Intradermal Nevus
Appearance: Dome-shaped or nodular raised lesion. Little pigment, often flesh-colored to light brown.
Features: May have hair. Often noticed since childhood.
Risk of malignancy: Almost none.
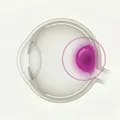
Eyelid nevus is a benign tumor in which immature pigment cells (nevus cells) proliferate from the basal layer of the epidermis into the dermis. The closer to the epidermis, the richer the melanin pigment, and the deeper in the dermis, the less pigment. As the main location of nevus cell nests shifts from superficial to deep layers, they are histologically classified into junctional nevus, compound nevus, and intradermal nevus.
It is the most common eyelid benign tumor by pathological diagnosis, accounting for 14 out of 64 eyes (22%). Many are present from childhood, and history taking is helpful for diagnosis. Among subtypes, intradermal nevus is the most common. The average number of common skin nevi per person is 10 to 401). Nevus of Ota is relatively common in Asians, with a reported frequency of 0.1 to 0.6%2).
The main histological subtypes of nevi occurring on the eyelid are shown below.
Junctional nevus: Occurs in the deep layer of the epidermis at the junction with the dermis. It is a well-defined, flat tumor containing abundant pigment, presenting a strong brown to black color. It may become malignant.
Intradermal nevus: Occurs within the dermis and is the most frequent type. It often presents as raised or nodular lesions with little brown pigmentation. It may have hair. Malignant transformation is extremely rare.
Compound nevus: Has characteristics of both junctional and intradermal nevi, with variable color. It may become malignant.
Blue nevus: Contains melanocytes in the dermis and presents a blue to blue-brown color.
Nevus of Ota: A blue nevus that occurs diffusely in the area of the first and second branches of the trigeminal nerve. It often causes pigmentation of the sclera and iris. It is more common in women and appears unilaterally.
It depends on the subtype. Intradermal nevi do not become malignant, so observation is possible if there are no cosmetic concerns. However, if rapid growth, color change, bleeding, or itching occurs, consult an ophthalmologist. Junctional and compound nevi rarely transform into malignant melanoma, so consider complete excision along with observation.
Most eyelid nevi are asymptomatic. The main complaint is cosmetic (pigmented raised lesion on the face), and pain or visual impairment is usually absent. Patients are often aware of the mass since childhood, and history taking is a clue to diagnosis.
They commonly occur at the eyelid margin between the eyelashes, but may also develop near the lacrimal punctum. Among nevi, intradermal nevus is the most common, where nevus cells in the dermis proliferate and push the epidermis upward in a dome shape. Compound nevi present as brown raised tumors, while junctional nevi present as black flat lesions. Color tends to be darker (black) in superficial layers and lighter in deeper layers.
The clinical features of each subtype are shown below.
Intradermal Nevus
Appearance: Dome-shaped or nodular raised lesion. Little pigment, often flesh-colored to light brown.
Features: May have hair. Often noticed since childhood.
Risk of malignancy: Almost none.
Compound and Junctional Nevus
Appearance: Compound nevus is a brown raised mass. Junctional nevus is black and flat.
Features: Rich in pigment, often black to dark brown. Relatively clear borders.
Risk of malignancy: Rarely transforms into malignant melanoma.
Nevus of Ota
Appearance: Diffuse bluish-gray to bluish-brown pigmentation in the distribution of the first and second branches of the trigeminal nerve.
Features: Pigmentation also in the sclera and iris. More common in women, unilateral. Congenital or adolescent onset.
Risk of malignancy: Risk of uveal melanoma has been reported.
Nevus of Ota may involve pigmentation of the iris and sclera, and an increased risk of pigmentary glaucoma (melanocytic glaucoma) has been noted. Additionally, an association between nevus of Ota and uveal malignant melanoma has been reported9), and regular ophthalmologic follow-up is recommended.
The following factors are involved in the development of eyelid nevus.
The following information is important in the diagnosis of eyelid nevus.
The main diseases that require differentiation from eyelid nevus are listed below.
| Differential Diagnosis | Key Differentiating Features |
|---|---|
| Malignant melanoma | Color variation, irregular borders, rapid change, meets ABCDE criteria |
| Basal cell carcinoma | Central ulceration, pearly luster, rapid growth |
| Seborrheic keratosis | Surface hyperkeratosis/verrucous, elderly |
| Blue nevus | Bluish color, deep dermis |
| Hemangioma | Red color, blanches with pressure |
For clinical differentiation, the ABCDE criteria (asymmetry, border irregularity, color variegation, diameter >6 mm, evolution) are useful. If dermoscopy shows an atypical pattern, malignancy should be suspected 3,4). Lesions present since childhood are likely benign (history is important). Definitive diagnosis is made by histopathological examination. If rapid change occurs, excisional biopsy should be performed without hesitation.
Treatment of eyelid nevus is determined based on subtype and risk of malignant transformation. Treatment strategies by subtype are shown in the table below.
| Subtype | Treatment | Malignant risk |
|---|---|---|
| Intradermal nevus | Shaving (open treatment) if cosmetic concern | Almost none |
| Compound nevus | Complete excision recommended | Rarely malignant melanoma |
| Junctional nevus | Complete excision recommended | Rarely malignant melanoma |
| Blue nevus | Observation or excision | Extremely rare |
| Nevus of Ota | Q-switched laser | Reports of uveal melanoma |
Treatment of intradermal nevus: Since it does not become malignant, surgery is indicated only if the patient desires removal for cosmetic reasons. Often, patients have lived with the nevus as part of their face since childhood and may have little desire for removal. The common surgical technique is partial excision (shaving) of the raised portion, leaving an open wound (open treatment).
Treatment of compound nevus and junctional nevus: Because there is a risk of transformation into malignant melanoma, albeit rare, complete excision is recommended while monitoring the condition. The excised specimen must always be sent for histopathological examination.
Treatment of nevus of Ota: Q-switched ruby laser and Q-switched alexandrite laser have been reported to be effective 5). Laser irradiation selectively destroys dermal melanocytes and improves pigmentation. Multiple sessions are often required.
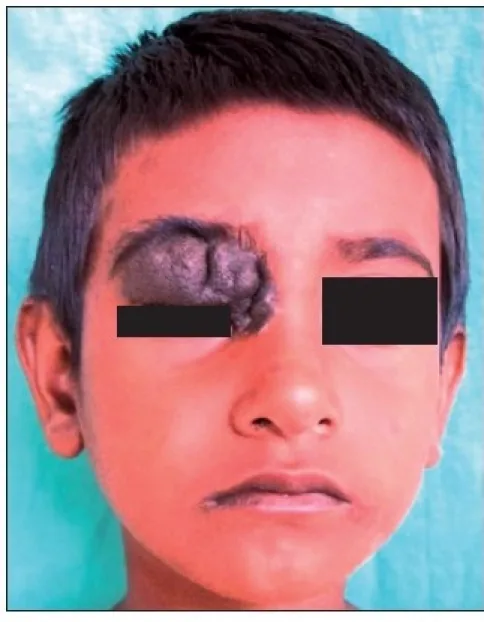
Treatment of congenital giant pigmented nevus: Staged excision and tissue expansion techniques are used 6). When the eyelid is involved, reconstructive surgery becomes complex, so management at a specialized facility is recommended.
For nevus of Ota, Q-switched ruby laser and Q-switched alexandrite laser are considered effective, and multiple sessions can improve pigmentation 5). For common intradermal nevi, compound nevi, and junctional nevi, excision (shaving or complete excision) is the standard treatment, and laser is not the first choice.
Nevus cells are immature pigment cells originating from melanocyte precursors that migrated from the neural crest 1). The histological subtype is determined by the depth of the nevus cell nests. If the nests are localized in the epidermal basal layer, it is a junctional nevus; if in the dermis, an intradermal nevus; and if spanning both, a compound nevus. Cells in superficial layers are rich in melanin, while those in deeper layers have less pigment. This pigment gradient accounts for the differences in macroscopic color among subtypes.
Approximately 80% of acquired nevi harbor BRAF V600E mutations 7). BRAF mutations activate cell proliferation signals (MAPK pathway), but alone they do not lead to malignancy and often cause oncogene-induced senescence. Transformation to malignant melanoma occurs when additional mutations such as TERT promoter mutations or CDKN2A deletions accumulate alongside BRAF mutations 7). The estimated risk of a common mole transforming into malignant melanoma is less than 0.0005% per mole per year, which is extremely low 1).
Nevus of Ota results from hyperplasia of dermal melanocytes. Proliferation of melanocytes scattered in the dermis of the first and second branches of the trigeminal nerve leads to blue to blue-gray pigmentation. Pigmentation may occur not only on the eyelid skin but also on the sclera, iris, and fundus. In Caucasian patients with oculodermal melanocytosis, the lifetime incidence of uveal melanoma is reported to be significantly higher 9), and regular fundus and intraocular pressure examinations are recommended.
Deep learning-based image diagnosis systems for skin tumors have been developed, and classification accuracy at the level of a dermatologist has been reported 8). Combining with dermoscopic images is expected to further improve the accuracy of differentiating nevi from malignant melanoma. Application to the eyelid area remains a future challenge.
A systematic review of staged excision using tissue expanders for congenital giant pigmented nevi has reported improvements in functional and cosmetic reconstruction 6). In cases involving the eyelid, technical refinements are progressing to minimize impact on visual function while performing excision and reconstruction.
A study of white patients with oculodermal melanocytosis reported a lifetime incidence of uveal melanoma of approximately 1 in 400, suggesting the usefulness of regular fundus examinations in patients with nevus of Ota 9).
Tsao H, Bevona C, Goggins W, et al. The transformation rate of moles (melanocytic nevi) into cutaneous melanoma. Arch Dermatol. 2003;139(3):282-288.
Hidano A, Kajima H, Ikeda S, et al. Natural history of nevus of Ota. Arch Dermatol. 1967;95(2):187-195.
Abbasi NR, Shaw HM, Rigel DS, et al. Early diagnosis of cutaneous melanoma: revisiting the ABCD criteria. JAMA. 2004;292(22):2771-2776.
Argenziano G, Soyer HP, Chimenti S, et al. Dermoscopy of pigmented skin lesions: results of a consensus meeting. J Am Acad Dermatol. 2003;48(5):679-693.
Chan HH, Kono T. The use of lasers and intense pulsed light sources for the treatment of pigmentary lesions. Skin Therapy Lett. 2004;9(8):5-7.
Kishi K, Matsuda N, Kubota Y, et al. Systematic review: staged excision and tissue expansion for giant congenital melanocytic nevi. J Plast Reconstr Aesthet Surg. 2022;75(1):1-15.
Shain AH, Yeh I, Kovalyshyn I, et al. The genetic evolution of melanoma from precursor lesions. N Engl J Med. 2015;373(20):1926-1936.
Esteva A, Kuprel B, Novoa RA, et al. Dermatologist-level classification of skin cancer with deep neural networks. Nature. 2017;542(7639):115-118.
Singh AD, De Potter P, Fijal BA, et al. Lifetime prevalence of uveal melanoma in white patients with oculo(dermal) melanocytosis. Ophthalmology. 1998;105(1):195-198.