Typical Type
Patient population: Predominantly men in their 30s–40s. Usually unilateral.
Pathology: Localized serous retinal detachment in the macula.
Course: Most cases resolve spontaneously within 3–4 months. Visual prognosis is generally good.

Central Serous Chorioretinopathy (CSC) is a disease in which serous retinal detachment (SRF) occurs in the macula. Fluorescein angiography (FA) shows leakage at the level of the retinal pigment epithelium (RPE). It is considered the fourth most common retinal disease after age-related macular degeneration, diabetic retinopathy, and retinal vein occlusion. It was first described by von Graefe in 1866 as “central recurrent retinitis,” and the current name was given by Gass et al. in 1967. In Japan, it was previously often used to refer only to typical cases (so-called central retinitis) with localized retinal detachment in the macula, but it is now classified into three subtypes. Many cases heal spontaneously, but in cases where SRF persists, visual prognosis may worsen.
Epidemiologically, the incidence is 9.9 per 100,000 men and 1.7 per 100,000 women, with a male-to-female ratio of about 8:14). In a study of 250 eyes, the mean age was 46.6 years, and 88.4% were male3).
This disease has the following three subtypes.
Typical Type
Patient population: Predominantly men in their 30s–40s. Usually unilateral.
Pathology: Localized serous retinal detachment in the macula.
Course: Most cases resolve spontaneously within 3–4 months. Visual prognosis is generally good.
Chronic Type
Patient population: More common in older adults. Often bilateral. Recently increasing.
Pathophysiology: Extensive RPE damage. Often recurs.
Course: SRF persists for 6 months or longer. Requires active treatment 5).
Bullous retinal detachment
Patient population: Common in cases with high-dose steroid use. Corresponds to what was previously called multiple posterior pole retinal pigment epitheliopathy in Japan.
Pathophysiology: Severe form with bullous retinal detachment. Accompanied by large RPE detachment (PED) and multiple white spots.
Course: The extent of SRF often reaches the lower fundus, and visual prognosis may be poor.
The recurrence rate is high, up to 50% within one year. This condition is considered part of the pachychoroid disease spectrum (a group of diseases characterized by choroidal thickening) 3)9).
In typical cases, spontaneous remission often occurs within 3 to 4 months, and visual prognosis is generally good. However, the recurrence rate reaches up to 50% within one year. If SRF persists for 6 months or longer, it is considered chronic and requires treatment such as PDT. In the pachychoroid group, the spontaneous resolution rate is lower (28.8%) than in the non-pachychoroid group (48%), and the recurrence rate is higher (31.2% vs. 10.4%), so caution is needed 3).
When subretinal fluid is absorbed, central scotoma and metamorphopsia improve relatively early. On the other hand, micropsia and decreased contrast sensitivity may persist. In the chronic type, recurrences are frequent, and many cases have poor visual acuity. Typical cases are often unilateral, while chronic type and bullous retinal detachment are often bilateral.
The following findings are observed with slit-lamp microscopy and various imaging tests.
In the chronic type, pigment epithelial damage is extensive, and retinal detachment extending downward with atrophic bands of the pigment epithelium may be observed. In bullous retinal detachment, the retinal detachment is severe and often extends to the lower fundus, accompanied by large pigment epithelial detachments and multiple white plaques.
The underlying cause of this condition remains unknown, but increased permeability of choroidal vessels is considered the essence. Stress and steroids are known to be related to the onset and exacerbation of this condition. The following risk factors have been reported.
| Risk factor | Strength of association | Notes |
|---|---|---|
| Steroids | OR 37.11) | All routes of administration |
| Type A personality | Moderate | Stress-related |
| Pregnancy | 0.008%/year7) | Frequent spontaneous remission after delivery |
| OSA | 61% of patients9) | Sympathetic hyperactivity |
| PDE5 inhibitors | Case report8) | Resolved after discontinuation |
Onset is explained by the multi-hit theory9). It is thought to occur in three stages: (1) anatomical predisposition (short axial length, scleral thickening, asymmetry of vortex vein drainage, etc.), (2) triggering events (steroids, stress, OSA, etc.), and (3) decompensation.
Steroids are the greatest risk factor for this disease, and the risk increases with all routes of administration, including eye drops, inhalation, injection, and oral intake. The odds ratio has been reported to be extremely high at 37.11). If diagnosed with this disease and currently using steroids, consult with your doctor and consider reducing or discontinuing them as much as possible. If steroids are being administered for a systemic disease, it is necessary to coordinate with other departments on a case-by-case basis.
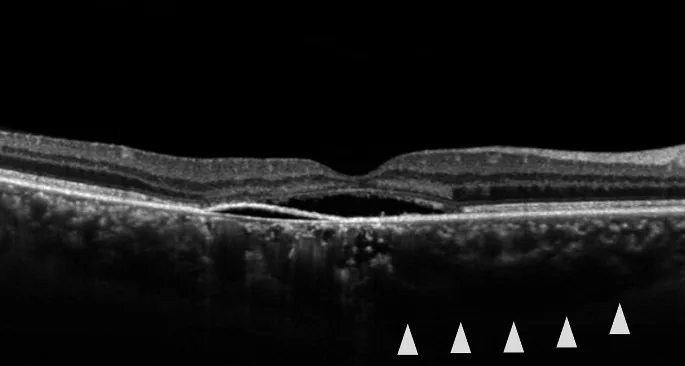
The diagnosis of this disease is based on a combination of FA and OCT. The main features of the examinations are shown below.
| Examination | Characteristic Findings | Main Role |
|---|---|---|
| FA | Punctate fluorescein leakage | Identification of leakage point and treatment planning |
| IA (ICG) | Choroidal hyperfluorescence | Evaluation of choroidal abnormalities and differential diagnosis |
| OCT | Visualization of SRF and pigment epithelial detachment | Follow-up and quantification |
Details of each examination are as follows.
There is no established drug therapy for this condition. Laser photocoagulation of fluorescein leakage points on FA is currently the only generally accepted treatment, and the treatment strategy is determined by the type, stage, and location of leakage points.
Typical acute cases have a tendency for spontaneous remission. In the acute phase of a first episode, natural absorption of subretinal fluid is awaited for 4 to 6 months. In typical cases, complete absorption often occurs within 3 to 4 months, and visual prognosis is good. The spontaneous resolution rate is 48% in the non-pachychoroid group, compared to 28.8% in the pachychoroid group3).
If symptoms are severe and the patient desires early improvement, photocoagulation may be indicated even in acute typical cases. If the patient is using steroids, reduce or discontinue as much as possible. If steroids are administered for systemic disease, manage in collaboration with other departments on a case-by-case basis.
In cases where detachment persists for more than 4 to 6 months after onset, recurrent cases, severe cases, or cases where the other eye has residual visual impairment due to this disease (chronic type, bullous retinal detachment), early absorption of subretinal fluid through treatment is necessary. This is because macular damage leads to poor visual prognosis.
Indicated when the leakage point is at least 500 μm from the fovea and outside the foveal avascular zone. Directly coagulate the leakage point identified on FA (200 μm spot size, 0.2 seconds, 70–120 mW). The coagulation intensity should be mild, producing a faint gray-white coagulation spot in the deep retina. Even leakage between the optic disc and macula is not problematic with mild coagulation. If leakage is within a pigment epithelial detachment, only the leakage point needs to be coagulated.
When photocoagulation is effective, retinal detachment resolves within a few weeks. However, improvement in subjective symptoms often takes longer. Complications include accidental foveal irradiation and postoperative choroidal neovascularization (CNV) (more likely with long wavelength, short duration, small spot coagulation).
A 577 nm yellow laser is used. Irradiation is performed with settings of 240 mW, 200 μm spot size, 200 ms, and 5% duty cycle4). Since energy is selectively delivered to the RPE, no scar is formed. It can be applied directly under the fovea.
Bodea F et al. (2024) reported that in patients with central serous chorioretinopathy treated with MPLT, SRF decreased at 2 weeks and resolved at 6 weeks after treatment4).
For this condition during pregnancy, micropulse laser is considered the only safe treatment option7).
Observation
Indications: First-line treatment for acute typical cases.
Duration: 4–6 months. Wait for spontaneous absorption.
Note: During steroid use, consider tapering or discontinuing in coordination with other departments.
PDT (Photodynamic Therapy)
Indications: First-line treatment for chronic central serous chorioretinopathy.
Method: Half-dose (half-fluence) PDT is recommended.
Evidence: Efficacy has been demonstrated in the PLACE and SPECTRA trials (12-week SRF resolution rate 78% vs. eplerenone 17%)9).
Micropulse Laser
Features: No scarring, can be applied directly under the fovea.
Advantage: The only treatment that can be used during pregnancy7).
Effect: SRF reduction at 2 weeks, resolution at 6 weeks4).
Used in cases where laser photocoagulation is not possible, such as when the leakage point is in the foveal avascular zone or in chronic types. Half-dose PDT (half the usual dose) or half-fluence PDT (half laser output) using verteporfin (Visudyne) is recommended as first-line treatment for chronic central serous chorioretinopathy9).
The results of major clinical trials are as follows9).
Mineralocorticoid receptor antagonist. Used at 50 mg/day. In the VICI trial (multicenter randomized double-blind placebo-controlled trial), eplerenone did not show a significant difference in BCVA improvement compared to placebo after 12 months of treatment9). It is considered less effective than PDT, and there are case reports of no response even after 10 months of treatment5).
A new treatment involving 6-minute irradiation with 590 nm yellow LED and 625 nm red LED5). A case has been reported where, in chronic central serous chorioretinopathy with serous pigment epithelial detachment unresponsive to eplerenone, best-corrected visual acuity improved from 20/80 to 20/25 and central retinal thickness decreased from 752 μm to 296 μm (at 1 month)5).
The incidence of this disease during pregnancy is reported to be 0.008% per year 7). Most cases resolve spontaneously within 3 months after delivery. If treatment is necessary, micropulse laser therapy (MPLT) is considered the only safe option during pregnancy, and it can be applied directly under the fovea without scarring 7).
For typical acute cases, observation for 4 to 6 months is the standard. For chronic cases with SRF persisting for more than 6 months, half-dose PDT is the first-line treatment 9). The SPECTRA trial showed a significant difference in complete SRF resolution at 12 weeks: 78% in the PDT group versus 17% in the eplerenone group. If the leakage point is outside the foveal avascular zone, laser photocoagulation (mild coagulation) is used; for points directly under the fovea or during pregnancy, micropulse laser is chosen. Eplerenone did not show a significant difference in BCVA improvement compared to placebo in the VICI trial 9).
The pathogenesis of this disease is explained by a complex mechanism centered on increased permeability of choroidal vessels. Based on IA studies, it is now thought that the primary lesion is in the choroid, with secondary RPE damage. Why the choroid is affected remains largely unknown. Stress and steroids are known to be associated with the onset and exacerbation of this disease.
On FA, this is shown as leakage of fluorescein from the pigment epithelium and pooling of dye in the subretinal space. OCT studies have shown that the choroid is thickened in this disease compared to normal eyes, providing morphological evidence of choroidal abnormality.
Cheung CMG et al. (2025) explain the pathogenesis of this disease using a multi-hit theory 9).
CSC eyes have significantly thicker anterior and posterior sclera compared to normal eyes 9). Since the vortex veins penetrate the sclera obliquely (intrascleral course of about 4 mm), scleral thickening may increase venous outflow resistance and cause choroidal congestion 9). In 62% of CSC eyes, loculation of fluid in the suprachoroidal space is observed, and 19% show ciliochoroidal effusion 9). In steroid-induced CSC, the sclera is thinner than in idiopathic CSC, suggesting a different role of the sclera in the pathology 9).
A new classification based on multimodal imaging has been proposed 9).
Within each category, there are subcategories of primary (first episode), recurrent, and resolved. Complex CSC has a higher risk of developing choroidal neovascularization (MNV) compared to Simple CSC. The agreement among 10 retinal specialists was κ=0.57 (moderate), and classification continues to be refined 9).
Pachydrusen are observed in over 40% of CSC patients 9). CSC eyes with pachydrusen have more extensive RPE abnormalities, and their morphology and distribution differ from conventional soft drusen. An association between Haller layer thickening and choriocapillaris thinning has been shown 9).
PBM is attracting attention as a new treatment that does not use verteporfin. It involves irradiation with a combination of 590 nm yellow LED and 625 nm red LED 5). It is expected as an alternative treatment in situations where verteporfin supply is a concern.
The presence or absence of pachychoroid findings is becoming increasingly important as a prognostic factor for this disease. Compared to the non-pachychoroid group, the pachychoroid group has a higher recurrence rate (31.2% vs 10.4%) and a lower spontaneous resolution rate (28.8% vs 48%) 3). The presence or absence of pachychoroid findings is an important indicator in determining treatment strategy.
Reports of onset and recurrence of this disease after COVID-19 vaccination have been accumulating 6). It has been suggested that vaccine-induced activation of the HPA axis and steroid hormone-like effects may be involved.