IOL displacement and dislocation
Diagnosis and surgical indications for IOL drop due to Zinn zonule rupture. Selection of intrascleral fixation (flange technique, forceps technique).
An intraocular lens (IOL) is an artificial lens permanently placed inside the eye after removing the cloudy lens during cataract surgery. An eye with an IOL is called pseudophakia. Unlike correction with glasses or contact lenses, an IOL is directly integrated into the eye’s optical system, so there is no image magnification or minification, providing the most natural and physiological visual function.
The most widely used IOL today is the foldable IOL, which consists of an optic and haptics. The standard optic diameter is 6.0 mm, and it can be inserted through a 2.4–2.8 mm small incision in phacoemulsification (PEA) using an injector. In some difficult cases, a 7.0 mm large-optic IOL is used, which improves visibility during fundus examination and centration stability during extracapsular fixation.
Classification by material is shown below.
| Material | Characteristics | Main Indications |
|---|---|---|
| Hydrophobic acrylic | Less posterior capsule opacification. Beware of glistenings. | Current standard choice |
| Hydrophilic acrylic | High biocompatibility. Risk of calcium deposits (IOL opacification) with long-term use. | Special design IOL |
| Silicone | Good foldability. Not suitable for eyes with silicone oil tamponade. | Some PIOLs |
| PMMA | Rigid material. Requires enlarging the incision to 5–7 mm. | Special fixation IOLs, secondary implantation |
In terms of structure, there are 1-piece lenses where the optic and haptics are made of the same material, and 3-piece lenses where they are made of different materials. The 1-piece is typically used for in-the-bag fixation, while the 3-piece is suitable for out-of-the-bag fixation, scleral fixation, and suturing. Care must be taken because if a 1-piece lens is mistakenly fixated outside the bag (in the ciliary sulcus), the haptics may rub against the iris, causing iris pigment dispersion and prolonged inflammation.
Spherical IOLs cause spherical aberration, where the focal points of paraxial rays passing near the optical axis and peripheral rays are misaligned. In contrast, aspheric IOLs reduce spherical aberration by varying the curvature of each refractive surface to converge peripheral and paraxial rays to the same focal point. Currently, almost all IOLs adopt aspheric designs, improving contrast sensitivity. However, aspheric designs increase coma-like aberration with decentration or tilt, so in cases where the Zinn zonules are weak and IOL fixation is unstable, spherical IOLs may be chosen.
Tinted (yellow filter) IOLs reduce the transmittance of short-wavelength light (blue light), approximating the spectral transmittance of the adult human crystalline lens, and are expected to reduce the risk of retinal phototoxicity.
In 1981, Sanders et al. proposed the SRK formula (Sanders-Retzlaff-Kraff formula), systematizing IOL power calculation1). In 1984, Mazzocco developed the foldable silicone IOL, paving the way for small-incision surgery. Japan was the first country to approve foldable IOLs and has since played a pioneering role internationally in injector development. In 1953, Strampelli performed anterior chamber IOL implantation, but 70–80% of cases developed bullous keratopathy within a few years, leading to failure; however, this became a driving force for subsequent IOL design improvements.
The most widely used IOL, designed to focus at a single point. It is the standard option covered by insurance (selective medical treatment). If postoperative emmetropia is targeted, distance vision is good without glasses, but reading glasses are needed for near vision. Its strengths are the highest contrast sensitivity and minimal glare and halo at night.
The following options are available for postoperative presbyopia management:
→ For details, see Monofocal Intraocular Lens.
Multifocal IOLs provide good vision at multiple distances and are premium IOLs aimed at reducing spectacle dependence (spectacle independence). Because light efficiency is distributed, contrast sensitivity may decrease and glare/halo may occur; this should be thoroughly explained preoperatively. These are elective treatments and require patient out-of-pocket payment.
Focuses on two points: distance and near. It was somewhat unsuitable for intermediate distances (e.g., computer work at 50–80 cm). Types include diffractive (AcrySof IQ ReSTOR, TECNIS Multifocal) and refractive.
In diffractive types, the 0th-order diffracted light focuses on distance and the 1st-order on near, with higher-order diffracted light (about 18%) causing reduced high-frequency contrast sensitivity. Apodized designs improve distance vision in dim light by reducing step heights at the pupil periphery.
A meta-analysis including 8 RCTs showed that multifocal IOLs were superior to monofocal IOLs in uncorrected near visual acuity (UCNVA, proportion of 6/6 or better: RR 0.20, 95% CI 0.07–0.58, 782 eyes) and spectacle independence rate (RR 0.63, 95% CI 0.55–0.73, 1,000 eyes). However, the frequency of halos (RR 3.58, 95% CI 1.99–6.46, 662 eyes) was significantly higher with multifocal IOLs. 3)
Trifocal IOLs focus on three points: distance, intermediate, and near, and are currently the mainstream multifocal IOLs. The first trifocal IOL was clinically introduced in 20104), and current representative products include AcrySof IQ PanOptix (Alcon), AT LISA tri (Carl Zeiss), FineVision (PhysIOL), and TECNIS Synergy (J&J Vision).
In a meta-analysis of 22 trials involving 2,200 eyes, trifocal IOLs were superior to EDOF IOLs in near visual acuity (UCNVA: MD = 0.12 logMAR, p < 0.00001) and spectacle independence rate (OR = 0.26, p = 0.02). Meanwhile, there were no significant differences between the two groups in distance visual acuity (UDVA) and intermediate visual acuity (UIVA), and the Quality of Vision score was significantly higher for trifocal IOLs (MD = 1.24, p = 0.03).5)
In a systematic review of trifocal IOLs, trifocal IOLs showed significantly better intermediate visual acuity compared to bifocal IOLs (DCIVA: MD −0.16 logMAR, 95% CI −0.22 to −0.10), but there were no significant differences in CDVA, distance visual acuity, near visual acuity, contrast sensitivity, or patient satisfaction.4)
Extended Depth of Focus (EDOF) IOLs distribute light over a continuous focal range rather than a single focus, providing a wide depth of focus from distance to intermediate. The first product to receive CE marking appeared in 20144). Representative products include TECNIS Symfony (J&J Vision), TECNIS Eyhance, and AcrySof IQ Vivity (Alcon).
In the aforementioned meta-analysis (22 trials), EDOF IOLs were slightly superior to trifocal IOLs in corrected distance visual acuity (CDVA: MD = −0.01 logMAR, p = 0.01), and there was no significant difference in the frequency of glare and halos between the two groups. 5) The ESCRS guidelines recommend EDOF IOLs as “an option for patients who prioritize intermediate vision while minimizing optical side effects.” 4) In a US registration trial evaluating the EDOF IOL (AcrySof IQ Vivity), the best-corrected distance visual acuity (BCVA) was equivalent to that of the monofocal control group (monocular CDVA 0.00 logMAR), and the reduction in mesopic contrast sensitivity was moderate. 17)
→ For details, see Multifocal Intraocular Lens.
Toric IOLs are designed to correct regular corneal astigmatism. The optic has markings along the flat meridian, and these marks are aligned with the steep corneal meridian during capsular bag fixation. For every 1° of axis misalignment, the corrective effect decreases by approximately 3.3%. A 30° misalignment not only eliminates the corrective effect but may also worsen visual function compared to a non-toric IOL.
General indications for toric IOL (varies by institution):
The ESCRS guidelines (2024) recommend considering a toric IOL in eyes with corneal astigmatism of 1.0 D or more, and there is strong evidence (GRADE ++) for 2.0 D or more. 4) A meta-analysis of 13 trials showed that toric IOLs were superior to non-toric IOLs (with or without relaxing incisions) in postoperative UDVA (MD −0.07 logMAR, 95% CI −0.10 to −0.04) and the rate of not achieving 20/25 (RR 0.59, 95% CI 0.50 to 0.70). 14)
Cases with zonular weakness, posterior capsule rupture, or insufficient pupil dilation are often contraindicated because accurate axis alignment is difficult. For calculation, use each manufacturer’s online calculator or the built-in Barrett Toric formula or Haigis-T formula.
→ For details, see Toric intraocular lens.
This is an IOL fixed to the posterior surface of the iris or ciliary sulcus while preserving the natural lens, and is a form of refractive surgery. A representative example is the ICL (Implantable Collamer Lens; EVO+ ICL), a posterior chamber PIOL made of collamer material with high biocompatibility. It does not ablate the cornea, is reversible, and can be used for a wide range of moderate to high myopia.
The Japanese Ophthalmological Society’s Refractive Surgery Guidelines (8th edition) sets the recommended age for phakic intraocular lens surgery at 21 to 45 years, and the indicated refractive range at myopia of 6 D or more. Caution is required for moderate myopia of 3 to less than 6 D and high myopia exceeding 15 D. In addition to the same contraindications as excimer laser surgery (active external ocular inflammation, cataract, uveitis, etc.), shallow anterior chamber, corneal endothelial damage, and progressive keratoconus are additional contraindications. 6)
Essential preoperative evaluations include corneal endothelial cell density measurement, anterior segment imaging (including anterior chamber depth), and corneal diameter measurement (horizontal diameter). 6)
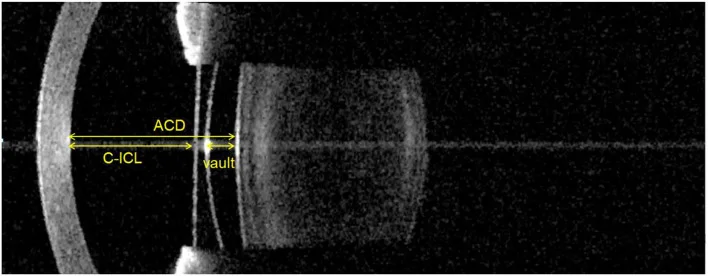
→ For details, see Phakic Posterior Chamber Lens (ICL).
This is an auxiliary IOL that is additionally placed over an existing monofocal IOL and fixed in the ciliary sulcus. Even in eyes that have previously received a monofocal IOL, it allows for presbyopia correction, astigmatism correction, and myopia correction as a retrofit. The distance from the in-the-bag IOL is relatively constant, and long-term stability is considered good.
→ For details, see Add-on Intraocular Lens.
| Type | Focal Range | Contrast Sensitivity | Glare/Halo | Astigmatism Correction | Insurance Coverage | Estimated Additional Cost (Both Eyes) |
|---|---|---|---|---|---|---|
| Monofocal | One point (distance or near) | ◎ | None | △ (toric separately) | ○ (selected medical care) | 0 to tens of thousands of yen |
| Bifocal (diffractive) | Distance + Near | ○ | ++ (moderate) | △〜○ | × | 300,000–500,000 yen |
| Trifocal | Distance + Intermediate + Near | ○ | ++ (Moderate) | △〜○ | × | 400,000–600,000 yen |
| EDOF | Distance to intermediate (continuous) | ◎〜○ | + (mild) | △〜○ | × | 350,000–550,000 yen |
| Toric (monofocal) | 1 point (distance priority) | ◎ | None | ◎ | × (selected medical treatment) | 100,000–150,000 yen |
| Phakic IOL (PIOL/ICL) | Distance (no corneal correction needed) | ◎ | None | △~○ with EVO+ | × | 500,000–700,000 yen |
| Add-on | Existing IOL + presbyopia correction | ○ | + | ○ | × | 300,000–450,000 yen |
Costs are approximate estimates for bilateral self-pay treatment. Varies greatly by facility and selected lens.
IOL selection directly affects postoperative vision, so careful preoperative confirmation of patient expectations is essential. The ESCRS cataract guidelines state that “evaluation of patient expectations and detailed informed consent are the most important prerequisites for appropriate IOL selection.” 4) Information to be gathered:
Conditions suitable for multifocal IOLs (trifocal/EDOF):
Meta-analyses have shown that the rate of IOL exchange within one year increases when spectacle independence is not achieved or optical side effects do not improve after multifocal IOL implantation. 3)
Toric IOLs are effective for correcting regular corneal astigmatism, but there are several prerequisites.
To account for the effect of posterior corneal astigmatism, it is recommended to use the Barrett Toric formula or the Haigis-T formula built into optical biometers. 7) Additionally, ocular cyclotorsion between sitting and supine positions averages 4–5°, so photo-based marking or image-guided systems (CALLISTO eye, VERION) for automatic axis alignment are useful for improving accuracy. In a prospective study comparing image-guided systems with manual marking, the image-guided group had significantly less postoperative residual astigmatism (0.33 D vs. 0.51 D, p = 0.003). 19)
For those who drive at night frequently or primarily perform precise visual tasks, a monofocal IOL may be more suitable. Conversely, for those who want to handle a wide range of distances—such as smartphones, computers, and reading—without glasses, a multifocal IOL may be a candidate. However, depending on the eye condition (macular disease, glaucoma, dry eye, etc.), multifocal IOLs may not be appropriate. Preoperative consultation with the attending physician is most important.
For precise IOL power calculation, simultaneous measurement of multiple parameters using an optical biometer is standard. Compared to the ultrasound A-scan method, optical biometry has smaller measurement errors. In Japan, it was covered by insurance as “optical axial length measurement” in 2010 and rapidly became widespread. The adoption of FD (Fourier domain) technology has improved the measurement success rate from about 90% to approximately 98%, and the standard deviation of measurements is now within 0.02 mm. 7)
| Device | Measurement Method | Main Features |
|---|---|---|
| IOLMaster 700 (Carl Zeiss) | SS-OCT (Swept Source) | Axial length, corneal refractive power, anterior chamber depth, lens thickness, corneal diameter, gold standard |
| ARGOS (Santen/Santek) | FD-OCT (Segmented method) | Measures refractive index of each tissue individually. Tends to measure axial length shorter than IOLMaster 700 |
| OA-2000 (Tomey) | FD-OCT | Displays B-scan image and A-scan waveform side by side |
| Ultrasound A-mode method | Ultrasound (1,550 m/s) | Applicable to all cases but has large error (considered the cause of most postoperative refractive errors)7) |
Optical biometry displays axial length 0.2–0.3 mm longer than ultrasound, so it is essential to use IOL constants (e.g., A-constant) specific to the measurement device. Since ARGOS uses a segmented approach, interchangeable use of IOL constants with IOLMaster 700 must be avoided. 7)
| Formula | Generation | Characteristics | Primary Use |
|---|---|---|---|
| SRK/T formula | 3rd generation | Used in over 90% of cases in Japan. Extensive clinical experience, A-constant | First-line for standard axial length (22–25 mm) |
| Haigis formula | 3rd generation | Uses anterior chamber depth as an independent variable. Three constants a0, a1, a2 (requires optimization with ≥200 eyes) | For shallow/deep anterior chambers |
| Barrett Universal II formula | 4th generation | Based on thick lens theory. Good accuracy for low-power IOLs. Free calculation available on APACRS website. | Long axial length, short axial length, low-power IOL |
| Hill-RBF | AI (machine learning) | Pattern recognition from big data. Not formula-based. | Long axial length, high myopia |
| Kane formula | AI (machine learning + theoretical formula) | Sex also used as a variable | Extreme high myopia (axial length ≥ 30 mm) |
In Japan, the third-generation SRK/T formula is widely used, but it is advisable to compare multiple calculation results depending on axial length and anterior segment anatomy (ocular biometry). Approximately 15% of patients seeking cataract surgery have eyes with irregular biometry in terms of axial length and corneal refractive power, so caution is needed when selecting the calculation formula. 7)
A multicenter study of 13,301 eyes showed that the Barrett Universal II, Olsen, and Haigis formulas were significantly more accurate than SRK/T, Holladay 1, and Hoffer Q (Holladay 2 had nearly equivalent accuracy), and fourth-generation and later formulas were superior for both long and short axial lengths. 15) Another meta-analysis focusing on short axial length (AL < 22 mm) also found that Barrett Universal II had significantly smaller MAE than Haigis and SRK/T (p < 0.05). 18)
In a study of 80 eyes with high myopia (axial length ≥ 30 mm), AI formulas (Kane, Hill-RBF) showed significantly smaller mean absolute error (MAE) compared to SRK/T (Kane: 0.51 D, Hill-RBF: 0.52 D, Barrett Universal II: 0.66 D, SRK/T: significant difference, p < 0.05). The rate of refractive error > 1.0 D was suppressed to 7.5% with Kane and Hill-RBF, whereas it reached 42.5% with SRK/T. 8) In eyes with axial length ≥ 32 mm, the Kane formula achieved the lowest MAE (0.44 D) and MedAE (0.40 D). 8) In another report by Rong 2019 targeting axial length ≥ 28 mm, the MedAE of Barrett Universal II was 0.37 D, significantly smaller than Haigis’s 0.46 D (p = 0.038). In eyes with axial length ≥ 30 mm, Barrett Universal II showed better accuracy than Haigis. 16)
| Case | Recommended Formula | Notes |
|---|---|---|
| Standard eye (AL 22–25 mm) | SRK/T, Haigis, Barrett Universal II | Compare multiple formulas |
| Long axial length (AL > 26 mm) | Barrett Universal II, Hill-RBF, Kane | Note accuracy differences with low-power IOLs15,16) |
| Short axial length (AL < 22 mm) | Barrett Universal II, Haigis, Holladay 2 | Risk of high refractive error. Large ELP prediction error18) |
| After corneal refractive surgery (post-LASIK) | Haigis-L, Shammas No-history, Barrett True-K, OKULIX | Compare multiple formulas using ASCRS online calculator9,20) |
| Ciliary sulcus (extracapsular) fixation | Calculate with SRK/T etc., then adjust by −1.0 D | For long axial length, low power requires less adjustment; for short axial length, high power requires −2.0 D |
| Intrascleral fixation (forceps method) | Same correction as extracapsular fixation | Varies by surgical technique |
| Keratoconus / post-keratoplasty | Dedicated formula (Seitz-Langenbucher etc.) or expert judgment | Standard formulas have large errors |
After LASIK, the cornea is locally flattened, so auto-keratometry tends to overestimate central corneal power, leading to postoperative hyperopic shift. Even without preoperative LASIK data, formulas such as Haigis-L, Shammas No-history, and Barrett True-K are available on optical biometers, and comparing multiple formulas using the free online calculator from the American Society of Cataract and Refractive Surgery (ASCRS) is a useful strategy. 7,9) In a study of 110 post-LASIK eyes, the accuracy of Barrett True-K alone was comparable to the average of multiple formulas (MAE 0.41 D vs. 0.42 D, p = 0.81), indicating its reliability even when used alone. 20)
Before surgery, set the target refraction based on the patient’s lifestyle, work, and hobbies (predicted spherical equivalent).
| Lifestyle | Recommended Target Refraction |
|---|---|
| Distance vision priority (driving, sports) | Emmetropia (0.00 D) to −0.25 D |
| Balanced type | −0.25 to −0.50 D (slight myopic shift) |
| Near vision priority (reading, detailed work) | −1.50 to −2.00 D (monovision candidate) |
| Monovision (non-dominant eye) | −1.50 to −2.00 D |
| Multifocal IOL (most products) | Emmetropia (must be within ±0.25 D) |
For toric IOL calculation, using formulas built into optical biometers (such as the Haigis-T formula or Barrett Toric formula) eliminates the need for manual input of measurement values and reduces the risk of entry errors. The basic principle is to consistently use optical axial length, corneal power, and IOL constants. 7)
Mild refractive errors (within ±0.5 D) can be corrected with glasses. For larger errors (≥±1.5 D), consider adding an add-on IOL or performing IOL exchange. With multifocal IOLs, even errors less than 0.5 D can significantly reduce patient satisfaction, so precise preoperative measurements and careful selection of calculation formulas are especially important.
After IOL implantation, the natural lens accommodation (change in thickness due to ciliary muscle contraction) is lost. However, a phenomenon called “pseudoaccommodation” may occur clinically, allowing slight near vision. Pseudoaccommodation is caused by corneal multifocality, astigmatism, the pinhole effect of the pupil, and spherical aberration of the IOL. Even with monofocal IOLs, pseudoaccommodation of about 0.5 to 1.0 D may be observed.
Monofocal IOLs provide contrast sensitivity equal to or better than that of a young normal lens. On the other hand, multifocal IOLs reduce contrast sensitivity in the high spatial frequency range due to light dispersion. In eyes with mild cataract or IOL implantation, contrast sensitivity may be reduced even if visual acuity is unchanged. A meta-analysis of trifocal IOLs and EDOF IOLs (22 studies) found no significant difference in contrast sensitivity between the two groups. 5) In multifocal IOLs in general (Cochrane meta-analysis including 8 RCTs), contrast sensitivity is slightly reduced compared to monofocal IOLs, but the clinical significance is unclear. 3)
If posterior capsule opacification (PCO) develops within 5 years after surgery, contrast sensitivity may be significantly reduced. Hydrophobic acrylic IOLs have been shown to have a lower incidence of PCO compared to hydrophilic acrylic IOLs, and a square-edge optic design is also effective in preventing it. 10)
Halo refers to a ring of light seen around a light source, and glare refers to dazzling or clouding of vision caused by a light source. With multifocal IOLs, halos (RR 3.58, 95% CI 1.99–6.46) and glare are significantly increased compared to monofocal IOLs. 3) EDOF IOLs have fewer optical side effects than traditional bifocal or trifocal IOLs, but they do not completely disappear. 5)
Management strategies:
Immediately after insertion of a non-tinted IOL, patients may report a bluish tint to vision (cyanopsia) because the IOL transmits more short-wavelength light than the natural human lens. Tinted (yellow filter) IOLs reduce this phenomenon. Neural adaptation occurs within days to weeks, and most patients no longer notice it.
Glistenings are punctate reflections seen in the optic of hydrophobic acrylic IOLs. Those occurring in the deep layers are called glistenings, while those on the surface are called subsurface nano glistenings (SSNG). Both are caused by aqueous humor entering small gaps within the optic material and do not represent material degradation. They usually do not affect visual function, but decreased visual function has been reported in patients with reduced retinal function. Currently available IOLs have improved manufacturing processes that reduce these phenomena. In hydrophilic acrylic IOLs, calcium phosphate may deposit on the surface with long-term use, causing severe opacification (calcium deposition). 10)
Modern cataract surgery is highly safe, but posterior capsule opacification (treatable with laser) occurs in about 20–40% of patients within 5 years. Serious complications (endophthalmitis, expulsive hemorrhage) are rare at 0.006–0.04%. IOL dislocation tends to increase over time due to aging and weakening of the zonules, especially in patients with advanced age, high myopia, or pseudoexfoliation syndrome.
| Complication | Incidence (approximate) | Overview | Management |
|---|---|---|---|
| Posterior capsule opacification (PCO) | 20–40% at 5 years postoperatively | Lens epithelial cells proliferate on the posterior capsule behind the IOL, reducing visual function | Nd:YAG laser posterior capsulotomy |
| IOL decentration or dislocation | Several percent per year in eyes with weakened zonules | Zonular rupture due to aging, trauma, or pseudoexfoliation syndrome causes the IOL to fall into the vitreous cavity | Intrascleral fixation, suturing, or IOL exchange11) |
| Pupillary capture (iris capture) | A few percent after intrascleral fixation, etc. | Condition where the IOL optic dislocates anterior to the pupil. | Mydriasis, postural change, exchange to IOL with larger optic diameter12) |
| IOL opacification | Hydrophobic glistening: a few percent; hydrophilic calcium deposition: several years later | Visual function decline due to material degradation | IOL exchange10) |
| Glaucoma after PIOL | Intraocular pressure elevation after ICL surgery 1–5% | Angle closure and pupillary block causing elevated intraocular pressure | Preoperative anterior chamber depth assessment and postoperative regular management13) |
IOL displacement and dislocation
Diagnosis and surgical indications for IOL drop due to Zinn zonule rupture. Selection of intrascleral fixation (flange technique, forceps technique).
IOL Opacification
Mechanisms and management of glistening, SSNG, calcium deposition, and posterior capsule opacification. Information influencing IOL material selection.
Biometry (IOL Power Calculation)
Simultaneous measurement of axial length, corneal refractive power, and anterior chamber depth using an optical biometer. Biometry for IOL Power Calculation
Intrascleral fixation
Flange technique and forceps technique for implanting IOL haptics into the sclera without sutures. Avoids risks of endophthalmitis and suture degradation. Intrascleral fixation IOL
Sanders DR, Retzlaff J, Kraff MC. Comparison of the SRK II formula and other second generation formulas. J Cataract Refract Surg. 1988;14(2):136-141.1)
European Society of Cataract and Refractive Surgeons. ESCRS recommendations for cataract surgery. Draft version September 2024. Chapter 4.6: patient selection for correcting presbyopia. https://www.escrs.org/escrs-guideline-for-cataract-surgery/2)
de Silva SR, Evans JR, Kirthi V, Ziaei M, Leyland M. Multifocal versus monofocal intraocular lenses after cataract extraction. Cochrane Database Syst Rev. 2016;12(12):CD003169. doi:10.1002/14651858.CD003169.pub4. PMID:27943250; PMCID:PMC6463930.3)
European Society of Cataract and Refractive Surgeons. ESCRS recommendations for cataract surgery. Draft version September 2024. Chapters 4.6-4.7 and 6.1-6.3. https://www.escrs.org/escrs-guideline-for-cataract-surgery/4)
Karam M, Alkhowaiter N, et al. Extended Depth of Focus Versus Trifocal for Intraocular Lens Implantation: An Updated Systematic Review and Meta-Analysis. J Refract Surg. 2022. 22 studies, 2200 eyes.5)
日本眼科学会屈折矯正委員会. 屈折矯正手術のガイドライン(第 8 版). 日眼会誌. 2024;128(2):135-142.6)
Werner L. Intraocular lenses: overview of designs, materials, and pathophysiologic features. Ophthalmology. 2021;128(11):e74-e93.7)
Suzuki Y, Kamoi K, Uramoto K, Ohno-Matsui K. Artificial intelligence driven intraocular lens power calculation in extreme axial myopia. Sci Rep. 2025. 80 eyes, axial length ≥30.0 mm, Institute of Science Tokyo.8)
American Academy of Ophthalmology. Cataract in the Adult Eye Preferred Practice Pattern®. AAO; 2021.9)
Grzybowski A, Markeviciute A, Zemaitiene R. A narrative review of intraocular lens opacifications: update 2020. Ann Transl Med. 2020;8(22):1547. doi:10.21037/atm-20-4207. PMID:33313292; PMCID:PMC7729367.10)
Tripathi M, Rao S, Sinha R. Scleral-fixated IOLs - A comprehensive review of current practices and emerging trends. Indian J Ophthalmol. 2025;73(7):933-945. doi:10.4103/IJO.IJO_2812_24. PMID:40586185; PMCID:PMC12356440.11)
Choi SR, Jeon JH, Kang JW, Heo JW. Risk factors for and management of pupillary intraocular lens capture after intraocular lens transscleral fixation. J Cataract Refract Surg. 2017;43(12):1557-1562. PMID: 29335100. doi:10.1016/j.jcrs.2017.08.021.12)
日本眼科学会屈折矯正委員会. 屈折矯正手術のガイドライン(第 8 版). 有水晶体眼内レンズ手術の禁忌・慎重事項. 日眼会誌. 2024;128(2):135-142.13)
Kessel L, Andresen J, Tendal B, Erngaard D, Flesner P, Hjortdal J. Toric intraocular lenses in the correction of astigmatism during cataract surgery: a systematic review and meta-analysis. Ophthalmology. 2016;123(2):275-286. doi:10.1016/j.ophtha.2015.10.002. PMID:26601819.14)
Melles RB, Holladay JT, Chang WJ. Accuracy of intraocular lens calculation formulas. Ophthalmology. 2018;125(2):169-178. doi:10.1016/j.ophtha.2017.08.027. PMID:28951074.15)
Rong X, He W, Zhu Q, et al. Intraocular lens power calculation in eyes with extreme myopia: comparison of Barrett Universal II, Haigis, and Olsen formulas. J Cataract Refract Surg. 2019;45(6):732-737.16)
McCabe C, Berdahl J, Reiser H, et al. Clinical outcomes in a U.S. registration study of a new EDOF intraocular lens with a nondiffractive design. J Cataract Refract Surg. 2022;48(11):1297-1304. PMID: 35616507. PMCID: PMC9622364. doi:10.1097/j.jcrs.0000000000000978.17)
Wang Q, Jiang W, Lin T, Wu X, Lin H, Chen W. Meta-analysis of accuracy of intraocular lens power calculation formulas in short eyes. Clin Exp Ophthalmol. 2018;46(4):356-363. doi:10.1111/ceo.13058. PMID:28887901.18)
Webers VSC, Bauer NJC, Visser N, et al. Image-guided system versus manual marking for toric intraocular lens alignment in cataract surgery. J Cataract Refract Surg. 2017;43(6):781-788. doi:10.1016/j.jcrs.2017.03.041. PMID:28732612.19)
Ferguson TJ, Downes RA, Randleman JB. IOL power calculations after LASIK or PRK: Barrett True-K biometer-only calculation strategy yields equivalent outcomes as a multiple formula approach. J Cataract Refract Surg. 2022;48(7):784-789. doi:10.1097/j.jcrs.0000000000000883. PMID:35067661.20)