Stages 0–1
Stage 0 (Normal): Blurring of the nasal, superior, and inferior borders. Radial NFL.
Stage 1 (Very early): Obscuration of the nasal disc border. No elevation of the border. Subtle grayish halo. Concentric or radial choroidal folds.

Papilledema is bilateral optic disc swelling due to elevated intracranial pressure (ICP). The term “papilledema” (choked disc) is reserved for ICP elevation, while other causes of disc swelling are termed “optic disc edema.” Increased pressure in the subarachnoid space around the optic nerve compresses the nerve, leading to axoplasmic flow stasis and disc swelling.
The annual incidence of IIH is 1.15 per 100,000 (US data 1997–2016, female 1.97 vs male 0.36) 2), with the highest rate in ages 18–44 (2.47 per 100,000). Prevalence in US women aged 18–55 is 3.44 per 10,000 (95% CI 2.61–5.39) 3). By race, incidence per 100,000 is Black 2.05 > White 1.04 > Hispanic 0.67 > Asian/Pacific Islander 0.16 2), generally consistent with obesity prevalence geographically (Moran I=0.20, P=0.03) 3).
Normal ICP is <250 mmH2O in adults and <280 mmH2O in children. Fulminant IIH (FIH) occurs in 2–3% of all IIH patients and is an emergency characterized by rapid visual loss within 4 weeks of symptom onset. 1)
Papilledema is a term limited to elevated ICP, while optic disc swelling from other causes (e.g., optic neuritis, ischemia, infiltrative lesions) is distinguished as optic disc edema. This distinction is important because it directly links to identifying the cause and selecting appropriate treatment.
Acute papilledema is often asymptomatic in the early stage, and central visual function (best-corrected visual acuity, color vision) is usually preserved.
In children, elevated intracranial pressure is often discovered due to esotropia associated with bilateral abducens nerve palsy.
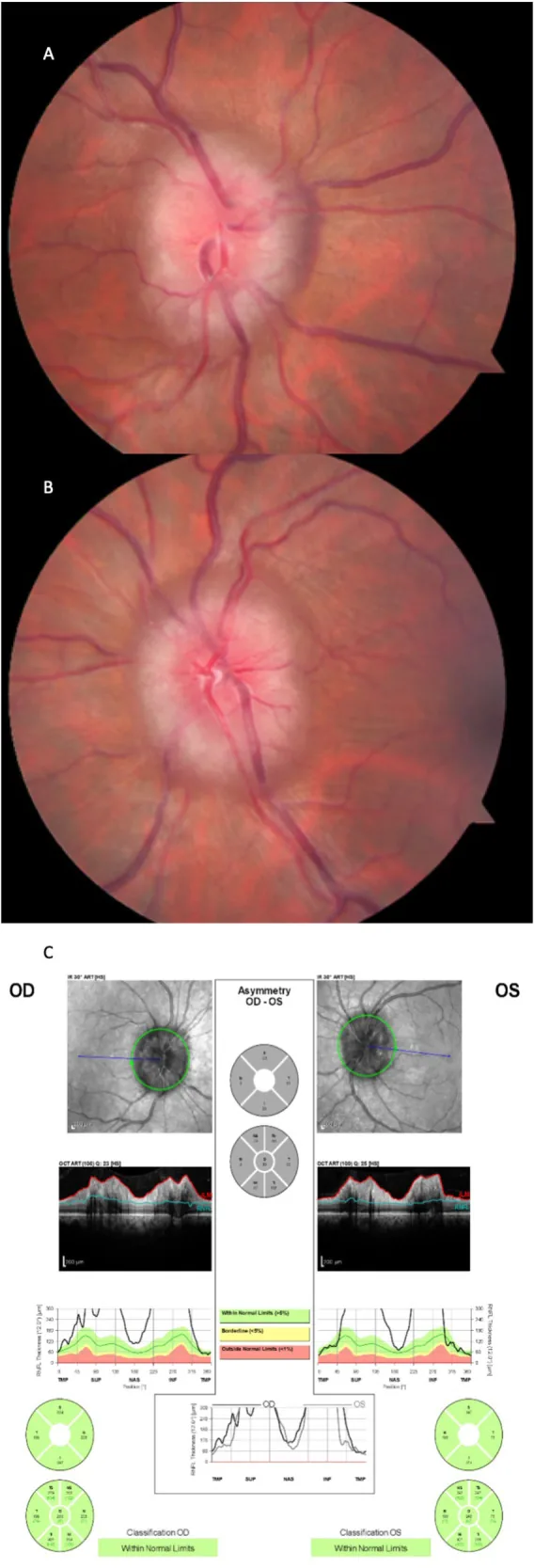
Fundus findings in acute papilledema:
Additional findings in chronic papilledema: Optic disc pallor, gliosis, optociliary shunt vessels, refractive bodies. In the chronic phase, dead nerve fibers do not swell, so edema may resolve even if elevated ICP persists (atrophic end-stage).
If there is bilateral optic disc redness with good visual acuity, always consider papilledema in the differential diagnosis. In papilledema, visual acuity remains nearly normal until the end stage.
The severity of papilledema is assessed using the Frisén scale (stages 0–5) and used as an indicator of visual prognosis.
Stages 0–1
Stage 0 (Normal): Blurring of the nasal, superior, and inferior borders. Radial NFL.
Stage 1 (Very early): Obscuration of the nasal disc border. No elevation of the border. Subtle grayish halo. Concentric or radial choroidal folds.
Stages 2–3
Stage 2 (early): Blurring of all borders. Elevation of the nasal border. Complete peripapillary halo.
Stage 3 (moderate): Increased optic disc diameter. Partial obscuration of segments of major vessels. Finger-like extensions at the outer edge of the halo.
Stages 4–5
Stage 4 (marked): Elevation of the entire optic disc. Complete obscuration of major vessel segments on the disc.
Stage 5 (severe): Dome-shaped protrusion. Halo narrow with smooth borders. Obliteration of the physiologic cup.
TVO is a characteristic symptom of papilledema, but it is not a direct predictor of vision loss. However, if intracranial hypertension persists for a long time, it can progress from severe peripheral visual field loss to central vision loss. If TVO occurs frequently, it is important to promptly see a specialist and investigate the cause.
The rate of papilledema formation depends on the rate of intracranial pressure increase. With gradual ICP elevation, it may appear over several weeks; with rapid elevation, it can appear within hours to one day.
In cases of IIH, a 5–10% weight loss improves ICP symptoms and papilledema. Normalization of ICP (remission) requires a 24% reduction in body weight (per protocol analysis of IIHWT) 15). If lifestyle modification alone is insufficient, bariatric surgery is an option. Weight management is part of conservative treatment and may need to be combined with medication or surgery depending on the degree of papilledema and visual dysfunction.
Evaluation proceeds in the following order: ophthalmoscopy to confirm bilateral papilledema → fluorescein angiography → OCT → CT/MRI plus contrast-enhanced MRV → lumbar puncture (LP). First, measure blood pressure to rule out malignant hypertension, then perform emergency imaging (CT → MRI plus contrast-enhanced MRV) to assess for mass lesions and venous sinus thrombosis. Perform LP after confirming no risk of brain herniation.
MRI plus contrast-enhanced MRV is optimal; evaluation of venous sinus stenosis/occlusion is essential in IIH. The following signs of high ICP are assessed:
| Criteria | Description |
|---|---|
| 1. Papilledema | Bilateral papilledema present |
| 2. Neurologic examination | Normal except for cranial nerve abnormalities |
| 3. Neuroimaging | Normal brain parenchyma, no ventricular enlargement, no mass, no abnormal contrast enhancement |
| 4. CSF composition | Normal |
| 5. CSF opening pressure | Adults ≥250 mmH2O, children ≥280 mmH2O |
Suspected diagnosis: bilateral papilledema present + criteria 1-4 met but CSF pressure below threshold. 25-30 cmH2O is a gray zone and reassessment is recommended.
It has been reported that 17.8% of IIH diagnoses are inaccurate or premature, and 13.0% are misdiagnoses 18). The most common cause of misdiagnosis is failure to perform MRV (42.4%) in atypical patients (men, normal BMI, children), leading to missed CVST.
Pseudopapilledema can be caused by high hyperopia, drusen, tilted optic disc, myelinated nerve fibers, hamartoma, etc. The following findings suggest pseudopapilledema.
Differential diagnosis list: Buried drusen, tilted disc, diabetic papillopathy, hypertensive papillopathy, posterior scleritis, perioptic neuritis, uveitis (sarcoidosis, VKH), optic neuritis, thyroid eye disease, CRVO, NAION, infiltrative optic neuropathy, optic nerve sheath meningioma.
It is reported that 10% of patients with papilledema have treatable iron deficiency anemia, and screening is recommended.
In choked disc (papilledema due to increased ICP), vision may be preserved in the early stages. This is because although increased ICP causes stasis of axoplasmic flow in the optic nerve head, the effect on afferent visual function is still mild. In contrast, optic neuritis often causes acute vision loss, and this difference can be a clue for differentiation.
The three goals of treatment are: ① treatment of the underlying cause, ② preservation of visual function, and ③ relief of symptoms (such as headache).
If intracranial pressure is lowered early, papilledema is quickly absorbed and visual function is preserved. If treatment is delayed and visual impairment appears, it becomes irreversible.
| Drug | Dose | Notes |
|---|---|---|
| Acetazolamide (first-line) | 250–500 mg twice daily → up to 2–4 g/day | IIHTT (2014) showed efficacy for mild visual field loss in IIH16). No consistent effect on headache. Not covered by insurance in Japan. |
| Topiramate (second-line) | Individualized dosing | Lowers ICP and promotes weight loss. Also useful for migraine-like headaches. Teratogenic → contraindicated in pregnancy. |
| Furosemide | Adjunctive use | Alternative when acetazolamide is not tolerated. |
| Methylprednisolone IV | 1 g/day for 3 days | Temporary measure only for fulminant IIH. Common practice in North America. |
Main side effects of acetazolamide: paresthesia (tingling in hands and feet), taste disturbance (metallic taste), gastrointestinal symptoms (nausea, vomiting, diarrhea), fatigue, kidney stones. Chronic compensated metabolic acidosis, mild hypokalemia.
| Procedure | Visual improvement rate | Headache improvement rate | Major complications |
|---|---|---|---|
| Optic nerve sheath fenestration (ONSF) | 59% | 44% | Complications 10–15% (including 1–2% vision loss from CRAO/CRVO). Recurrence rate 34% at 1 year, 45% at 3 years1) |
| CSF shunting (LPS/VPS) | 54% | 80% | Shunt failure 43–50%, infection, migration1) |
| Dural venous sinus stenting (VSS) | 78% | 82–83% | Antiplatelet therapy required for 6 months postoperatively. Reoperation rate 10–18%1) |
| Bariatric surgery | — | — | IIHWT RCT showed sustained ICP reduction and weight loss over 2 years15) |
Indications for VSS (venous sinus stenting): Confirmation of transverse sinus stenosis with pressure gradient >8 mmHg. ONSF does not lower ICP. Unilateral ONSF may have insufficient effect on the contralateral side. Lumbar-peritoneal (LP) shunt is often chosen as the primary surgical intervention for IIH.
2–3% of IIH patients. Characterized by rapid visual loss within 4 weeks of symptom onset. Inpatient management is standard, with aggressive medical and surgical treatment.
Mean CSF opening pressure is extremely high at 54.1 cmH2O (range 29–70) 6). In a series of 16 patients by Thambisetty et al. 6), 50% became legally blind despite aggressive treatment, and all had residual visual field defects and optic atrophy. Visual recovery is difficult if baseline HVF MD is less than -7 dB. 1) Higher initial pRNFL is associated with worse long-term visual prognosis. 1)
There is a report of FIH recovery with acetazolamide alone at up to 4 g/day (36-year-old male, BMI 47.3, CSF OP 45 cmH2O → papilledema resolved in 4 months, 20/20 vision recovered in both eyes at 6 months) 8). When FIH is complicated by malignant hypertension, diagnostic delay averages 3.2 months, and final visual acuity can be extremely poor (20/400 to light perception) 7).
The prevalence of IIH during pregnancy is 16 per 100,000. 61% of cases occur in the first trimester 9). Pregnancy itself is not considered an etiological factor for IIH. Visual prognosis is similar to that in non-pregnant women, and it is not usually considered a high-risk pregnancy.
Prognosis: IIH recurrence rate 9–28%2). Poor prognostic factors: weight gain, severity of papilledema, and baseline headache severity2).
Acetazolamide is established as a first-line treatment for IIH, but it is contraindicated when cerebral venous thrombosis is the cause, as it may worsen the condition. It is also contraindicated in cryptococcal meningitis. It is essential to first identify the cause and select treatment based on the underlying etiology. Note that it is not covered by insurance in Japan.
Although classified as FDA category C, systematic reviews have not found a causal relationship with congenital malformations9). Even high-dose use (≥1 g/day) in the first trimester did not show an increased risk of malformations. It should always be used in consultation with an obstetrician, and in many cases, use is limited to after 20 weeks of gestation.
Fulminant IIH (FIH) is a subtype of IIH characterized by rapid and severe visual loss within 4 weeks of symptom onset. It occurs in 2–3% of IIH patients. While typical IIH progresses relatively slowly, FIH can worsen within days and lead to irreversible blindness without prompt intervention6).
Increased pressure in the subarachnoid space around the optic nerve compresses the nerve, leading to stasis of axoplasmic flow and resulting in papilledema. Specifically, the following pathway occurs.
Increased ICP → increased pressure in the subarachnoid space around the optic nerve → optic nerve constriction → stagnation of anterograde axonal transport → nerve edema (papilledema). Persistent pressure → intra-neural ischemia → axonal loss → optic atrophy → visual impairment. In the atrophic stage, dead nerve fibers do not swell, so papilledema may disappear even if ICP elevation continues (end-stage atrophy).
CSF outflow obstruction hypothesis
Delayed CSF drainage via arachnoid granulations and lymphatic pathways leads to increased ICP. Minocycline disrupts cAMP signaling in arachnoid granulations and reduces CSF absorption 13).
Venous sinus pressure elevation hypothesis
Bilateral transverse sinus stenosis is observed in over 90% of IIH patients 2). The chain: obesity → increased intra-abdominal pressure → increased intrathoracic pressure → impaired cerebral venous return → increased ICP. The effectiveness of VSS supports this hypothesis.
Metabolic and hormonal abnormality hypothesis
Androgen dysregulation is suggested to be involved 2). Excess leptin → choroid plexus hyperactivation → excessive CSF production. GLP-1 receptors are present in the choroid plexus, and GLP-1 RAs have been shown to reduce CSF production in rat models.
In an RCT comparing bariatric surgery versus community weight management intervention, the bariatric surgery group sustained ICP reduction and weight loss over 2 years 15). Cost-effectiveness after 5 years was also superior to dietary therapy. The amount of ICP reduction correlated with the degree of weight loss.
GLP-1RAs such as exenatide are attracting attention as new candidates for IIH treatment. They reduce CSF secretion in the choroid plexus via decreased Na⁺/K⁺-ATPase activity. In humans, a report showed ICP reduction of 5.7 ± 2.9 cmH₂O (P = 0.048) 2.5 hours after administration. A dual mechanism of weight loss promotion and direct ICP reduction is expected. However, attention should be paid to the risk of weight rebound after discontinuation leading to IIH onset.
Reports on the safety and efficacy of transverse sinus stenting are accumulating 4, 5). An RCT comparing VSS vs VPS is ongoing in the UK. Visual improvement rate of VSS is 78%, headache improvement rate 82–83% (meta-analysis). Reoperation rate ranges from 10% to 18%.
11β-HSD1 (11β-hydroxysteroid dehydrogenase type 1) inhibitors are under investigation as novel therapeutic targets for IIH 1).
There is discussion proposing a definition based on “vision-threatening” rather than the time criterion of “within 4 weeks” 1). The time criterion may miss cases with rapid progression.
Papilledema has been reported in patients who do not fit the traditional risk profile 1). Examples include IIH in non-obese, non-female individuals; MIS-C (multisystem inflammatory syndrome in children)-related cases; and VITT after the AstraZeneca vaccine.
Significant interstate differences have been identified within the United States (Moran I = 0.20, P = 0.03) 3). In states such as Texas and Oklahoma, a discrepancy has been observed with high obesity rates but low IIH prevalence, the cause of which remains unknown.
Bonelli L, Menon V, Arnold AC, Mollan SP. Managing idiopathic intracranial hypertension in the eye clinic. Eye. 2024;38:2472-2481.
Toshniwal SS, Kinkar J, Chadha Y, et al. Navigating the enigma: a comprehensive review of idiopathic intracranial hypertension. Cureus. 2024;16(3):e56256.
Fraz MA, Kim BM, Chen JJ, et al. Nationwide prevalence and geographic variation of idiopathic intracranial hypertension among women in the United States. Ophthalmology. 2025;132:476-483.
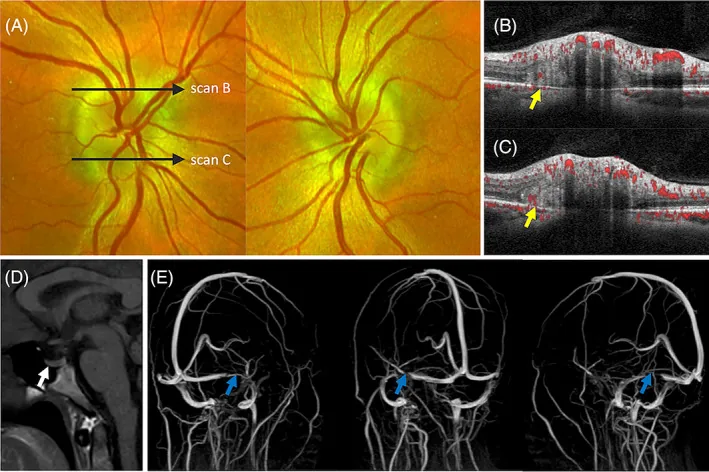
Ghorbani M, Griessenauer CJ, Wipplinger C, et al. Intracranial hypertension and papilledema secondary to an unruptured arteriovenous malformation: review of the literature. Neuroradiol J. 2025;38(4):387-393.
Waser B, Wood HM, Mews P, Lalloo S. Transverse sinus stenting for treatment of papilloedema secondary to a large brain herniation into a dural venous sinus with associated tectal plate lesion: case report and literature review. Interv Neuroradiol. 2021;27(6):756-762.
Thambisetty M, Lavin PJ, Newman NJ, Biousse V. Fulminant idiopathic intracranial hypertension. Neurology. 2007;68(3):229-232.
Aldhahwani B, Shah SM, Jiang H, Lam BL. Severe visual loss from concurrent fulminant idiopathic intracranial hypertension and malignant arterial hypertension: prompt suspicion matters. Am J Ophthalmol Case Reports. 2024;36:102201.
Srivastava O, Micieli JA. Resolution of fulminant idiopathic intracranial hypertension treated with acetazolamide. Case Rep Neurol. 2022;14:432-436.
Palermo M, Trevisi G, D’Arrigo S, Sturiale CL. Idiopathic intracranial hypertension in pregnancy: a systematic review on clinical course, treatments, delivery and maternal-fetal outcome. Eur J Neurol. 2025;32:e70186.
Alves S, Sousa N, Cardoso L, Alves J. Multidisciplinary management of idiopathic intracranial hypertension in pregnancy: case series and narrative review. Braz J Anesthesiol. 2022;72(6):790-794.
Regev T, Fried-Regev N, Leeman S, et al. Transverse venous sinus stenting for fulminant idiopathic intracranial hypertension during pregnancy: a report of two cases and literature review. Quant Imaging Med Surg. 2025;15(5):4796-4815.
Selvaraj J, Veeranki V, Kommaraju SY, et al. Abortion and fulminant idiopathic intracranial hypertension. Cureus. 2021;13(2):e13501.
Paramo R, Leishangthem L. Optic atrophy secondary to minocycline-induced idiopathic intracranial hypertension. BMJ Case Rep. 2023;16:e252731.
Mollan SP, Davies B, Silver NC, et al. Idiopathic intracranial hypertension: consensus guidelines on management. J Neurol Neurosurg Psychiatry. 2018;89(10):1088-1100.
Mollan SP, Mitchell JL, Ottridge RS, et al. Effectiveness of bariatric surgery vs community weight management intervention for the treatment of idiopathic intracranial hypertension: a randomized clinical trial. JAMA Neurol. 2021;78:678-686.
Wall M, McDermott MP, Kieburtz KD, et al. Effect of acetazolamide on visual function in patients with idiopathic intracranial hypertension and mild visual loss: the idiopathic intracranial hypertension treatment trial. JAMA. 2014;311:1641-1651.
Pasricha SV, Bhayana R, Wu PE. Supine headache and papilledema: a case and review of cerebral venous sinus thrombosis. Clin Case Rep. 2023;11:e07329.
Fisayo A, Bruce BB, Newman NJ, Biousse V. Overdiagnosis of idiopathic intracranial hypertension. Neurology. 2016;86:341-350.