Choroidal Nevus
1. What is a choroidal nevus?
Section titled “1. What is a choroidal nevus?”A choroidal nevus is a pigmented lesion caused by benign proliferation of melanocytes in the choroid. It appears as a gray-black, roundish subretinal lesion on fundus examination and usually follows a stationary (non-growing) course.
The prevalence is about 5% in Westerners and about 0.3% in Japanese, with a relatively high distribution in the posterior pole. It may also be found near the equator or the far periphery near the ora serrata. In many cases, it is discovered incidentally during routine eye examinations in adults or during evaluation of other diseases.
The frequency of malignant transformation is reported as 0.5–3% per 10 years in Western reports. It is considered even rarer in Japanese, but if malignant transformation occurs, it is treated as choroidal malignant melanoma (choroidal melanoma). About 10% of choroidal melanomas are reported to arise from a preexisting nevus.
A choroidal nevus is a benign pigmented lesion and is usually not a problem. However, if multiple risk factors such as thickness or fluid accumulation are present, regular follow-up is necessary. Follow your doctor’s instructions for regular check-ups, and if you experience changes in vision, floaters, or flashes of light, seek medical attention promptly.
2. Main symptoms and clinical findings
Section titled “2. Main symptoms and clinical findings”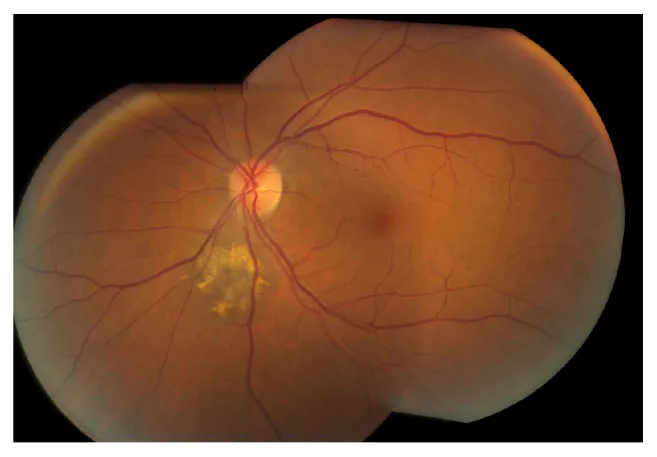
Subjective symptoms
Section titled “Subjective symptoms”The majority of choroidal nevi are asymptomatic and discovered without subjective symptoms. If the lesion extends to the macula or near the optic disc, or if subretinal fluid is present, the following symptoms may appear.
- Decreased vision and metamorphopsia: when the macula is involved
- Photopsia: due to mechanical stimulation of the retina
- Floaters: Rarely occur due to vitreous involvement
Clinical Findings
Section titled “Clinical Findings”Typical findings of choroidal nevus are as follows:
- Gray-black, roundish subretinal elevation
- Diameter usually within 3 optic disc diameters (DD)
- Height (thickness) less than 2 mm, low profile, ≤1/5 of diameter
- Relatively well-defined borders
- Usually stationary (non-progressive) course
Orange pigment (lipofuscin deposition) is an important predictor of malignant transformation. It is deposition of aging pigment produced by the RPE (retinal pigment epithelium) and appears as hyperautofluorescence on fundus autofluorescence.
Presence of subretinal fluid is a concerning finding and suggests disruption of the RPE barrier. If subretinal fluid is present, further evaluation for malignant transformation is necessary.
Risk Assessment for Malignant Transformation Using TFSOM-UHHD
Section titled “Risk Assessment for Malignant Transformation Using TFSOM-UHHD”To differentiate choroidal nevus from choroidal melanoma, the TFSOM-UHHD criteria, which evaluate the following 8 risk factors, are used.
| Factor | Criteria | Significance |
|---|---|---|
| Thickness | Greater than 2 mm | Increase in tumor volume |
| Fluid | Presence of subretinal fluid | RPE barrier breakdown |
| Symptoms | Photopsia and floaters | Indicator of tumor activity |
| Orange pigment | Lipofuscin deposition | Tumor metabolic activity |
| Margin | Within 3 mm of the optic disc | Risk of optic disc infiltration |
| Ultrasound Hollow (hypoechoic) | Internal hypoechogenicity | Tumor vascular density |
| Halo absent | No halo (light ring) | Absence of benign indicator |
| Drusen absent | No drusen | Absence of chronic changes |
The relationship between the number of risk factors and the probability of growth within 5 years is shown below.
| Number of risk factors | 5-year growth probability |
|---|---|
| 0 | Approximately 3% |
| 1 | Approximately 38% |
| 2 or more | >50% |
3. Causes and Risk Factors
Section titled “3. Causes and Risk Factors”Pathogenesis
Section titled “Pathogenesis”Choroidal nevus is a benign proliferation of melanocytes in the choroid, and the exact cause is unknown. It often occurs sporadically.
Risk Factors for Malignant Transformation
Section titled “Risk Factors for Malignant Transformation”The following systemic and clinical factors are thought to increase the risk of malignant transformation.
- Light-colored iris and fair skin (Caucasian/Northern European descent)
- Congenital ocular melanocytosis (ocular disease associated with nevus of Ota)
- Family history of uveal melanoma
- BAP1 tumor predisposition syndrome (germline mutation of BRCA1-associated protein 1)
Each factor of TFSOM-UHHD reflects the local risk of malignant transformation.
- Orange pigment (lipofuscin): Accumulation of aging pigment from RPE indicates tumor activity
- Subretinal fluid: Suggests disruption of the RPE barrier by the tumor
- Thickness ≥2 mm: Reflects increase in tumor volume
- Tumor contact with the optic disc: Increased risk of optic nerve invasion
- Internal low reflectivity on ultrasound: Suggests presence of intratumoral vessels
Genetic Abnormalities and Malignant Transformation
Section titled “Genetic Abnormalities and Malignant Transformation”Malignant transformation to choroidal melanoma involves accumulation of genetic abnormalities. Main associated abnormalities include monosomy 3 and BAP1 mutations. These mutations are frequently detected in choroidal melanoma but are usually not found in benign nevi.
4. Diagnosis and Examination Methods
Section titled “4. Diagnosis and Examination Methods”Fundus Examination and Fundus Photography
Section titled “Fundus Examination and Fundus Photography”Fundus examination (ophthalmoscopy, wide-field fundus photography) is the basis for screening and follow-up. Serial fundus photography is essential for determining tumor growth, and wide-field fundus photography (e.g., Optos) is also useful.
Optical Coherence Tomography (OCT)
Section titled “Optical Coherence Tomography (OCT)”OCT is excellent for visualizing subretinal fluid and RPE changes. EDI-OCT (enhanced depth imaging OCT) allows evaluation of choroidal structure, and the following findings are observed:
- Optical choroidal shadowing
- Presence of subretinal fluid
- RPE changes (atrophy, elevation)
- Subretinal hyperreflective material (SRHM)
Fundus Autofluorescence
Section titled “Fundus Autofluorescence”Orange pigment (lipofuscin) appears as brighter hyperfluorescence than drusen. It is useful for evaluating lipofuscin deposition and helps assess the risk of malignant transformation.
Fluorescein Angiography (FA)
Section titled “Fluorescein Angiography (FA)”FA (fluorescein angiography) shows a hyperfluorescent pattern. Blocking fluorescence due to pigment and hyperfluorescence due to lipofuscin deposition are mixed.
Ultrasonography (B-mode, Color Doppler)
Section titled “Ultrasonography (B-mode, Color Doppler)”B-mode ultrasound is used to measure tumor height and evaluate internal echo patterns. Color Doppler ultrasound can confirm the presence or absence of pulsatile blood flow at the tumor base. Choroidal nevi are often color Doppler negative (no tumor blood flow), while melanomas are often color Doppler positive. This finding is an important distinguishing feature between nevi and melanomas.
Differential Diagnosis of Choroidal Tumors (Three Major Diseases)
Section titled “Differential Diagnosis of Choroidal Tumors (Three Major Diseases)”The characteristics of the main differential diagnoses of choroidal tumors are shown below.
| Malignant Melanoma | Hemangioma | Metastatic Tumor | |
|---|---|---|---|
| Color | Black, gray, brown | Orange-red | Yellow-white |
| Shape | Tall | Fusiform | Plate-like |
| Retinal detachment | None to moderate | None to mild | Marked |
| Growth | Relatively slow | None | Fast |
Other differential diagnoses include congenital hypertrophy of the retinal pigment epithelium (CHRPE), choroidal hemorrhage, and melanocytoma.
It varies greatly depending on the number of risk factors. Among the 8 items of TFSOM-UHHD, if there are 0 risk factors, the probability of growth within 5 years is about 3%. However, with 1 risk factor it rises to about 38%, and with 2 or more it exceeds 50%. Regular follow-up with ultrasound and fundus photography is important, and if enlargement is observed, promptly switch to treatment for choroidal malignant melanoma.
5. Standard Treatment
Section titled “5. Standard Treatment”The basic treatment for choroidal nevus is observation. Currently, there is no drug therapy or surgical indication for the nevus itself, and regular fundus examinations are performed to monitor for enlargement of the lesion.
Low-risk lesions (no risk factors, small nevus)
Section titled “Low-risk lesions (no risk factors, small nevus)”Small nevi without risk factors according to TFSOM-UHHD are managed as low risk.
- Observation interval: Regular fundus examination every 12–24 months
- Record growth using fundus photography and B-mode ultrasound
- If no abnormal findings, continue follow-up at the same interval
Intermediate/High-Risk Lesions (with risk factors)
Section titled “Intermediate/High-Risk Lesions (with risk factors)”If one or more TFSOM-UHHD risk factors are present, careful regular observation is performed.
- Re-examine 3 months after initial examination (check for growth)
- Thereafter, continue every 6 months with fundus photography and ultrasound for life
- For small tumors with 3 or more risk factors, consider prompt treatment initiation without waiting for growth documentation
When Growth Is Confirmed
Section titled “When Growth Is Confirmed”If tumor growth is confirmed during follow-up, the treatment strategy is changed to that for choroidal malignant melanoma. Specific treatment options are determined based on tumor size and visual function, and include radiation therapy (plaque brachytherapy, proton beam therapy) or enucleation (see the article on choroidal and ciliary body melanoma for details).
The observation interval varies depending on the risk. For low-risk lesions without risk factors, an interval of once every 12 to 24 months is standard. For intermediate- or high-risk lesions with risk factors, re-examination is performed after the first 3 months, followed by lifelong fundus examination and ultrasound every 6 months. Continuing regular observation is most important.
6. Pathophysiology and Detailed Mechanisms
Section titled “6. Pathophysiology and Detailed Mechanisms”Mechanism of Benign Proliferation
Section titled “Mechanism of Benign Proliferation”A choroidal nevus is a lesion caused by localized benign proliferation of choroidal melanocytes. Normal choroidal melanocytes originate from neural crest cells and have pigment-producing ability. In the benign proliferation stage, cell morphology is preserved, and invasive growth or metastasis does not occur.
Molecular Mechanisms of Malignant Transformation
Section titled “Molecular Mechanisms of Malignant Transformation”Malignant transformation from choroidal nevus to melanoma requires the accumulation of multiple genetic abnormalities.
- GNAQ/GNA11 mutations: Initiating mutations detected in about 90% of choroidal melanomas. They cause constitutive GTP-binding activation, persistently activating multiple signaling pathways such as the MAPK pathway.
- Monosomy 3: A secondary mutation strongly associated with metastatic risk.
- BAP1 mutations: Associated with the highest metastatic risk. Also known as the causative gene for BAP1 tumor predisposition syndrome.
- SF3B1 mutations: Moderate metastatic risk, characterized by late metastasis.
- EIF1AX mutations: Lowest metastatic risk.
These malignant mutations are usually not found in the nevus stage. It is thought that the accumulation of genetic abnormalities leads to a continuous change from benign to malignant.
Biological Significance of Risk Factors
Section titled “Biological Significance of Risk Factors”Each item of TFSOM-UHHD reflects the following pathology.
Orange pigment (lipofuscin): An aging pigment produced by the RPE. It reflects the metabolic activity of the tumor and the interaction between the tumor and RPE; accumulation increases with higher tumor activity.
Subretinal fluid: A finding suggesting disruption of the RPE barrier by the tumor. When the tumor grows and becomes active, the RPE pump function decreases, and fluid accumulates under the retina.
Internal low reflectivity on ultrasound: Suggests high density of intratumoral vessels. Choroidal melanoma often has abundant intratumoral vessels and shows internal low reflectivity. Benign nevi usually have a uniform, high-reflectivity internal echo pattern.
Color Doppler positive (pulsatile flow): Indicates that the tumor has its own blood supply. Nevi are often color Doppler negative, which is an important distinguishing feature from melanoma.
7. Latest Research and Future Perspectives
Section titled “7. Latest Research and Future Perspectives”Validation of TFSOM-UHHD Criteria Accuracy
Section titled “Validation of TFSOM-UHHD Criteria Accuracy”Validation studies of the sensitivity and specificity of the TFSOM-UHHD criteria are ongoing, and improvements in clinical accuracy are expected. In the future, more precise risk stratification incorporating a 5-year risk calculation model may be realized.
Application of OCT Angiography
Section titled “Application of OCT Angiography”Non-invasive visualization of intratumoral vessels using OCT angiography (OCTA) is being studied. Combined with color Doppler ultrasound, it is expected to further improve the accuracy of differentiating nevi from melanoma.
Quantitative Assessment of Orange Pigment
Section titled “Quantitative Assessment of Orange Pigment”Development of a quantitative assessment method for lipofuscin (orange pigment) using fundus autofluorescence is progressing. The goal is to improve risk prediction accuracy by quantifying the amount of pigment.
Risk Stratification Using Molecular Biomarkers
Section titled “Risk Stratification Using Molecular Biomarkers”Research on non-invasive genomic assessment using liquid biopsy (e.g., circulating tumor DNA in blood) is advancing. In the future, it may be combined with fundus findings for early detection of malignant transformation.
8. References
Section titled “8. References”- Finger PT. Laser treatment for choroidal melanoma. Surv Ophthalmol. 2023;68(2):211-224.
- Qureshi MB, Lentz PC, Xu TT, White LJ, Olsen TW, Pulido JS, et al. Choroidal Nevus Features Associated with Subspecialty Referral. Ophthalmol Retina. 2023;7(12):1097-1108. PMID: 37517800.
- Chien JL, Sioufi K, Surakiatchanukul T, Shields JA, Shields CL. Choroidal nevus: a review of prevalence, features, genetics, risks, and outcomes. Curr Opin Ophthalmol. 2017;28(3):228-237. PMID: 28141766.
