive choroidopathy |
| Chorioretinal atrophy | Loss of fluorescence due to tissue atrophy | Macular dystrophy, retinitis pigmentosa, atrophic AMD, pathologic myopia |
| Optic disc hypofluorescence | Ischemia/infiltration of optic nerve tissue | Ischemic optic neuropathy, melanocytoma |
Hyperfluorescence is subdivided by cause as follows.
The following morphological vascular abnormalities are observed.
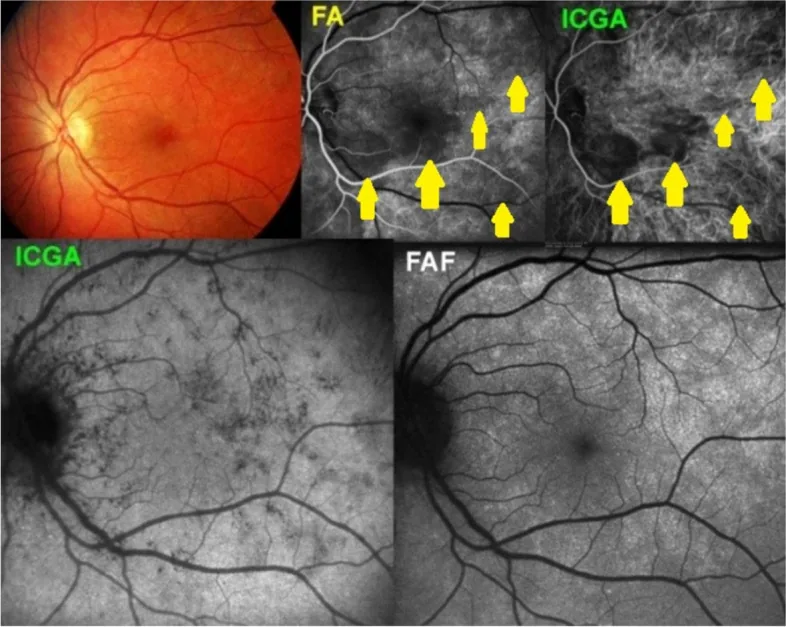
Choroidal vessels are difficult to evaluate because they are obscured by the RPE. ICG angiography (ICGA) is used complementarily to evaluate type 1 MNV (choroidal neovascularization) with fluorescein leakage.
Microaneurysms, non-perfusion areas (NPA), and neovascularization are the main findings. FA helps determine the leakage pattern of macular edema (focal/diffuse/cystoid) and provides the basis for deciding laser photocoagulation sites 3). In the late phase, marked leakage from neovascularization is observed. Ultra-widefield FA improves the accuracy of evaluating peripheral NPA.
FA is essential for assessing CNV activity. Classic CNV (type 2) shows well-defined hyperfluorescence in the early phase and leaks in the late phase. Occult CNV (type 1) is located beneath the RPE and appears as stippled hyperfluorescence or fibrovascular PED in the late phase. Geographic atrophy appears as a window defect throughout the angiogram.
FA is used to evaluate delayed filling, venous dilation and tortuosity, capillary non-perfusion areas, and collateral circulation. An NPA of 10 disc areas or more is considered ischemic and indicates a risk of neovascular glaucoma. It is also important for understanding the leakage pattern of macular edema.
Characteristic inkblot or smokestack leakage points are seen at the level of the RPE. Multiple leakage points suggest chronic CSC. FA and ICGA are used together to determine the treatment area for photodynamic therapy (PDT).
In Vogt-Koyanagi-Harada disease, punctate hyperfluorescence due to multifocal choroidal leakage and dye pooling in areas of serous retinal detachment are observed. In Behçet’s disease, vessel wall staining of retinal vasculitis and non-perfusion areas (NPA) are seen. In Eales disease, peripheral NPA and neovascularization are characteristic.
Data from the Japanese Ophthalmological Society’s Fundus Angiography Implementation Standards (Revised Edition) are shown 4).
Side effects occur at the following frequencies according to severity.
| Severity | Incidence |
|---|---|
| All side effects | 1.1–11.2% |
| Mild | 1.4–8.1% |
| Moderate | 0.2–1.5% |
| Severe | 0.005–0.48% |
| Death | 0.0005–0.002% |
Mild (often resolves spontaneously)
Moderate
Severe (extremely rare)
The diagnosis of anaphylaxis is made when any of the following three criteria are met4).
Anaphylaxis Management Flowchart:
Even after successful treatment, there is a risk of biphasic anaphylaxis (recurrence within 6–8 hours after symptom resolution), so observation for at least 8 hours is necessary, and hospitalization for 24 hours is recommended4).
Differentiation from vasovagal reflex: Vasovagal reflex presents with bradycardia, hypotension, pallor, and cold sweat, but can be differentiated from anaphylaxis by the absence of skin findings (urticaria, flushing). For vasovagal reflex, supine positioning, leg elevation, and fluid resuscitation are effective4).
This is a normal reaction due to fluorescein excretion by the kidneys, and there is no need to worry. Yellowing of the skin resolves in 2–3 hours, and yellow urine resolves by the next day.
Fluorescein crosses the placenta and is detected in breast milk for 72 hours, so administration is generally avoided in pregnant and breastfeeding women2)3). If retinal vascular information is needed, non-invasive OCT angiography (OCTA) is recommended as an alternative2).
Fluorescein sodium is a yellow-red water-soluble dye with a molecular weight of 376 Da. When excited at wavelengths of 465–490 nm (488 nm with SLO), it emits yellow-green fluorescence at 520–530 nm. After intravenous administration, approximately 70–80% binds to plasma proteins (mainly albumin), and about 20–30% fluoresces as free form. It is excreted by the kidneys (cleared within 1–2 days), with minimal hepatic metabolism.
Indocyanine green (ICG) angiography is used complementarily with FA. Their characteristics are shown below.
| Parameter | FA | ICG angiography |
|---|---|---|
| Molecular weight | 376 Da | 775 Da |
| Protein binding rate | Approximately 70–80% | Approximately 98% |
| Main observation target | Retinal vessels | Choroidal vessels |
| Excitation wavelength | 465–490 nm | Approximately 805 nm |
| Fluorescence wavelength | 520–530 nm | Approximately 835 nm (near-infrared) |
| Excretion route | Kidney | Liver |
ICG has a high protein binding rate of 98%, so it hardly leaks out of the choroidal vessels, making it suitable for evaluating choroidal blood flow. ICGA complements FA in the evaluation of polypoidal choroidal vasculopathy (PCV), choroidal hemangioma, and type 1 MNV.
OCTA is a non-invasive test that analyzes the phase information of OCT to visualize the movement of red blood cells. It does not require contrast agents and can separate and depict the retinal vascular plexus into three layers at the capillary level 3). Its usefulness as an alternative to FA in evaluating retinal vessels during pregnancy has been demonstrated 2).
| Item | FA | OCTA |
|---|---|---|
| Contrast agent | Required | Not required |
| Invasiveness | Venipuncture and side effects possible | Non-invasive |
| Dynamic information | Can evaluate leakage and filling delay | Cannot evaluate (structure only) |
| Depth resolution | Two-dimensional only | Layer-by-layer analysis possible |
| Imaging range | Wide angle (up to 200°) | Limited (3–12 mm) |
| Peripheral evaluation | Easy | Difficult |
OCTA cannot detect fluorescence leakage outside blood vessels, so FA remains essential for assessing the activity of macular edema, determining CNV activity (presence of leakage), and evaluating vascular wall inflammation in retinal vasculitis. Using both complementarily enables more precise fundus evaluation.
The observable field of view and characteristics differ depending on the imaging device used.
| Device | Field of view | Main features |
|---|---|---|
| Fundus camera | 55° | Standard, widely used |
| SLO/HRA | 30–102° | High contrast, confocal |
| Optos | 200° | Ultra-widefield single-shot imaging of periphery |
The ultra-widefield imaging device (Optos) can capture the peripheral retina in a single shot, and is useful for evaluating peripheral lesions in diabetic retinopathy and retinal degenerative diseases.
In cystoid macular edema after acute retinal necrosis, it has been suggested that the petaloid leakage pattern on FA may serve as a predictive biomarker for treatment response 1). Research is ongoing to quantify FA dynamic information and utilize it for predicting the efficacy of anti-VEGF therapy and photodynamic therapy.
The introduction of RetCam3 has made FA feasible in children. It is expected to be applied for vascular evaluation in retinopathy of prematurity and pediatric retinal diseases.
With the 200° ultra-widefield FA using Optos, the imaging time for peripheral lesions has been significantly reduced. Its utility has been demonstrated for evaluating peripheral capillary non-perfusion areas in diabetic retinopathy and for extensive assessment of congenital retinal vascular diseases (e.g., FEVR).