Infectious Uveitis
Key Points at a Glance
Section titled “Key Points at a Glance”1. What is infectious uveitis?
Section titled “1. What is infectious uveitis?”Uveitis is a general term for intraocular inflammation. Based on etiology, it is broadly classified into exogenous (infectious) uveitis and endogenous (non-infectious) uveitis. Infectious uveitis refers to intraocular inflammation caused directly or via immune-mediated mechanisms by pathogens such as bacteria, viruses, fungi, and parasites. The annual incidence of uveitis is estimated at 17–52 per 100,000 population, and the prevalence at 38–714 per 100,000 population 1,16).
In an epidemiological survey conducted by the Japanese Society for Ocular Inflammation in 2002 (3,060 cases), infectious uveitis accounted for approximately 16% of all cases 1,7). Major diseases included herpetic iritis 3.6%, bacterial endophthalmitis 3.8%, acute retinal necrosis (ARN) 1.3%, ocular toxoplasmosis 1.1%, ocular toxocariasis 1.1%, CMV retinitis 0.8%, and HTLV-1-associated uveitis 1.0%. Internationally, toxoplasmosis and tuberculous uveitis are reported to account for over 50% of infectious uveitis in developing countries 2,13), showing marked regional differences 8).
Identifying the cause of infectious uveitis is important because the treatment strategy differs fundamentally from that of non-infectious uveitis. Non-infectious uveitis is primarily treated with immunosuppression (steroids and immunomodulatory drugs), whereas infectious uveitis requires specific treatment targeting the pathogen first, and careless steroid administration can rapidly worsen the disease.
2. Classification
Section titled “2. Classification”2-1. Classification by Location
Section titled “2-1. Classification by Location”Uveitis is classified into the following four types based on the primary site of inflammation (SUN Working Group classification):
| Classification | Site | Representative diseases (infectious) |
|---|---|---|
| Anterior uveitis | Iris and ciliary body (iritis, iridocyclitis) | HSV/VZV iritis, CMV anterior uveitis, Lyme disease |
| Intermediate uveitis | Vitreous, pars plana | HTLV-1-associated uveitis, Lyme disease |
| Posterior uveitis | Retinochoroid, posterior vitreous | CMV retinitis, ocular toxoplasmosis, ocular toxocariasis |
| Panuveitis | Full-thickness | ARN, tuberculous, syphilitic, fungal endophthalmitis |
2-2. Classification by pathogen
Section titled “2-2. Classification by pathogen”Infectious uveitis is classified into four groups according to the type of pathogen.
The herpesvirus family (HHV-1 to 8) is the most common, with HSV-1/2, VZV, CMV, and HTLV-1 being clinically important. Depending on the host’s immune status, various clinical presentations occur: in immunocompetent individuals, HSV/VZV cause anterior uveitis and ARN; in immunocompromised individuals, CMV retinitis and PORN are typical.
Bacterial
Section titled “Bacterial”Major pathogens include Mycobacterium tuberculosis, Treponema pallidum (syphilis), Bartonella (cat scratch disease), and Borrelia (Lyme disease). In endogenous bacterial endophthalmitis, Gram-negative rods (e.g., Escherichia coli, Klebsiella) are common, with rapid progression and poor prognosis.
Fungal
Section titled “Fungal”Candida, Aspergillus, and Cryptococcus cause endogenous infections in immunocompromised hosts (e.g., central venous catheter, immunosuppression, HIV infection). Endophthalmitis following candidemia is particularly problematic.
Parasitic
Section titled “Parasitic”Ocular toxoplasmosis (Toxoplasma gondii) and ocular toxocariasis (Toxocara canis/cati) are representative diseases. Toxoplasmosis can reactivate even after adult-acquired infection, causing lymphadenopathy and acquired retinochoroiditis.
2-3. Classification by route of infection
Section titled “2-3. Classification by route of infection”| Classification | Route of infection | Representative diseases |
|---|---|---|
| Endogenous | Hematogenous dissemination (other organs → eye) | Bacterial endophthalmitis, fungal endophthalmitis, tuberculosis, syphilis, CMV retinitis |
| Exogenous | Direct invasion (trauma/surgery) | Postoperative infectious endophthalmitis, post-traumatic endophthalmitis |
3. Overview of Viral Uveitis
Section titled “3. Overview of Viral Uveitis”Uveitis involving viruses in the inflammation of the uvea, retina, and vitreous is called viral uveitis. Human herpesviruses (HHV) have a strong affinity for retinal tissue and cause various ocular diseases. Viral uveitis is characterized by diverse clinical presentations depending on the host’s immune status, driven by direct viral toxicity and inflammation triggered by immune responses. Comprehensive detection using PCR of intraocular fluids has greatly improved diagnostic rates.
3-1. Herpesvirus Pathogens and Ocular Manifestations
Section titled “3-1. Herpesvirus Pathogens and Ocular Manifestations”| Virus | Ocular manifestations in immunocompetent patients | Ocular manifestations in immunocompromised patients |
|---|---|---|
| HSV-1/2 (HHV-1/2) | Herpetic iridocyclitis, ARN | PORN (rare) |
| VZV (HHV-3) | Herpes zoster ophthalmicus, herpetic iridocyclitis, ARN | PORN |
| CMV (HHV-5) | Corneal endotheliitis, CMV anterior uveitis | CMV retinitis, immune recovery uveitis (IRU) |
| EBV (HHV-4) | Mild uveitis (rare) | ARN-like lesions (rare) |
| HTLV-1 | Intermediate to panuveitis (veil-like vitreous opacities) | Same as left (severe) |
| Rubella virus | Association with Fuchs heterochromic iridocyclitis | ─ |
3-2. Acute retinal necrosis (ARN)
Section titled “3-2. Acute retinal necrosis (ARN)”ARN presents as acute iridocyclitis with pigmented keratic precipitates and is a necrotizing retinitis in which yellowish-white granular lesions in the peripheral fundus rapidly coalesce and expand. VZV is the most common cause (often severe cases), followed by HSV-1/2. Rhegmatogenous retinal detachment with retinal tears occurs at a high rate (approximately 75%), and visual prognosis is poor18).
Japanese Ocular Inflammation Society ARN Diagnostic Criteria (Definite diagnosis group):
- Initial ocular findings: ① anterior chamber cells or mutton-fat KP, ② one or more yellow-white lesions (peripheral), ③ retinal arteritis, ④ optic disc hyperemia, ⑤ inflammatory vitreous opacities, ⑥ elevated intraocular pressure; ① and ② are essential.
- Progression items: one or more of rapid circumferential expansion, retinal tear/retinal detachment, vascular occlusion, optic atrophy, response to anti-herpetic drugs.
- Intraocular fluid test: PCR of aqueous humor or vitreous fluid positive for HSV-1, HSV-2, or VZV.
The clinical diagnostic group (not requiring intraocular fluid test) can be diagnosed when initial findings ① and ② are present, plus two items from ③ to ⑥ and one progression item.
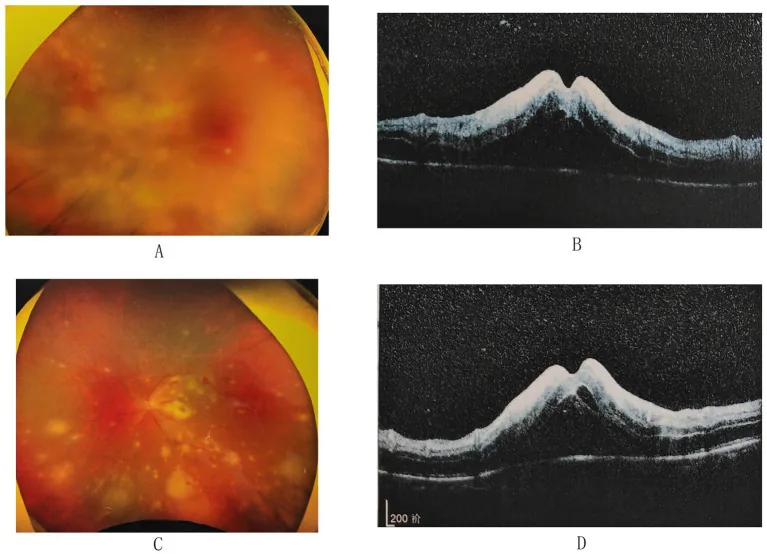
3-3. CMV retinitis
Section titled “3-3. CMV retinitis”CMV is an opportunistic infection that occurs in immunocompromised patients, primarily those with AIDS, and is most common in AIDS patients whose CD4-positive T-cell count has dropped to 50–100/µL 20). There are three clinical types: (1) peripheral granular type (fan-shaped lesions with white granules), (2) posterior polar vasculitis type (hemorrhage and edema), and (3) frosted branch angiitis type (perivascular whitening of large vessels), which often coexist. Immune recovery uveitis (IRU) following immune recovery is an important complication 15), and ophthalmic management after initiation of antiretroviral therapy is necessary.
3-4. HTLV-1-associated uveitis (HAU)
Section titled “3-4. HTLV-1-associated uveitis (HAU)”HAU occurs in approximately 0.1% of HTLV-1 carriers and is more common in regions such as Kyushu and Okinawa 12). Characteristic findings include white granular keratic precipitates, iris nodules, vitreous opacities that are veil-like, stringy, or granular, and white granular deposits around retinal vessels. Hyperthyroidism is frequently associated. Visual prognosis is relatively good, but recurrence may occur after steroid tapering or discontinuation.
3-5. Rubella virus and Fuchs heterochromic iridocyclitis
Section titled “3-5. Rubella virus and Fuchs heterochromic iridocyclitis”Fuchs heterochromic iridocyclitis (characterized by the triad of heterochromia, iridocyclitis, and cataract) has been suggested to be associated with rubella virus. In congenital rubella syndrome, transplacental infection within the first three months of pregnancy leads to retinochoroiditis with a salt-and-pepper fundus appearance.
4. Overview of Bacterial Uveitis
Section titled “4. Overview of Bacterial Uveitis”4-1. Tuberculous Uveitis
Section titled “4-1. Tuberculous Uveitis”Intraocular inflammation caused by Mycobacterium tuberculosis involves two mechanisms: direct infection and immune-mediated (hypersensitivity reaction). The clinical forms are diverse, including choroidal granulomas (tubercles), serous retinal detachment, shotgun choroiditis, vasculitis, and panuveitis.
IGRA (QuantiFERON®-TB Gold Plus or T-SPOT®.TB) is useful for diagnosis. Note that the tuberculin skin test may be negative in elderly patients and those with impaired cell-mediated immunity such as AIDS. Regarding visual prognosis in tuberculous uveitis, it has been reported that best-corrected visual acuity falls below 3/60 in about one-third of cases 3). The diagnostic criteria for tuberculous uveitis proposed by the SUN Working Group (2021) include: (1) anterior uveitis with iris nodules, (2) serpiginous-like choroiditis, (3) choroidal nodules (tuberculomas), (4) multifocal choroiditis in cases of active systemic tuberculosis, and (5) occlusive retinal vasculitis 5). In India and Indonesia, approximately 22.9–48.0% of infectious uveitis cases are attributed to tuberculosis 3,14), highlighting its particular importance in high tuberculosis prevalence regions.
4-2. Syphilitic Uveitis
Section titled “4-2. Syphilitic Uveitis”Intraocular inflammation caused by Treponema pallidum is known as the “great mimicker” and can present as anterior, posterior, or panuveitis. In recent years, a global resurgence has been observed, particularly among men who have sex with men (MSM), and HIV-coinfected individuals have approximately twice the risk of ocular syphilis4). Worldwide, an estimated 5.7 to 6 million new syphilis infections (ages 15–49) are reported annually4), and ocular involvement occurs in about 1–1.5% of syphilis patients4,10).
Diagnosis is made by serological tests (TPHA, RPR/VDRL), interpreted using a combination of treponemal and non-treponemal methods. Treatment follows neurosyphilis guidelines: intravenous benzylpenicillin G (Aqacillin®) 24 million units/day for 10–14 days is first-line, with intravenous ceftriaxone as an alternative. Be alert for the Jarisch-Herxheimer reaction (fever and inflammation exacerbation within 24 hours of treatment).
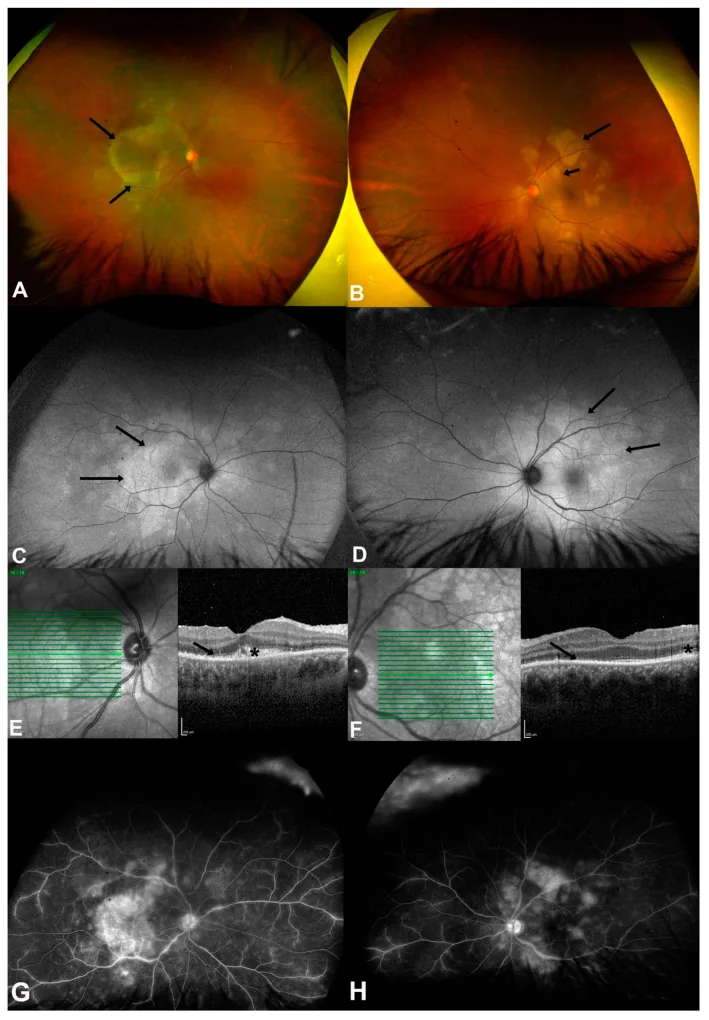
4-3. Cat Scratch Disease (Bartonella Infection)
Section titled “4-3. Cat Scratch Disease (Bartonella Infection)”Infection caused by Bartonella henselae, with a history of cat contact being an important interview item. Fundus findings include optic neuritis (macular star sign), macular stellate whitish spots, and serous retinal detachment, often presenting as posterior uveitis. Diagnosis is made by measuring anti-Bartonella henselae antibody titers, and antibiotic treatment with azithromycin, doxycycline, or rifampicin is administered.
4-4. Lyme Disease
Section titled “4-4. Lyme Disease”An infectious disease caused by Borrelia spirochetes transmitted by ticks, with various ocular manifestations (uveitis, retinal vasculitis, optic neuritis, keratitis, etc.) appearing in stages 2 to 3. Erythema migrans is a characteristic rash in stage 1, and a history of tick bite is a diagnostic clue. Diagnosis is made by elevated serum IgM antibody titers via ELISA, and treatment is with penicillin or tetracycline antibiotics. Differentiation from syphilis and sarcoidosis is necessary.
4-5. Hansen’s Disease (Leprosy)
Section titled “4-5. Hansen’s Disease (Leprosy)”A chronic granulomatous infection caused by Mycobacterium leprae, with ocular involvement reported in 70-80% of cases (30-40% in recent new patients). Chronic granulomatous anterior uveitis is predominant, with characteristic findings including iris pearls, iris atrophy, small pupil, and tent-shaped peripheral anterior synechiae. Multidrug therapy (rifampicin, dapsone, clofazimine) is used, and steroid eye drops are added when uveitis is present.
4-6. Endogenous Bacterial Endophthalmitis
Section titled “4-6. Endogenous Bacterial Endophthalmitis”Endogenous bacterial endophthalmitis, in which infection spreads hematogenously to the eye from other organ infections (e.g., liver abscess, endocarditis, pneumonia), is primarily caused by Gram-negative bacilli (e.g., Klebsiella, Escherichia coli) and has an extremely poor prognosis. It progresses more rapidly than exogenous (postoperative) Gram-positive coccal infection, and systemic search for and treatment of the infection source are essential.
5. Overview of Fungal Uveitis
Section titled “5. Overview of Fungal Uveitis”Fungal endophthalmitis is mostly endogenous, with Candida spp. being the most common, and Aspergillus and Cryptococcus are also important pathogens.
Main risk factors:
- Central venous nutrition (IVH) and indwelling catheters
- Long-term broad-spectrum antibiotic use
- Immunosuppression (HIV infection, organ transplantation, malignancy, chemotherapy)
- Diabetes mellitus
- Intravenous drug use
Fungal endophthalmitis is characterized by white spherical or cotton-like vitreous opacities (“clumped opacities”) and progresses more slowly than bacterial endophthalmitis. Early stages may be asymptomatic or present only with mild floaters, making them easy to overlook. As it progresses, redness, eye pain, and panuveitis appear. 30% of endogenous fungal endophthalmitis occurs in both eyes8).
Diagnosis is made by blood/catheter tip culture, β-D-glucan, Candida antigen measurement, and vitreous culture. Treatment is based on a combination of antifungal drugs (voriconazole, amphotericin B, fluconazole) and vitrectomy.
6. Overview of Parasitic Uveitis
Section titled “6. Overview of Parasitic Uveitis”6-1. Ocular Toxoplasmosis
Section titled “6-1. Ocular Toxoplasmosis”Toxoplasma gondii (an intracellular parasitic protozoan) is estimated to infect about one-third of the world’s population, and the antibody positivity rate in Japanese adults is reported to be 20–30% 11). Felids are the definitive hosts, and infection occurs through oral ingestion of oocysts from cat feces or consumption of undercooked meat.
Ocular toxoplasmosis accounts for approximately 1% of the causes of uveitis 11). The fundus findings in acquired infection are peripheral, poorly demarcated white to milky-white exudative chorioretinitis, and “satellite lesions” adjacent to old scarred lesions are characteristic findings upon recurrence. It is accompanied by anterior chamber inflammation and vitreous opacities (“headlights in the fog”). In congenital infection, scarred lesions with central gray-white fibrous proliferation and pigmentation are seen in both maculae.
Diagnosis is primarily based on characteristic ocular findings and measurement of serum antibody titers (IgG, IgM). Intraocular fluid testing using the Q value (Goldmann-Witmer coefficient) or PCR is also useful. Treatment involves oral administration of spiramycin acetate ester (acetylspiramycin) or clindamycin 1.2 g/day for 4 to 6 weeks, combined with steroids 0.5 mg/kg/day to control exudative lesions.
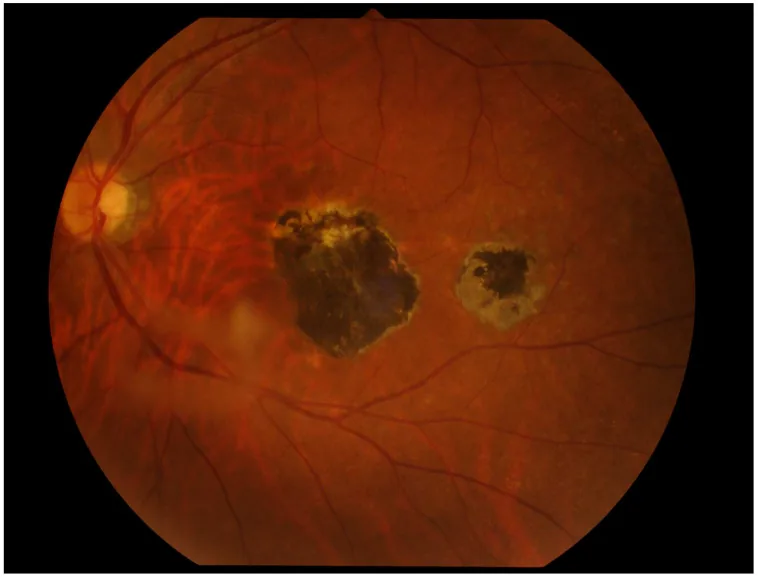
6-2. Ocular Toxocariasis
Section titled “6-2. Ocular Toxocariasis”This is a larval migration disease caused by the dog roundworm (Toxocara canis) or cat roundworm (Toxocara cati), and is often unilateral. There are three types: endophthalmitis type, posterior pole mass type, and peripheral mass type (most common), which can cause severe vitreous opacities and retinal detachment. Diagnosis is made by serum antibody testing (e.g., Toxocara CHECK® ELISA) and characteristic ocular findings. Treatment includes oral anthelmintics (diethylcarbamazine), oral steroids, and may involve cryocoagulation, laser photocoagulation, or vitrectomy.
7. Diagnostic Strategy
Section titled “7. Diagnostic Strategy”7-1. Key Points in History Taking
Section titled “7-1. Key Points in History Taking”Systematic history taking is essential for the diagnosis of infectious uveitis:
- Age and sex: CMV retinitis is common in AIDS patients (mostly men in their 20s-40s), HAU in residents of Kyushu and Okinawa
- Region and travel history: Travel to areas with high tuberculosis prevalence (e.g., Southeast Asia, Africa)
- Animal contact history: Cats (Toxoplasma, Bartonella), Dogs (Toxocara)
- Dietary history: Raw meat or raw fish consumption (Toxoplasma)
- Sexual history and HIV testing history: Syphilis, CMV
- Immunosuppressed state: HIV, malignancy, organ transplant, immunosuppressant use
- IVH or catheter history: Fungal endophthalmitis
- History of herpes zoster or herpes labialis: VZV or HSV iritis
7-2. Differentiation by Ocular Findings
Section titled “7-2. Differentiation by Ocular Findings”| Finding | Suspected Infectious Disease |
|---|---|
| Mutton-fat KP | HSV/VZV iritis, ARN, tuberculous uveitis |
| Anterior uveitis with high intraocular pressure (≥25 mmHg) | HSV/VZV iritis, CMV anterior uveitis |
| Peripheral confluent white necrotic lesions | ARN (VZV/HSV) |
| White infiltrates with posterior pole hemorrhage | CMV retinitis (vasculitic type) |
| Massive vitreous opacities (progressive) | Fungal endophthalmitis |
| Veil-like or string-like vitreous opacities | HTLV-1-associated uveitis |
| Old scar + satellite lesions | Ocular toxoplasmosis |
| Peripheral white mass + vitreous strands | Ocular toxocariasis |
| Granulomatous iritis + choroidal granuloma | Tuberculous uveitis, syphilitic |
| Optic neuritis + macular star | Cat scratch disease (Bartonella) |
7-3. Systemic examination
Section titled “7-3. Systemic examination”| Examination | Target disease |
|---|---|
| Serum TPHA・RPR/VDRL | Syphilitic uveitis |
| IGRA (QuantiFERON・T-SPOT) | Tuberculous uveitis |
| CMV antigenemia (C7-HRP method)・CMV-PCR | CMV retinitis |
| Anti-HTLV-1 antibody | HTLV-1-associated uveitis |
| Anti-HIV antibody / CD4 count | CMV / PORN / ocular syphilis |
| β-D-glucan / Candida antigen | Fungal endophthalmitis |
| Anti-Toxoplasma IgG/IgM | Ocular toxoplasmosis |
| Anti-Toxocara antibody (ELISA) | Ocular toxocariasis |
| Anti-Bartonella henselae antibody | Cat scratch disease |
| Blood culture and echocardiography | Endogenous bacterial endophthalmitis |
7-4. Intraocular fluid testing (aqueous humor and vitreous humor)
Section titled “7-4. Intraocular fluid testing (aqueous humor and vitreous humor)”Testing using intraocular fluid is the most important definitive method for diagnosing infectious uveitis, and specimens are obtained by anterior chamber paracentesis (aqueous humor collection) or vitrectomy (vitreous humor collection)9).
PCR method
Section titled “PCR method”DNA is extracted from aqueous humor or vitreous humor, and DNA of each pathogen is detected by PCR. Multiplex real-time PCR allows comprehensive detection of multiple viruses from a small sample volume and is particularly useful for differential diagnosis of ARN19).
Infectious uveitis kit (advanced medical care): Can simultaneously detect HSV-1/2, VZV, CMV, EBV, HHV-6/7, HTLV-1, Toxoplasma gondii DNA, etc., and is used for diagnosing co-infections with multiple pathogens and refractory cases. However, note that PCR may not detect CMV retinitis in the early stage (before anterior chamber inflammatory cells appear).
Goldmann-Witmer ratio (Q value, antibody ratio)
Section titled “Goldmann-Witmer ratio (Q value, antibody ratio)”This method detects the presence of pathogen-specific antibody production within the eye and is calculated using the following formula:
Q value = (Intraocular fluid virus antibody titer ÷ Intraocular fluid IgG level) ÷ (Serum virus antibody titer ÷ Serum IgG level)
- Q value >1: Possible local antibody production within the eye
- Q value ≥6: Significant local antibody production confirmed, allowing identification of the virus as the causative agent
Note that the Q value may be underestimated within the first 10 days of onset because intraocular antibody production is insufficient. Etiological diagnosis cannot be made based solely on serum antibody titers (most adults are seropositive due to past infection)6).
Intraocular fluid PCR usage flow
Section titled “Intraocular fluid PCR usage flow”眼内炎症患者 ├─ ウイルス性疑い(高眼圧・前房炎症・壊死性網膜炎) │ → 前房水PCR(HSV/VZV/CMV)+ Q値 ← 第一選択 │ ─ 前房水で陰性かつ壊死性病変 → 硝子体液PCR │ ─ 免疫不全者 → マルチプレックスPCR(先進医療) ├─ 寄生虫疑い(衛星病巣・周辺部腫瘤) │ → 眼内液Q値(Toxoplasma)または眼内液PCR ├─ 真菌疑い(塊状混濁・リスク因子あり) │ → 硝子体液培養 + β-D-グルカン(血液) └─ 細菌疑い(急速進行・IVH歴) → 硝子体液グラム染色・培養(至急)8. Principles of treatment
Section titled “8. Principles of treatment”8-1. Specific treatment by pathogen
Section titled “8-1. Specific treatment by pathogen”| Pathogen | First-line treatment | Notes |
|---|---|---|
| HSV (ARN, iritis) | ACV 10 mg/kg ×3 times IV for 2 weeks → VACV 1,000 mg ×3 times/day oral | Combine with steroid eye drops and IV for anti-inflammation |
| VZV (ARN, iritis) | ACV 10-15 mg/kg x3 IV + VACV oral | PORN follows CMV treatment |
| CMV (retinitis) | GCV 5 mg/kg x2 IV for 2-3 weeks (induction) → VGCV 900 mg x2/day oral (maintenance) | Immune recovery is the curative treatment |
| CMV (anterior uveitis) | GCV gel 0.15% eye drops (73% of specialists choose as first-line) ± oral VGCV | For chronic course, consider 12-month maintenance therapy (73% of specialists choose GCV gel 0.15%) 2) |
| Tuberculosis | INH, RFP, PZA, EMB (standard 6-month regimen) | Corticosteroids used as appropriate |
| Syphilis | Benzylpenicillin G 24 million units/day IV for 10–14 days | Watch for Jarisch-Herxheimer reaction |
| Fungal (Candida) | Voriconazole or amphotericin B ± vitrectomy | Monitor with β-D-glucan index |
| Toxoplasma | Acetylspiramycin + steroid or clindamycin | Caution in pregnancy (use spiramycin) |
| Toxocara | Diethylcarbamazine + steroid | Consider laser/vitrectomy |
| Lyme disease | Amoxicillin or doxycycline for 3 weeks | Add steroid eye drops for ocular involvement |
| Leprosy | MDT (rifampicin, DDS, clofazimine) | Steroids needed during leprosy reaction |
8-2. Appropriate Use of Steroids
Section titled “8-2. Appropriate Use of Steroids”When using steroids for infectious uveitis, anti-pathogen treatment must precede or be given concurrently. The role of steroids is to reduce secondary tissue damage caused by inflammation, and they are used in combination with appropriate pathogen treatment for many infectious uveitis conditions such as ARN, toxoplasmosis, tuberculous uveitis, and Hansen’s disease uveitis.
On the other hand, for CMV retinitis (a pure opportunistic infection), treatment of the underlying disease (AIDS) to improve immune function is the curative therapy, and steroids are generally not used.
8-3. Mydriatics and Intraocular Pressure Management
Section titled “8-3. Mydriatics and Intraocular Pressure Management”To prevent posterior synechiae, tropicamide-phenylephrine eye drops (1–6 times/day) are used for anterior segment inflammation. For elevated intraocular pressure (characteristic of HSV/VZV iritis and CMV anterior uveitis), intraocular pressure-lowering drugs such as carbonic anhydrase inhibitors and beta-blockers are selected, but prostaglandin-related drugs carry a risk of exacerbating inflammation and should be used with caution.
9. Risk Matrix by Immunosuppressive Condition
Section titled “9. Risk Matrix by Immunosuppressive Condition”| Background | Pathogens Requiring Special Attention | Priority Tests |
|---|---|---|
| HIV infection (CD4 <50/µL) | CMV (retinitis, ARN, PORN), fungi (Cryptococcus), syphilis | Intraocular fluid PCR (multiplex), CMV antigen, β-D-glucan |
| After solid organ transplant / immunosuppressant use | CMV, fungi (Aspergillus, Candida), EBV | CMV-PCR, β-D-glucan, blood culture |
| Malignancy / after chemotherapy | Fungi (Candida, Aspergillus), CMV | β-D-glucan, blood culture |
| Long-term IVH / catheter placement | Candida endophthalmitis | β-D-glucan / blood culture (ophthalmology consultation mandatory) |
| High tuberculosis risk (travel / contact history) | Tuberculosis (choroidal granuloma / vasculitis type) | IGRA / chest CT |
| MSM (men who have sex with men) / HIV+ | Syphilis (ocular syphilis approx. 1–1.5%), CMV | TPHA, RPR, HIV testing |
| Immunocompetent (middle-aged and older) | HSV/VZV iridocyclitis, ARN | Anterior chamber PCR, Q value |
| Childhood/pet contact history | Toxocara/Toxoplasma | Serum antibody (ELISA) |
10. Frequently Asked Questions
Section titled “10. Frequently Asked Questions”When it is difficult to differentiate between infectious and non-infectious uveitis based on clinical findings alone, or when serological tests are negative despite suspicion of infection, anterior chamber paracentesis is actively considered. In particular, when necrotizing retinitis (ARN, PORN, CMV retinitis) is suspected, intraocular fluid PCR is performed as early as possible because early definitive diagnosis directly affects treatment choice and prognosis. Multiplex PCR is useful for cases that are PCR-negative in the early stage or refractory cases.
First, intraocular fluid PCR for pathogen identification should be performed promptly, and specific antimicrobial therapy should be initiated as soon as the pathogen is identified. Since abrupt discontinuation of steroids may cause rebound inflammation, the dose should be carefully tapered after starting anti-pathogen treatment. In particular, when steroids have been administered alone for tuberculous uveitis, the risk of reactivation and dissemination of latent tuberculosis is increased, so systemic evaluation (chest CT, IGRA) should be performed urgently in collaboration with a respiratory physician.
After treatment, Toxoplasma remains as drug-resistant cysts within atrophic scarred lesions. Recurrence occurs in approximately 5–30% of cases, triggered by immunosuppression or pregnancy. For patients with frequent recurrences, long-term prophylactic treatment with trimethoprim-sulfamethoxazole may be considered. During pregnancy, if a Toxoplasma antibody-negative pregnant woman has a primary infection, there is a risk of vertical transmission to the fetus (approximately 40%), so she should be advised to avoid contact with cat feces, soil, and consumption of raw meat.
11. Related Articles
Section titled “11. Related Articles”Viral Uveitis
Section titled “Viral Uveitis”Bacterial and Other Infectious Uveitis
Section titled “Bacterial and Other Infectious Uveitis”References
Section titled “References”- 日本眼炎症学会ぶどう膜炎診療ガイドライン作成委員会. ぶどう膜炎診療ガイドライン. 日眼会誌. 2019;123(6):635–702.
- Thng ZX, Putera I, Testi I, et al. The Infectious Uveitis Treatment Algorithm Network (TITAN) Report 2—global current practice patterns for the management of Cytomegalovirus anterior uveitis. Eye. 2023. https://doi.org/10.1038/s41433-023-02503-3
- Putera I, Schrijver B, ten Berge JCEM, et al. The immune response in tubercular uveitis and its implications for treatment: From anti-tubercular treatment to host-directed therapies. Prog Retin Eye Res. 2023;95:101173.
- Chauhan K, Fonollosa A, Giralt L, et al. Demystifying Ocular Syphilis – A Major Review. Ocul Immunol Inflamm. 2023. https://doi.org/10.1080/09273948.2023.2217246
- Jabs DA, et al. Standardization of Uveitis Nomenclature (SUN) Working Group. Classification Criteria for Tubercular Uveitis. Am J Ophthalmol. 2021;228:142–149.
- 八代成子. ウイルス性ぶどう膜炎の診断と治療. 眼科. 2007;49:1193–1198.
- 大野重昭. ぶどう膜炎の疫学と分類. 日眼会誌. 2019;123(6).
- Tsirouki T, Dastiridou A, Symeon CI, et al. A focus on the epidemiology of uveitis. Ocul Immunol Inflamm. 2018;26(1):2–16.
- 慶野博. 感染性ぶどう膜炎の眼内液診断. 臨床眼科. 2019.
- Oliver SE, Aubin M, Atwell L, et al. Ocular Syphilis — Eight Jurisdictions, United States, 2014–2015. MMWR Morb Mortal Wkly Rep. 2016;65:1185–1188.
- 岡田アナベルあやめ. 眼トキソプラズマ症の診断と治療. 眼科. 2018.
- 中尾久美子. HTLV-1関連ぶどう膜炎の疫学と治療. 日本臨床. 2020.
- La Distia Nora R, et al. Infectious uveitis etiology in developing vs. developed countries. Curr Opin Ophthalmol. 2021.
- Agrawal R, et al. Collaborative Ocular Tuberculosis Study (COTS)—Report 1. Ophthalmology. 2019;126(11):1566–1577.
- 八代成子. CMV網膜炎・免疫回復ぶどう膜炎の管理. 眼科. 2007.
- Kempen JH, et al. The prevalence of uveitis in the United States. Arch Ophthalmol. 2004;122(4):543–557.
- Groen-Hakan F, et al. Diagnostic value of aqueous humor analysis for uveitis. Ocul Immunol Inflamm. 2020.
- 堤雅幸. 急性網膜壊死診断基準(日本眼炎症学会). 日眼会誌. 2020.
- 蕪城俊克. 眼内液PCRによるウイルス性ぶどう膜炎診断. 眼科. 2021.
- Bodaghi B, et al. Blindness prevention in HIV patients: CMV retinitis management update. Prog Retin Eye Res. 2023.
