Outer retina predominant type
White Dot Syndromes
Key Points at a Glance
Section titled “Key Points at a Glance”1. What are white dot syndromes?
Section titled “1. What are white dot syndromes?”White dot syndromes, a concept named by Gass in 1977, refer to a group of inflammatory diseases characterized by multiple white or yellowish-white spotty lesions in the fundus. Although the definition of target diseases varies among researchers, it is now widely used as a group of non-infectious idiopathic inflammatory diseases primarily involving the outer retina, RPE, choriocapillaris, and choroid 1).
In the Uveitis Clinical Practice Guidelines (Jpn J Ophthalmol 2019;123(6):635-696), APMPPE, MEWDS, PIC, multifocal choroiditis, birdshot retinochoroidopathy, serpiginous choroiditis, and AZOOR are listed as independent diseases under the classification of posterior uveitis, and when the inclusive term “white dot syndromes” is used, it encompasses these diseases 2).
Classification concepts
Section titled “Classification concepts”With recent advances in multimodal imaging (including OCT-A), white dot syndromes are now classified into the following three groups based on the primary layer of involvement1).
Choriocapillaris-dominant type
APMPPE, Serpiginous choroiditis, PIC
- Occlusive vasculitis of the choriocapillaris is central to the pathology
- OCT-A shows choriocapillaris flow void
- Outer retinal and RPE changes are secondary
Choroidal stroma-dominant type
- Primary lesion is lymphocytic infiltration of the choroidal stroma
- OCT-A shows flow void in Haller’s layer, with initial sparing of choriocapillaris
- Chronic progressive course with strong association with HLA-A29
Furthermore, the concept of AZOOR complex has been proposed, integrating MEWDS, AZOOR, PIC, MFC, AMN, AIBSE, and AAOR as a continuum with a common genetic autoimmune/inflammatory basis3).
Epidemiology in Japan
Section titled “Epidemiology in Japan”According to statistics from the Japanese Society for Ocular Inflammation, the proportion of each white dot syndrome among all uveitis cases is as follows2).
| Disease | Proportion of all uveitis |
|---|---|
| MEWDS | Approximately 1–2% (domestic reports) |
| APMPPE | Rare (no clear annual incidence statistics) |
| PIC | Rare |
| Serpiginous choroiditis | Approximately 0.3% |
| Birdshot retinochoroidopathy | Rare (more common in Caucasians, extremely rare in Japan) |
| AZOOR | Rare (increasing reports in recent years) |
2. Common clinical features
Section titled “2. Common clinical features”Although symptoms vary by disease, the following clinical features are common to the white dot syndromes1, 2).
Common patterns of subjective symptoms
Section titled “Common patterns of subjective symptoms”- Decreased visual acuity: ranges from mild (MEWDS, AZOOR) to severe (serpiginous choroiditis, PIC with CNV)
- Photopsia: the most common symptom reflecting outer retinal/photoreceptor damage
- Scotoma/visual field defect: often paracentral or central scotomas corresponding to lesion location
- Metamorphopsia: occurs with macular lesions or CNV
Presence of anterior segment inflammation
Section titled “Presence of anterior segment inflammation”| Disease group | Anterior chamber/vitreous inflammation |
|---|---|
| MEWDS, APMPPE, PIC | Usually none (APMPPE may have mild) |
| MFC (MFCwP) | Anterior chamber inflammation + vitritis present (differentiating from PIC) |
| Birdshot | No to minimal anterior chamber inflammation, vitritis present |
| Serpiginous choroiditis | Anterior chamber inflammation and vitritis are usually mild |
| AZOOR | Usually none |
3. Differential diagnosis approach
Section titled “3. Differential diagnosis approach”Age, Sex, Unilateral vs Bilateral, and Recurrence-Based Classification
Section titled “Age, Sex, Unilateral vs Bilateral, and Recurrence-Based Classification”Young women, unilateral, spontaneous resolution
Young to middle-aged women, bilateral, CNV risk
Young to middle-aged, bilateral, acute onset
- Peak incidence in 20s–30s (mean age 25), no sex difference
- Bilateral, acute, tendency for spontaneous resolution
- Watch for cerebral vasculitis (urgent evaluation if neurological symptoms appear)
Middle-aged and older adults, bilateral, chronic progressive
Birdshot・Serpiginous choroiditis
- Birdshot: 40s–60s, slightly more common in women
- Serpiginous: 30s–50s, slightly more common in men
- Both diseases are chronic, recurrent, and require long-term immunosuppression
- HLA-A29 positivity rate in Birdshot (Caucasians): 80–98%
Differential Diagnosis Flowchart
Section titled “Differential Diagnosis Flowchart”眼底に白点状病変 │ ├─ 片眼性? │ ├─ YES → MEWDS・AZOOR・AMN を考慮 │ │ ↳ FA で初期過蛍光 → MEWDS │ │ ↳ 眼底ほぼ正常・ERG 異常 → AZOOR │ └─ NO(両眼性) │ ├─ 急性発症・後極部大型白斑? │ └─ YES → APMPPE(FA 蛍光逆転現象を確認) │ ├─ 後極部小病変・近視女性・硝子体炎なし? │ └─ YES → PIC を考慮(CNV 検索:OCTA 必須) │ ├─ 小病変・硝子体炎あり・周辺部にも病変? │ └─ YES → MFC(MFCwP)を考慮 │ ├─ 乳頭周囲から蛇行状に進展・男性多め? │ └─ YES → 蛇行状脈絡膜炎(結核除外が最優先) │ └─ 後極部散弾状病変・中高年・HLA-A29? └─ YES → Birdshot 網脈絡膜症4. Comparison of Major Diseases
Section titled “4. Comparison of Major Diseases”Main Comparison Table: 7-Disease Matrix
Section titled “Main Comparison Table: 7-Disease Matrix”| Item | APMPPE | MEWDS | PIC | MFC (MFCwP) | Birdshot | Serpiginous choroiditis | AZOOR |
|---|---|---|---|---|---|---|---|
| Typical age of onset | 20s–30s (average 25 years) | 20s–50s | 18–40s (average 36 years) | Average 30 years | 40s–60s | 30s–50s | Mainly mid-30s |
| Sex | No sex difference | Female predominance (1:4) | Female predominance (approx. 90%) | Female predominance (more common in white women) | Slightly more females | Slightly more males | Female predominance (approx. 75%) |
| Unilateral/Bilateral | Mostly bilateral | Unilateral (≥95%) | Mostly bilateral (80%) | Bilateral | Bilateral | Bilateral | Unilateral → bilateral progression (eventually 76% bilateral) |
| Main symptoms | Decreased vision, central scotoma, metamorphopsia | Decreased vision, photopsia, blurred vision | Scotoma, decreased vision, metamorphopsia | Floaters, decreased vision, photopsia | Decreased vision, night blindness, color vision abnormality | Decreased vision, paracentral scotoma | Photopsia, visual field defect (fundus nearly normal) |
| Characteristics of fundus white spots | Large cream-colored white spots in the posterior pole (1/4 to 1/2 disc diameter) | Pale gray-white multiple small spots from posterior pole to equator (100–200 μm) | Yellow-white small spots in the posterior pole (100–300 μm), 12–25 in number | Yellow-gray spots from posterior pole to periphery (45–350 μm), with vitreitis | Shotgun-like cream-colored spots from posterior pole to equator (1/4 to 1/2 disc diameter) | Geographic gray-yellow lesions extending tortuously from the peripapillary area | Fundus nearly normal (acute phase), outer layer atrophy in late phase |
| OCT findings | Ellipsoid zone disruption + outer retinal hyperreflectivity, partial atrophy after recovery | Ellipsoid zone marked disruption/disappearance (acute phase) → recovery | Sub-RPE hyperreflective elevation + EZ break (5-stage evolution) | Sub-RPE hyperreflectivity + EZ break (similar to PIC) | Choroidal lesions, cystoid macular edema, EZ disappearance indicates poor prognosis | Active phase: outer retinal hyperreflectivity, subretinal fluid. Scar phase: RPE atrophy | EZ (IS/OS) disappearance is the most important finding (corresponds to visual field defect) |
| FA findings | Early hypofluorescence → late hyperfluorescence (fluorescence reversal) | Persistent wreath-like hyperfluorescence from early phase | Active: early arterial hyperfluorescence → late leakage | Active: early hypofluorescence → late leakage | Lesion hyperfluorescence (no leakage) + vascular leakage and optic disc hyperfluorescence | Active: early hypofluorescence → late hyperfluorescence (leakage) | Usually normal or minimal abnormalities |
| ICGA Findings | Hypofluorescence in all phases (directly reflects choriocapillaris ischemia) | Late hypofluorescence (more extensive than white dots) | Mid-phase hypofluorescence, useful for detecting subclinical lesions | Early to all-phase hypofluorescence | Early/mid hypofluorescence → late isofluorescence (initial) → advanced: all-phase hypofluorescence | All-phase hypofluorescence (reflects choroidal circulatory disturbance) | Usually normal |
| FAF findings | Acute: hypo- or hyper-autofluorescence. Remission: hypo-autofluorescence | Acute: hyper-autofluorescence (multiple hyperfluorescent spots). After recovery: normalization | Active: hypo-autofluorescence (hypoAF), marginal hyper-autofluorescence halo | Active: hypo-autofluorescence | Peripapillary confluent hypo-autofluorescence (seen in 73%) | Active: hyper-autofluorescence margin + hypo-autofluorescence halo. Inactive: hypo-autofluorescence | Zonal hyper- to hypo-autofluorescence abnormalities |
| OCT-A findings | Choriocapillaris flow void (high concordance with FA/ICGA) | Choriocapillaris generally preserved (some transient flow void) | Choriocapillaris flow void (at sites of inflammatory lesions) | Choriocapillaris flow void | Haller layer flow void (early) → advanced: full-thickness flow void | Choriocapillaris flow void (severe) | Choriocapillaris generally preserved |
| Recurrence | Rare (basically single episode) | Recurrence in about 10% | High (chronic recurrent) | High (recurrent inflammatory episodes) | High (chronic with repeated relapses and remissions) | High (repeats at intervals of 3 months to 4 years) | Most stabilize within 6 months; some progress |
| CNV complication rate | Rare | Rare | 40–76% (high risk) | Up to 60% | Subretinal CNV: Rare | Up to 35% | Almost none |
| HLA associated | None | HLA-B51 (preliminary reports) | HLA-DR2, HLA-DRB1*15 | IL-10 haplotype association | HLA-A29 (80–98% in Caucasians) | HLA-B7, HLA-A2 (reported associations) | None (immunological predisposition) |
| Treatment strategy | Observation (spontaneous resolution), severe: steroids | Observation, severe: short-term steroids, CNV: anti-VEGF | Observation (no CNV), CNV: anti-VEGF + steroids, immunosuppressants | Steroids + immunomodulatory therapy, CNV: anti-VEGF | Steroids + mycophenolate mofetil / adalimumab (long-term) | Steroids + immunosuppressants (including alkylating agents), CNV: anti-VEGF | Observation, severe: steroid pulse |
| Visual prognosis | Good (often spontaneous resolution) | Good (caution for recurrence and CNV) | High risk of poor prognosis if CNV develops | Risk of poor prognosis due to CNV and macular edema | Without treatment, 16–22% have visual acuity ≤0.1 at 10 years | Irreversible if fovea involved; up to 25% of eyes have final visual acuity <20/200 | Mostly stable. Poor prognosis in cases with progressive outer layer damage |
Serpiginous choroiditis and birdshot retinochoroidopathy have the poorest visual prognosis. In serpiginous choroiditis, up to 25% of eyes have a final visual acuity of less than 20/200, and in birdshot, without treatment, 16–22% of patients have visual acuity of 0.1 or less over 10 years2, 4). PIC and MFC have a higher risk of poor outcomes when complicated by CNV. APMPPE and MEWDS have a strong tendency for spontaneous resolution and a good prognosis.
Detailed Clinical Features of Each Disease
Section titled “Detailed Clinical Features of Each Disease”APMPPE (Acute Posterior Multifocal Placoid Pigment Epitheliopathy)
Section titled “APMPPE (Acute Posterior Multifocal Placoid Pigment Epitheliopathy)”APMPPE commonly occurs in individuals in their 20s to 30s (average age 25) with no gender predilection. It is thought to be caused by occlusive vasculitis of the afferent arterioles of the choriocapillaris, and viral infections are suspected as triggers1, 2).
Prodromal symptoms and course
- About half of patients have cold-like symptoms (influenza, EB virus, chickenpox, streptococcal infection, etc.)
- Multiple cream-colored disc-shaped white spots, 1/4 to 1/2 optic disc diameter, appear in the posterior pole of both eyes
- The white spots begin to fade from the center within a few days and disappear after 7–12 days, leaving mild depigmentation
- Usually a single episode resolves spontaneously (recurrence is rare)
- Visual prognosis is generally good, but may be poor in severe cases or those progressing to geographic choroiditis
Specific complication: Cerebral vasculitis (MCAT)
APMPPE complicated by central nervous system vasculitis (MCAT: multiple cerebral arterial thrombosis) is a serious complication. If headache, fever, or neurological symptoms appear, emergency brain MRI and MRA should be performed. In cases with cerebral vasculitis, methylprednisolone pulse therapy and collaboration with neurology are necessary2).
Relationship with the placoid chorioretinitis spectrum
APMPPE, together with PPM (persistent placoid maculopathy) and RPC (relentless placoid chorioretinitis), forms the “placoid chorioretinitis spectrum,” with choriocapillaris ischemia as a common pathological basis5).
MEWDS (Multiple Evanescent White Dot Syndrome)
Section titled “MEWDS (Multiple Evanescent White Dot Syndrome)”MEWDS predominantly affects women in their 20s to 50s (male-to-female ratio 1:4) and is characterized by unilateral, acute onset, and spontaneous resolution.
Characteristic clinical features
- Pale gray-white multiple small spots (100–200 μm) at the deep retinal to RPE level in the posterior pole to the equator
- Foveal granularity: observed in 74–96% of patients, may be the only residual finding after white spots disappear. Near-infrared FAF (NIR-FAF) shows a characteristic pattern9)
- Orange-dot appearance: characteristic finding on fundus photography and near-infrared fundus imaging
- Ivory lesion: A faint, blurred white change at the posterior pole of the fundus
- Approximately 50% of cases are preceded by cold-like prodromal symptoms
- Annual incidence is about 0.22 per 100,000 people, with recurrence in 10% of cases
Wreath-like hyperfluorescence on FA
Characteristic wreath-like hyperfluorescence from the early phase of FA is a key point in diagnosing MEWDS. The white spot lesions show hyperfluorescence from the early phase of FA and do not enlarge in the late phase. This early hyperfluorescence is an important distinguishing feature from the early hypofluorescence (fluorescence reversal phenomenon) of APMPPE1, 9).
Continuity with AZOOR complex
MEWDS primarily involves transient destruction of the ellipsoid zone (IS/OS line) of photoreceptors and is understood as a disease belonging to the AZOOR complex. OCT can confirm disruption or loss of the ellipsoid zone in the acute phase, which improves with visual recovery3).
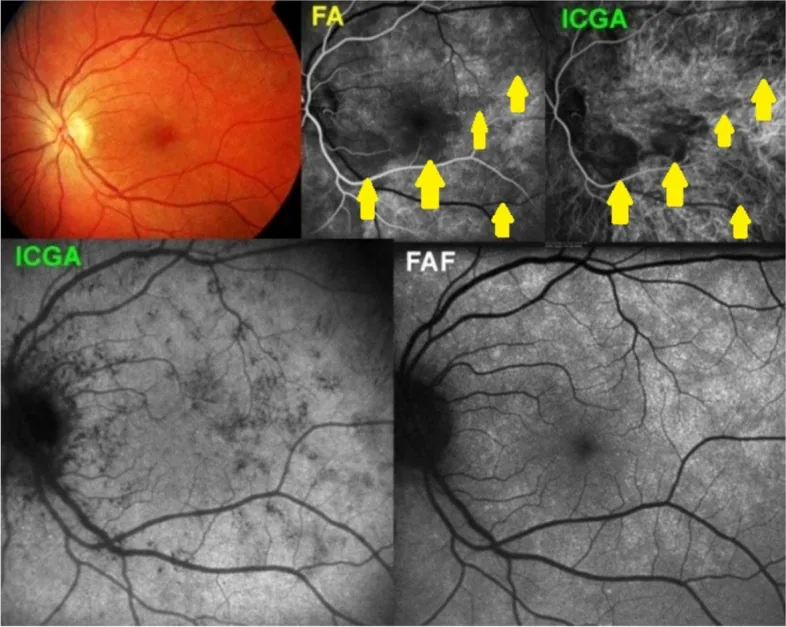
PIC (Punctate Inner Choroidopathy)
Section titled “PIC (Punctate Inner Choroidopathy)”PIC predominantly occurs in young women (approximately 90%) aged 18–40 years with myopia (average around -5 D).
Characteristic clinical features
- Yellow-white small spots of 100–300 μm confined to the posterior pole, usually 12–25 in number
- No anterior chamber inflammation or vitritis (this is the most important distinguishing feature from MFC)
- Active lesions can be seen on OCT as hyperreflective elevations beneath the RPE
- Scarring leaves small atrophic lesions
CNV complication (40–76%) is the major clinical problem
The most important complication of PIC is CNV, with reported rates of 40–76%7, 8). CNV is more likely to occur due to the following factors:
- Bruch membrane fragility due to myopic choroidal thinning
- Bruch membrane disruption due to sub-RPE inflammation
- Increased local production of inflammatory cytokines (e.g., VEGF)
OCT-A has been shown to be more sensitive than FA for CNV screening, and regular OCT-A monitoring is recommended. Sudden worsening of metamorphopsia is a sign of CNV development and requires prompt evaluation.
Association with systemic diseases
Coexistence of PIC and sarcoidosis has been reported; in cases with multiple lung lesions, perform chest CT, serum ACE, and lysozyme tests. Association with HLA-DR2 and HLA-DRB1*15 has also been reported3).
MFC (Multifocal Choroiditis, MFCwP)
Section titled “MFC (Multifocal Choroiditis, MFCwP)”MFC (multifocal choroiditis with panuveitis; MFCwP) is on the same spectrum as PIC, but the key distinguishing feature is the presence of vitritis and anterior chamber inflammation7).
Characteristic clinical features
- Multiple yellow-gray spots measuring 45–350 μm appear not only in the posterior pole but also in the mid-periphery.
- Characterized by a chronic recurrent course (repeated inflammatory episodes)
- Epiretinal membrane (ERM) is frequently associated (up to 35%), affecting long-term visual prognosis
- In some cases, inflammation cannot be controlled without immunosuppressive therapy
Treatment considerations
MFC tends not to resolve spontaneously, and many cases require long-term immunomodulatory therapy. When steroids alone are insufficient, methotrexate (MTX), azathioprine (AZA), or mycophenolate mofetil (MMF) are used. When CNV is present, a dual approach with anti-VEGF therapy and immunomodulatory therapy is important7, 8).
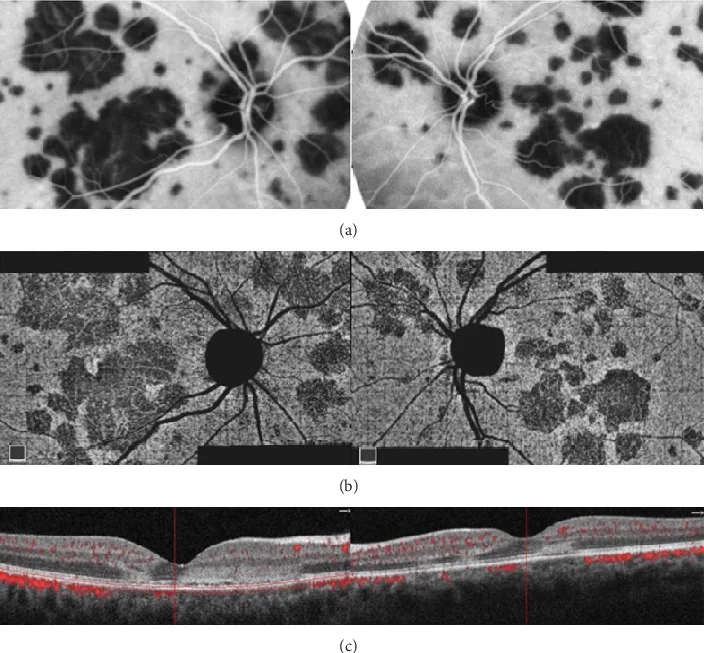
Birdshot Chorioretinopathy (Birdshot Retinochoroidopathy)
Section titled “Birdshot Chorioretinopathy (Birdshot Retinochoroidopathy)”Birdshot typically occurs in middle-aged to older adults aged 40–60 (average 50s), with a slight female predominance (1.5:1). It is more common in Caucasians and has one of the strongest genetic associations among known diseases, linked to HLA-A29 (relative risk 50–224 times in Caucasians)4).
Characteristic fundus findings
- Multiple cream-colored spots (1/4 to 1/2 disc diameter) resembling birdshot, symmetrically distributed from the posterior pole to the equator
- Spots transition to scar lesions without pigmentation
- May be accompanied by retinal vasculitis and optic disc swelling
Characteristic functional changes
- Night blindness and color vision abnormalities: Appear early and may precede visual acuity loss
- Negative-type full-field ERG: Observed early, with a-wave amplitude decreasing as the disease progresses
- Delayed 30-Hz flicker ERG: The most sensitive indicator for monitoring activity; can detect abnormalities earlier than visual acuity loss17)
Precautions in Japanese patients
Because the frequency of HLA-A29 is low in Japanese individuals, the diagnostic sensitivity of HLA-A29 is limited. Diagnosis should emphasize clinical findings from the SUN 2021 classification criteria (birdshot fundus lesions, minimal anterior chamber inflammation, and presence of vitritis)10).
Long-term complications
- Cystoid macular edema (CME): main cause of vision loss
- Optic disc swelling and optic atrophy
- With steroid implants (fluocinolone), intraocular pressure elevation occurs in up to 40% of cases, sometimes requiring trabeculectomy
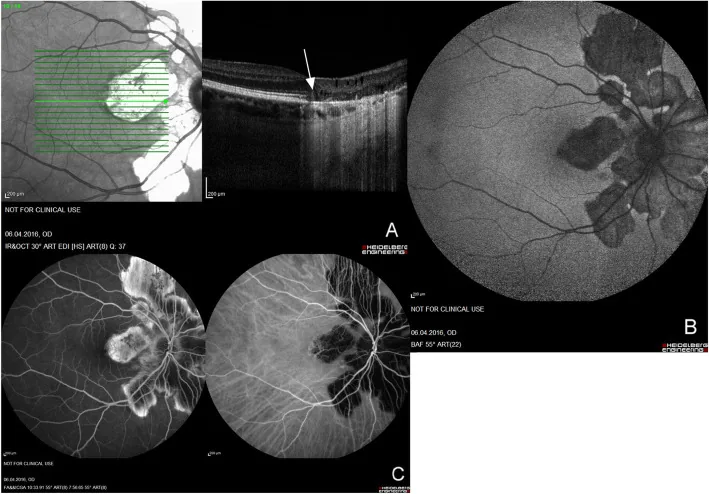
Serpiginous choroidopathy
Section titled “Serpiginous choroidopathy”Serpiginous choroidopathy is a bilateral chronic choroiditis that occurs in the 30s to 50s (slightly more common in men), characterized by geographic gray-yellow lesions that extend in a serpentine pattern from the peripapillary area.
Characteristic progression pattern
- Starts centripetally from the peripapillary area, with the lesion margin gradually expanding in a serpentine pattern
- Active phase: a gray-white border appears at the lesion margin
- Scar phase: becomes fixed as chorioretinal atrophic lesions
- During recurrence, new inflammation always appears from the margin of the existing scar (this is characteristic)
- The recurrence interval varies greatly among individuals, ranging from 3 months to 4 years.
Most important: Differentiation from tuberculosis-associated type (SLC)
Tuberculous serpiginous-like choroiditis (SLC) closely resembles serpiginous choroidopathy in imaging findings, but the treatment strategy is fundamentally different:
| Differentiating feature | Serpiginous choroidopathy | Tuberculosis-associated type (SLC) |
|---|---|---|
| Lesion distribution | Peripapillary, centripetal | Posterior pole to periphery, multifocal |
| IGRA/TST | Negative | Positive |
| Lesion shape | Geographic, continuous | Multiple discontinuous small lesions common |
| Treatment | Steroids + immunosuppressants | Antitubercular drugs are essential |
Since immunosuppressive drugs can markedly worsen tuberculosis in SLC, IGRA (QuantiFERON) testing before treatment is absolutely essential2).
Management of CNV (up to 35%)
In serpiginous choroidopathy, CNV occurs in up to 35% of cases and can cause irreversible vision loss if it involves the fovea. Intravitreal injection of anti-VEGF therapy (bevacizumab, ranibizumab) is effective18).
AZOOR (Acute Zonal Occult Outer Retinopathy)
Section titled “AZOOR (Acute Zonal Occult Outer Retinopathy)”AZOOR is a disease concept proposed by Gass in 1992. It is an outer retinopathy that causes sudden vision loss, visual field defects, and photopsia despite a nearly normal fundus3).
Concept of AZOOR complex
The AZOOR complex, proposed by Jampol et al., is a concept that understands MEWDS, AZOOR, PIC, MFC, AMN (acute macular neuroretinopathy), AIBSE, and AAOR as a continuum with a common genetic autoimmune/inflammatory basis3).
Characteristic clinical features
- Frequently occurs in young women in their 20s to 50s with myopia.
- Photopsia (light perception) often appears in the early stage (especially light seen as带状 or arc-shaped)
- Starts unilaterally, eventually becomes bilateral in 76% of cases
- Fundus is almost normal in the acute phase (characterized by discrepancy between visual acuity loss and fundus findings)
- Visual field defects show irregular带状 patterns (often continuous with blind spots)
- May be associated with autoimmune diseases (Hashimoto’s disease, multiple sclerosis)
OCT and ERG are key to diagnosis
- The most important finding on OCT is disappearance or obscuration of the ellipsoid zone (IS/OS line).
- Functional recovery cannot be expected in areas where the outer layer has disappeared on OCT (also useful for prognosis prediction).
- Multifocal ERG can detect amplitude reduction even when the fundus appears normal (multifocal ERG is more sensitive than full-field ERG).
- Infrared FAF may delineate the border between the lesion and normal area.
Treatment and Prognosis
There is no established treatment for AZOOR. Mild cases may only require observation, but severe cases (with decreased visual acuity or extensive visual field defects) are treated with methylprednisolone pulse therapy (1,000 mg × 3 days) followed by oral prednisolone2). Most cases stabilize within 6 months, but visual field defects in areas with residual outer layer damage do not recover.
Both AZOOR and retrobulbar optic neuritis present with nearly normal fundus and decreased visual acuity and visual field, so differentiation is necessary. Key points for differentiation are: ① AZOOR shows reduced multifocal ERG amplitudes, while retrobulbar optic neuritis has normal ERG; ② AZOOR exhibits irregular带状 or arcuate visual field defects, whereas retrobulbar optic neuritis often presents with central scotomas; ③ AZOOR usually has mild RAPD, while retrobulbar optic neuritis shows marked RAPD; ④ On OCT, AZOOR shows ellipsoid zone loss, while retrobulbar optic neuritis shows optic disc swelling or RNFL thinning, which aids differentiation2, 3).
5. Use of Imaging Tests
Section titled “5. Use of Imaging Tests”Role Allocation by Modality
Section titled “Role Allocation by Modality”Clearly understanding the role of each imaging test is essential for accurate diagnosis and activity assessment of white dot syndromes1, 5).
| Modality | Strongest indication/role |
|---|---|
| FA (fluorescein angiography) | Evaluation of leakage from retinal vessels, RPE, and choriocapillaris. Confirmation of fluorescence reversal in APMPPE. Confirmation of wreath-like hyperfluorescence in MEWDS. Evaluation of vasculitis (Birdshot). |
| ICGA (indocyanine green angiography) | Direct evaluation of choroidal circulation disorders. Detection of lesions earlier than FA (especially Birdshot, APMPPE, serpiginous). Detection of lesions more extensive than clinical findings (MEWDS, PIC). Most sensitive for detecting active lesions. |
| FAF (Fundus Autofluorescence) | Noninvasive assessment of RPE damage. Activity evaluation (APMPPE, serpiginous). Diagnosis of MEWDS (early white dots show hyperautofluorescence). Chronicity assessment of Birdshot (peripapillary hypoautofluorescence 73%) |
| OCT (Optical Coherence Tomography) | Ellipsoid zone evaluation (diagnostic finding in MEWDS, AZOOR). Five-stage evolution assessment of lesions (PIC). Evaluation of CNV and macular edema. Prognosis prediction (EZ loss → poor visual prognosis) |
| OCT-A | Noninvasive detection of choriocapillaris flow void (APMPPE, serpiginous, PIC). Early and sensitive detection of CNV (higher sensitivity than FA in PIC, MFC). Layer-specific choroidal blood flow assessment in Birdshot. Treatment effect monitoring |
| Multifocal ERG / Full-field ERG | Diagnosis of AZOOR (ERG amplitude reduction even with nearly normal fundus). Monitoring of Birdshot activity (30Hz flicker delay is most sensitive). Assessment of treatment efficacy. |
Disease-specific guide: “Which test first?”
Section titled “Disease-specific guide: “Which test first?””When MEWDS is suspected
When APMPPE is suspected
When PIC/MFC is suspected
When serpiginous choroiditis is suspected
The strategy differs by disease. In APMPPE and serpiginous choroiditis, ICGA more directly depicts choroidal circulatory disturbances, so performing ICGA simultaneously with or after FA deepens understanding of the pathology. In MEWDS, FA’s wreath-like hyperfluorescence is diagnostically important. However, since these are invasive tests, many findings can now be replaced by OCT-A, and the combination of FAF and OCT-A is used for initial evaluation1, 5).
Meaning of hypofluorescent lesions on ICGA
Section titled “Meaning of hypofluorescent lesions on ICGA”Hypofluorescence of white dot syndrome lesions on ICGA is a direct reflection of choroidal blood flow disruption (choriocapillaris occlusion)1). ICGA is more sensitive than FA for evaluating choroidal circulation and has the following characteristics:
- Total-phase hypofluorescence: Seen in APMPPE, serpiginous choroiditis, PIC, and MFC. Reflects severe occlusion of choriocapillaris ischemia.
- Late hypofluorescence (normal on FA): Late hypofluorescence on ICGA in MEWDS is explained by ICG uptake changes due to RPE abnormality rather than choriocapillaris involvement (choriocapillaris is generally preserved on OCT-A)1).
- Haller layer flow void → full-thickness flow void in advanced stage: A two-stage progression pattern unique to Birdshot, starting from the choroidal stroma and extending to the choriocapillaris14).
ICGA is superior to FA and OCT-A in detecting clinically invisible subclinical lesions, and particularly in MEWDS and PIC, it reveals more extensive choroidal lesions than white spots1, 15).
FAF hypo- and hyperfluorescence pattern-based disease map
Section titled “FAF hypo- and hyperfluorescence pattern-based disease map”FAF (fundus autofluorescence) patterns reflect the metabolic state of the RPE and are useful for diagnosing white dot syndromes and assessing disease activity16).
| FAF pattern | Disease/stage | Meaning |
|---|---|---|
| High autofluorescence (hyper-AF) | MEWDS acute phase, Serpiginous active edge | Accumulation of photoreceptor degeneration products (e.g., A2E) in RPE |
| Low autofluorescence (hypo-AF) | APMPPE scar stage, PIC active lesion, Birdshot peripapillary | RPE loss or dysfunction |
| Central hypo-AF with peripheral hyper-AF halo | Serpiginous active border, PIC | RPE damage pattern at active border |
| Zonal abnormal AF | AZOOR | Corresponds to distribution of photoreceptor outer segment damage |
| Foveal granular hyperautofluorescence | MEWDS (NIR-FAF) | Noninvasive visualization of foveal granularity |
In Birdshot, peripapillary confluent hypoautofluorescence is observed in 73% of cases, useful as an indicator of chronicity17).
Ellipsoid zone (EZ) defect patterns on OCT
Section titled “Ellipsoid zone (EZ) defect patterns on OCT”Evaluation of the ellipsoid zone (EZ, formerly the IS/OS line) plays a central role in assessing activity and predicting prognosis in white dot syndromes1).
| EZ findings | Disease/stage | Prognosis |
|---|---|---|
| Marked EZ disruption → recovery | MEWDS acute phase → recovery phase | Good (EZ recovery linked to visual acuity recovery) |
| EZ loss (corresponding to lesion) | Active phase of AZOOR | No functional recovery in the lost area |
| EZ disruption + outer retinal hyperreflectivity | Acute phase of APMPPE | Partial residual atrophy after recovery |
| Sub-RPE hyperreflective elevation + EZ disruption | PIC/MFC (5-stage evolution) | Poor prognosis with CNV complication |
| EZ loss (with cystoid macular edema) | Birdshot advanced stage | Risk factor for poor visual prognosis |
Choriocapillaris flow void on OCT-A shows high concordance with FA and ICGA findings (particularly useful in APMPPE and serpiginous choroiditis) 5, 13).
FA Fluorescence Patterns: Filling Delay vs Hyperfluorescence vs Leakage
Section titled “FA Fluorescence Patterns: Filling Delay vs Hyperfluorescence vs Leakage”| FA Pattern | Disease | Clinical Significance |
|---|---|---|
| Early hypofluorescence → late hyperfluorescence (fluorescence reversal phenomenon) | APMPPE | Choriocapillaris ischemia. Early filling defect → delayed dye leakage from surrounding tissue. |
| Early wreath-like hyperfluorescence | MEWDS | Directly reflects RPE/photoreceptor damage. Differentiating point: does not enlarge in late phase |
| Early hypofluorescence → late leakage | Active serpiginous/MFC | Evidence of active choroiditis |
| Early arterial hyperfluorescence → late leakage | Active PIC | Suggests inflammatory CNV |
| Vascular leakage + optic disc hyperfluorescence (no leakage) | Birdshot | Direct evidence of retinal vasculitis |
| Usually normal to minimal | AZOOR | Characterized by dissociation: visual field defects and ERG abnormalities despite negative FA |
6. General Treatment
Section titled “6. General Treatment”Selection of Treatment Strategy
Section titled “Selection of Treatment Strategy”Treatment for white dot syndromes varies greatly depending on the natural course of the disease, severity, and presence of CNV.
Observation (no treatment needed) as a principle
Section titled “Observation (no treatment needed) as a principle”- Strong tendency for spontaneous resolution; often recovers without specific treatment2)
- MEWDS: White dots disappear and vision recovers within weeks. Oral steroids only when severe or with optic disc edema.
- APMPPE: White spots regress in 7–12 days; visual prognosis is usually good.
Diseases where steroid therapy is the mainstay
Section titled “Diseases where steroid therapy is the mainstay”APMPPE (severe cases, with papillitis) · PIC (active foveal lesions) · AZOOR (severe cases)
- Start with prednisolone 30–60 mg/day and taper gradually
- If APMPPE is complicated by cerebral vasculitis, methylprednisolone pulse therapy and collaboration with neurology are required
- Severity criteria for AZOOR: corrected visual acuity in the better eye < 0.3 (Japanese Ophthalmological Society diagnostic guidelines)
Diseases requiring long-term immunosuppressive therapy
Section titled “Diseases requiring long-term immunosuppressive therapy”Birdshot retinochoroidopathy, serpiginous choroiditis, MFC
Birdshot (long-term management)
- Start prednisolone 0.5–1 mg/kg/day
- Mycophenolate mofetil (MMF) 2–3 g/day (first-line immunosuppressant)
- Methotrexate (MTX) 10–25 mg/week
- Azathioprine (AZA) 1–3 mg/kg/day
- Refractory cases: adalimumab (93.2% used as first-line biologic agent)
- Without treatment, 16–22% have visual acuity ≤0.1 at 10 years
Serpiginous choroiditis (long-term management)
- Prednisolone 40–80 mg/day initially (tapered)
- Azathioprine 1–2.5 mg/kg/day (first-line maintenance therapy)
- Mycophenolate mofetil / methotrexate (alternatives)
- Refractory cases: chlorambucil (most potent; ≤0.2 mg/kg/day, weekly blood tests mandatory)
- Biologics: adalimumab (91.0% recommend as first-line)
- Exclude tuberculosis (if IGRA positive, start anti-tuberculosis drugs first) before initiating immunosuppressants
Immunomodulatory therapy for MFC
Section titled “Immunomodulatory therapy for MFC”Since MFC follows a chronic relapsing course, immunomodulatory drugs are often needed as maintenance therapy after steroid tapering.
| Drug | Dosage guide | Notes |
|---|---|---|
| Methotrexate (MTX) | 10–25 mg/week | Concomitant folic acid 1 mg/day. Monitor hepatotoxicity. |
| Azathioprine (AZA) | 1–3 mg/kg/day | TPMT activity testing recommended. Monitor for myelosuppression. |
| Mycophenolate mofetil (MMF) | 1–3 g/day | Gastrointestinal symptoms are common side effects |
| Cyclosporine (CsA) | 3–5 mg/kg/day | Renal function and blood pressure monitoring required |
| Adalimumab | 40 mg every 2 weeks (subcutaneous injection) | Refractory/steroid-dependent cases. Tuberculosis screening mandatory. |
Management of CNV Complication
Section titled “Management of CNV Complication”CNV is the most important complication affecting visual prognosis, and it occurs particularly frequently in PIC, MFC, and serpiginous choroiditis.
| Disease | CNV complication rate | Treatment |
|---|---|---|
| PIC | 40–76% | Intravitreal anti-VEGF injection (bevacizumab, ranibizumab, aflibercept) + steroids. OCTA PRN strategy. |
| MFC | Up to 60% | Intravitreal anti-VEGF injection + immunomodulatory therapy |
| Serpiginous choroiditis | Up to 35% | Intravitreal anti-VEGF (bevacizumab, ranibizumab) injection |
| MEWDS/APMPPE | Rare | Anti-VEGF (if CNV confirmed) |
| Birdshot | Rare | Systemic immunosuppression + anti-VEGF if CNV present |
Inflammatory CNV (iCNVM) differs from CNV in age-related macular degeneration; controlling the underlying inflammation is also important for preventing CNV recurrence. For PIC and MFC, a bidirectional approach with anti-VEGF and steroids (or immunosuppressants) is considered effective, and anti-VEGF monotherapy leaves a risk of recurrence7, 8).
Precautions in Treatment
Section titled “Precautions in Treatment”| Precaution | Relevant Disease |
|---|---|
| Prioritize exclusion of tuberculosis (before immunosuppression) | Serpiginous choroiditis, MFC |
| Consider HLA-A29 (diagnosis) | Birdshot (low sensitivity in Japanese) |
| Risk of intraocular pressure elevation with steroid implants | Birdshot (trabeculectomy required in up to 40%) |
| Bone marrow suppression and malignancy risk with chlorambucil | Serpiginous choroiditis (weekly blood tests mandatory) |
| Flare-up after COVID-19 infection or vaccination | PIC・MEWDS (flare-up reported) |
| Emergency management when cerebral vasculitis is present | APMPPE (headache, neurological symptoms → urgent brain MRI) |
The essential tests to be checked before starting immunosuppressive drugs for white dot syndromes are as follows: ① Exclusion of tuberculosis by IGRA (QuantiFERON) (highest priority in serpiginous choroiditis), ② Viral hepatitis screening by HBs antigen, HBc antibody, and HCV antibody (to prevent reactivation due to immunosuppression), ③ Chest X-ray/CT (to exclude tuberculosis and sarcoidosis), ④ Complete blood count and liver/kidney function tests (to confirm baseline values). For biologic agents such as adalimumab, tuberculosis screening is also mandatory according to the drug package insert 19, 20).
Inflammatory CNV (iCNVM) differs from age-related macular degeneration in that CNV may spontaneously regress when inflammation is controlled. Generally, a PRN (pro re nata) strategy is used, with administration each time active CNV is confirmed on OCT-A. Initial loading doses (3 consecutive injections) may be given, but combining with immunosuppressive therapy may reduce the number of required injections. Anti-VEGF alone carries a high risk of recurrence, and a bidirectional approach addressing both inflammation control and anti-VEGF is important 7, 8).
6.5 Association with Systemic Diseases
Section titled “6.5 Association with Systemic Diseases”Each disease in the white dot syndromes is known to be associated with specific systemic diseases or infections, and systematic exclusion workup before treatment is important.
| Disease | Associated Systemic Disease/Condition | Clinical Significance |
|---|---|---|
| APMPPE | Cerebral vasculitis (MCAT), streptococcal infection, EB virus | Headache, neurological symptoms → urgent brain MRI |
| Birdshot | HLA-A29 (80–98% in Caucasians), similar to sarcoidosis | HLA testing aids diagnosis (low sensitivity in Japanese) |
| Serpiginous choroiditis | Tuberculosis (SLC) · HLA-B7/A2 | IGRA positive → Anti-tuberculosis drugs first in principle |
| PIC | Sarcoidosis · HLA-DRB1*15 | Consider chest CT and ACE measurement |
| MFC | IL-10 haplotype, EBV, sarcoidosis | Re-evaluate systemic workup for chronic recurrent cases |
| MEWDS | COVID-19 infection, post-vaccination, HLA-B51 | SARS-CoV-2 infection acts as an immune trigger |
| AZOOR | Hashimoto’s disease, multiple sclerosis, autoimmune diseases | Consider thyroid function and autoantibody tests |
7. Special Notes and Latest Findings for Each Disease
Section titled “7. Special Notes and Latest Findings for Each Disease”APMPPE and the Placoid Chorioretinitis Spectrum
Section titled “APMPPE and the Placoid Chorioretinitis Spectrum”APMPPE is now understood as part of the “placoid chorioretinitis spectrum” along with persistent placoid maculopathy (PPM) and relentless placoid chorioretinitis (RPC). In these three diseases, OCT-A shows a common pattern of choriocapillaris flow void, indicating that choriocapillaris ischemia is a shared pathological basis 5).
Klufas et al. (2017) reported that OCT-A detects choriocapillaris flow void with high agreement with FA and ICGA in APMPPE, PPM, and RPC, supporting the concept of the placoid chorioretinitis spectrum 5).
MEWDS and Foveal Granularity
Section titled “MEWDS and Foveal Granularity”Foveal granularity is a diagnostic finding seen in 74–96% of MEWDS cases and may remain as the only sign after white spots disappear. Near-infrared FAF (NIR-FAF) reveals a characteristic foveal granular pattern 9).
Disease Spectrum of PIC and MFC
Section titled “Disease Spectrum of PIC and MFC”PIC and MFC (MFCwP) share a common genetic background (IL-10 haplotype, HLA-DRB1*15) and are considered different phenotypes of the same disease spectrum. The main differentiating points are the presence or absence of vitritis and anterior chamber inflammation, as well as the distribution of lesions1, 3).
| Differentiating point | PIC | MFC (MFCwP) |
|---|---|---|
| Vitritis | Absent | Present (important differentiating point) |
| Anterior chamber inflammation | None | Mild |
| Lesion distribution | Limited to posterior pole | Posterior pole + mid-periphery |
| Lesion size | 100–300 μm | 45–350 μm |
| CNV complication rate | 40–76% | Up to 60% |
Birdshot and HLA-A29
Section titled “Birdshot and HLA-A29”The association between Birdshot and HLA-A29 is one of the strongest genetic associations of any known disease, with a relative risk of 50–224 times in white patients4). However, since HLA-A29 carriers are rare in Japanese individuals, the sensitivity of HLA-A29 positivity in diagnosis is limited. Diagnosis should emphasize the clinical findings of the SUN 2021 classification criteria (fundus findings, minimal anterior chamber inflammation, vitritis)10).
AZOOR complex and viral triggers
Section titled “AZOOR complex and viral triggers”AZOOR is thought to develop when environmental triggers such as viral infection, vaccination, or drugs are added to genetic predisposition (e.g., IL-10 haplotypes), and is understood as part of the AZOOR complex along with MEWDS, PIC, AMN, and AIBSE3). Cases of MEWDS after COVID-19 infection or vaccination have been increasing worldwide, suggesting that SARS-CoV-2 may act as an immune trigger11).
Yes. Multiple reports have described the onset or recurrence of MEWDS, PIC, and serpiginous choroiditis after COVID-19 infection, suggesting that SARS-CoV-2 infection may act as an immune trigger11, 12). For MEWDS, a systematic review of 27 cases after COVID-19 vaccination found that mRNA vaccines (Pfizer-BioNTech) were most common. Patients with a history of these conditions should consult an ophthalmologist about monitoring before and after vaccination.
The recommended priority order for initial testing when white dot syndrome is suspected is as follows: ① FAF + OCT (noninvasive, allows initial evaluation of almost all diseases; can detect high AF in MEWDS, ellipsoid zone assessment, and RPE elevation in PIC) → ② OCT-A (early detection of CNV, evaluation of choriocapillaris flow void) → ③ FA + ICGA (for definitive diagnosis and activity assessment when needed). If serpiginous choroiditis is suspected, IGRA (to rule out tuberculosis) should be prioritized before FA. For suspected AZOOR, ERG (multifocal ERG) is essential1, 2).
8. Related Articles
Section titled “8. Related Articles”9. References
Section titled “9. References”- Testi I, Modugno RL, Pavesio C. Multimodal imaging supporting the pathophysiology of white dot syndromes. J Ophthalmic Inflamm Infect. 2021;11:32.
- 日本眼炎症学会・日本眼科学会. ぶどう膜炎診療ガイドライン(13〜18節:APMPPE・MEWDS・PIC・多発性脈絡膜炎・バードショット・蛇行状脈絡膜炎・AZOOR). 日本眼科学会雑誌. 2019;123(6):635-696.
- Jampol LM, Becker KG. White spot syndromes of the retina: a hypothesis based on the common genetic hypothesis of autoimmune/inflammatory disease. Am J Ophthalmol. 2003;135(3):376-379.
- Agrawal R, et al. The role of HLA-A29 in birdshot chorioretinopathy and immune checkpoint inhibitor-related uveitis. Am J Ophthalmol. 2025. doi:10.1016/j.ajo.2024.01.007
- Klufas MA, Phasukkijwatana N, Iafe NA, et al. Optical coherence tomography angiography reveals choriocapillaris flow reduction in placoid chorioretinitis. Ophthalmol Retina. 2017;1(1):77-91.
- Stattin M, Forster J, Ahmed D, Krepler K, Ansari-Shahrezaei S. Swept Source-Optical Coherence Tomography Angiography for Management of Secondary Choroidal Neovascularization in Punctate Inner Choroidopathy. Case Rep Ophthalmol. 2021;12:232-238.
- Spaide RF, Goldberg N, Freund KB. Redefining multifocal choroiditis and panuveitis and punctate inner choroidopathy through multimodal imaging. Retina. 2013;33(7):1315-1324.
- Leclaire MD, Clemens CR, Eter N, Mihailovic N. Choroidale Neovaskularisation infolge einer “punctate inner choroidopathy”, dargestellt mittels optischer Kohärenztomographie-Angiographie. Ophthalmologe. 2021;118:842-846.
- Mantovani A, Invernizzi A, Staurenghi G, Herbort CP Jr. Multiple evanescent white dot syndrome: a multimodal imaging study of foveal granularity. Ocul Immunol Inflamm. 2019;27(1):141-147.
- Standardization of Uveitis Nomenclature (SUN) Working Group. Classification criteria for birdshot chorioretinitis. Am J Ophthalmol. 2021;228:65-71.
- Chen N, Mandell M, Arjmand P. Multimodal imaging findings of multiple evanescent white dot syndrome in COVID-19 patients. IDCases. 2024;38:e02110.
- Seddigh S, Pinto A, Zaki AM, Gupta RR. Serpiginous choroiditis after COVID-19 infection. J Vitreoretinal Dis. 2025;9(2):246-252.
- Pakzad-Vaezi K, Khaksari K, Chu Z, Van Gelder RN, Wang RK, Pepple KL. Swept-source OCT angiography of serpiginous choroiditis. Ophthalmol Retina. 2018;2(7):712-719.
- Pepple KL, Chu Z, Weinstein J, Munk MR, Van Gelder RN, Wang RK. Use of en face swept-source optical coherence tomography angiography in identifying choroidal flow voids in 3 patients with birdshot chorioretinopathy. JAMA Ophthalmol. 2018;136(11):1288-1292. doi:10.1001/jamaophthalmol.2018.3474. PMID:30128478. PMCID:PMC6248174
- Khochtali S, Dridi T, Abroug N, et al. Swept-source optical coherence tomography angiography shows choriocapillaris flow reduction in multiple evanescent white dot syndrome. J Curr Ophthalmol. 2020;32(2):211-215.
- Yeh S, Forooghian F, Wong WT, et al. Fundus autofluorescence imaging of the white dot syndromes. Arch Ophthalmol. 2010;128(1):46-56.
- Minos E, Barry RJ, Southworth S, et al. Birdshot chorioretinopathy: current knowledge and new concepts in pathophysiology, diagnosis, monitoring and treatment. Orphanet J Rare Dis. 2016;11(1):61.
- Maleki A, Maldonado Cerda A, Garcia CM, et al. Chlorambucil combination therapy in refractory serpiginous choroiditis: a cure? Am J Ophthalmol Case Rep. 2021;21:101014.
- Niederer RL, Al-Janabi A, Engelbrecht C, et al. Immunomodulatory therapy prescribing practices for non-infectious uveitis: a survey of international experts. Br J Ophthalmol. 2024;108:482-489.
- Tomkins-Netzer O, et al. Treatment of non-infectious uveitis with biologics: a survey of the International Ocular Inflammation Society. Br J Ophthalmol. 2022;106:482-488.
