SS-OCT
Wavelength: 1310 nm (long wavelength)
Penetration depth: High (images the entire anterior segment in one frame)
Resolution: Lower than SD-OCT but sufficient for practical use
Representative model: CASIA2 (Tomey)
Anterior segment OCT (AS-OCT: Anterior Segment Optical Coherence Tomography) is a non-invasive imaging device that uses near-infrared light interference to obtain cross-sectional images of the cornea, anterior chamber, lens, and angle. It enables observation and quantitative evaluation of areas that cannot be examined with a slit lamp microscope, and is widely used in screening for angle closure, corneal diseases, before and after refractive surgery, and angle evaluation in glaucoma.
AS-OCT imaging was first reported by Izatt et al. in 1994. Initially, the same 830 nm wavelength used for retinal OCT was employed, but it had poor penetration through scattering tissues such as the sclera, making it unsuitable for angle imaging. Subsequently, devices using a longer wavelength of 1310 nm were developed, significantly improving scleral penetration and imaging speed.
Currently, Fourier-domain OCT (FD-OCT) is mainstream. Compared to time-domain OCT (TD-OCT), it offers superior measurement speed, resolution, and three-dimensional analysis capabilities. FD-OCT includes two types: swept-source OCT (SS-OCT) and spectral-domain OCT (SD-OCT).
SS-OCT
Wavelength: 1310 nm (long wavelength)
Penetration depth: High (images the entire anterior segment in one frame)
Resolution: Lower than SD-OCT but sufficient for practical use
Representative model: CASIA2 (Tomey)
SD-OCT
Wavelength: 840 nm (short wavelength)
Penetration depth: Shallow (difficult to image the entire anterior segment)
Resolution: Higher resolution than SS-OCT
Use: Suitable for detailed observation of the cornea and conjunctiva
AS-OCT is a diagnostic device that allows non-contact observation of the angle. Its resolution is superior to ultrasound biomicroscopy (UBM), but it cannot visualize the ciliary body 3). Its usefulness as an auxiliary diagnostic tool in glaucoma care is widely recognized 3).
Fundus OCT is a device that captures tomographic images of the retina, using a light source with a wavelength of 840–870 nm. AS-OCT is specialized for observing the anterior segment (cornea, angle, iris, etc.), and the SS-OCT method uses a longer wavelength of 1310 nm to enhance penetration into deep tissues. The observation target and wavelength used differ.
There are two types of anterior segment tomographic imaging: AS-OCT and ultrasound biomicroscopy (UBM). Both have commonalities and clear differences.
| Item | Anterior Segment OCT (AS-OCT) | Ultrasound Biomicroscopy (UBM) |
|---|---|---|
| Principle | Light (wavelength 0.7–1.3 μm) | Ultrasound (30–50 MHz) |
| Contact | Non-contact | Contact (requires water immersion) |
| Position | Sitting (some supine possible) | Supine |
| Resolution | 15 μm | 50 μm |
| Maximum scan range | 16 × 6 mm | 5 × 5 mm |
| Ciliary body observation | Unclear | Possible |
| Posterior iris surface | Unclear | Possible |
| Corneal surface / tear meniscus | Useful | Unsuitable |
| Image analysis software | Comprehensive | Limited |
| Immediately after surgery | Possible (no infection risk) | Difficult |
AS-OCT is the first choice in daily practice due to its advantages of being non-contact, fast, and high-resolution. On the other hand, UBM excels in observing the ciliary body, zonules, and posterior iris surface, which are difficult to visualize with AS-OCT. UBM is particularly useful when observation of the ciliary body is needed, such as in the diagnosis of malignant glaucoma or detailed evaluation of plateau iris.
AS-OCT is performed according to the following procedure.
CASIA2 (equipped with SS-OCT) performs automatic 360° angle analysis to calculate AOD500 around the entire circumference and quantifies narrow-angle risk with a narrow angle index. Combined with gonioscopy results, it can be used for staff education and patient explanations.
AS-OCT cannot detect neovascularization or pigmentation of the angle. Secondary causes such as peripheral anterior synechiae (PAS), pigmentation, and trabecular meshwork dysfunction may be missed if evaluated only with AS-OCT6).
No, it cannot. AS-OCT has the advantage of being non-contact and allowing imaging in dark conditions, but findings such as peripheral anterior synechiae, pigment deposition, and neovascularization may be difficult to detect with AS-OCT6). Gonioscopy should be performed in all patients with suspected glaucoma6).
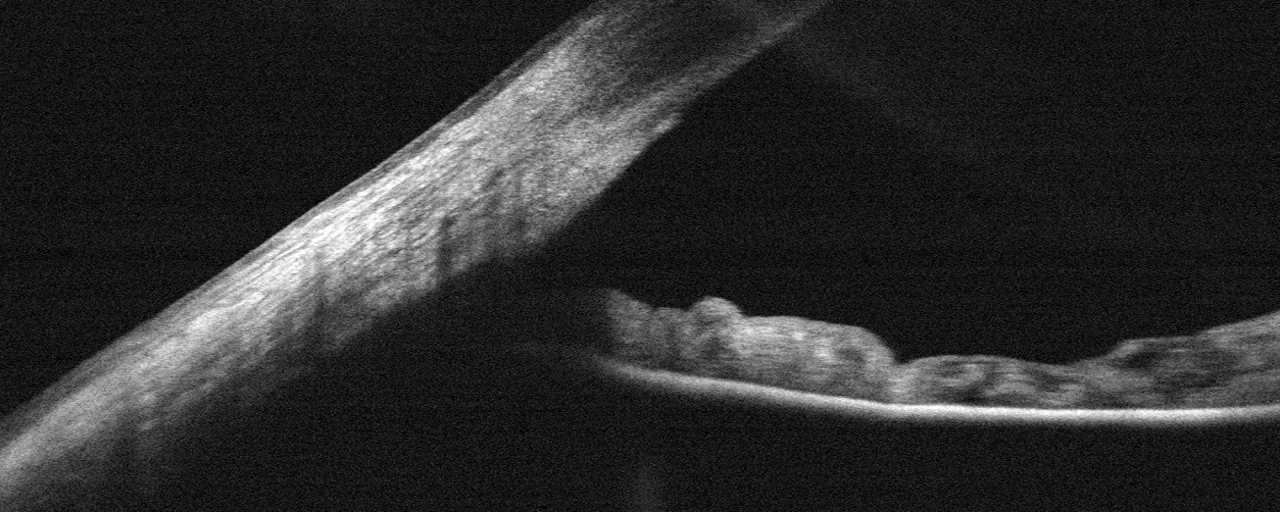
The most important landmark when interpreting AS-OCT images is the scleral spur. The scleral spur is visible as a structure where the inner surface of the sclera and the curvature of the cornea meet, with the sclera protruding inward. By evaluating the apposition between the iris and the inner wall of the corneosclera, angle closure can be detected.
However, it has been reported that the scleral spur is not visible in approximately 25% of cases when using scan protocols without image averaging.
The main parameters used for quantitative measurement of the anterior chamber angle are shown below.
| Parameter | Abbreviation | Definition |
|---|---|---|
| Angle Opening Distance | AOD | Distance between the iris and a point 500/750 μm anterior to the scleral spur |
| Angle recess area | ARA | Area enclosed by AOD, iris, and corneoscleral wall |
| Trabecular-iris space area | TISA | Trapezoidal area from scleral spur to AOD line |
Other measurable parameters include iris thickness, anterior chamber width, and lens vault.
AS-OCT is useful not only for angle assessment but also for detailed evaluation of corneal cross-sections.
Angle imaging cannot replace gonioscopy 6). Gonioscopy should be performed in all patients with suspected glaucoma 6).
In glaucoma clinical practice, AS-OCT is useful as an adjunct to gonioscopy or as an alternative when gonioscopy is difficult due to corneal disease or poor patient cooperation. Because it is non-contact and can be performed in dark conditions, angle assessment under physiological mydriasis is possible.
Based on iris morphology and the position of the lens relative to anterior segment structures, mechanisms of angle closure such as pupillary block and lens-induced angle closure can be differentiated 4). As a diagnostic aid for angle closure (PAC/PACS), it helps in decisions regarding laser iridotomy (LPI) and cataract surgery 4).
It is also useful as a patient education tool when recommending laser iridotomy 5). It has become indispensable for observing morphological changes of the iris such as shallow anterior chamber, narrow angle, and plateau iris.
AS-OCT is also applied in preoperative and postoperative evaluation of glaucoma surgery. It is used to assess the morphology of filtering blebs after trabeculectomy and to confirm the position of intraocular drainage devices.
Tanito et al. (2024) clearly visualized the status of the stent, which was difficult to assess with conventional 2D cross-sectional images, using raster scanning and 3D AS-OCT imaging in a case 2 years after PreserFlo MicroShunt (PFM) implantation. In the right eye, C-shaped deformation was confirmed, suggesting possible fin extrusion from the scleral pocket 1). Adding 3D images to 2D images significantly improved the accuracy of stent evaluation 1).
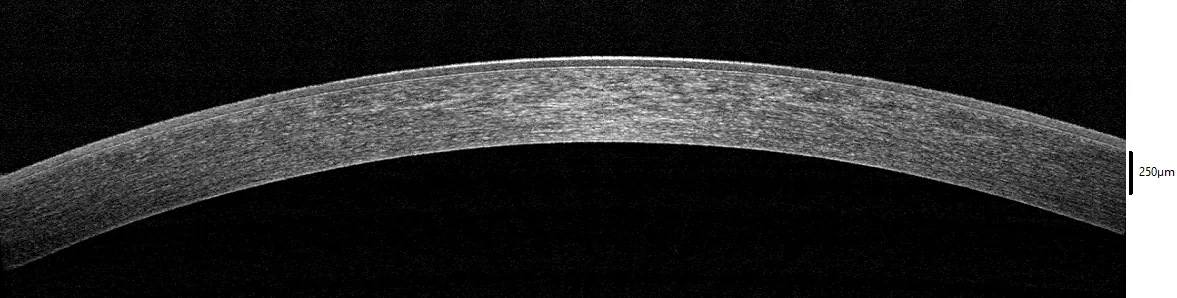
AS-OCT evaluates the depth of corneal opacities in cross-section and helps in selecting the surgical technique for corneal transplantation.
Before cataract surgery, AS-OCT is used for quantitative evaluation of the anterior segment.
AS-OCT is a non-contact examination; no instrument touches the eye. There is no pain or discomfort. No anesthetic eye drops are needed, and the examination takes only a few minutes.
AS-OCT utilizes the principle of the Michelson interferometer. Light from the source is split into a “reference arm” and a “sample arm” (irradiation to the eye), and the reflected light from each is interfered to obtain the reflection intensity from each depth in the tissue as an A-scan signal. The A-scan signal is converted into a depth-wise luminance distribution by Fourier transform, and a tomographic image is generated by two-dimensional scanning.
FD-OCT (Fourier-domain OCT) has two implementation methods.
SS-OCT at 1310 nm has a penetration depth that reaches the posterior lens surface and ciliary body, and has become the de facto standard for AS-OCT applications.
Optical coherence tomography angiography (OCTA) is a rapidly developing technology. It is considered less susceptible to floor effects than retinal nerve fiber layer measurements and may be more advantageous than OCT for progression assessment in advanced glaucoma, but standardized clinical applications have not yet been established 3).
Huang et al. (2024) conducted a bibliometric analysis of AS-OCT applications in glaucoma over 20 years (2004–2023), analyzing 931 reports. The United States had the most publications (288), followed by China (231) and Singapore (124). Aung Tin had the most publications (80) and citations (3595) 2).
The number of papers increased sharply after 2012, and since 2015, more than 60 papers have been published annually 2). Since 2018, advances in artificial intelligence (AI) have led to a notable shift from manual measurement to automated detection and recognition 2).
A recent research frontier is the automatic detection of angle closure using deep learning 2). Conventional AS-OCT image evaluation relies on manual measurement of various parameters, which is time-consuming, subjective, and has low reproducibility.
Deep learning algorithms learn directly from image data and demonstrate the ability to classify open, narrow, and closed angles with high accuracy. A 3D deep learning-based digital gonioscopy system (DGS) showed diagnostic accuracy comparable to ophthalmologists in detecting narrow iridocorneal angles and peripheral anterior synechiae 2).
FD-mode AS-OCT operating at a wavelength of 1310 nm is enabling rapid three-dimensional cube scans of the anterior segment. This is expected to allow the following evaluations:
3D AS-OCT has also demonstrated utility in postoperative evaluation of glaucoma surgical devices, clearly visualizing the overall deformation and displacement of stents that were difficult to assess with 2D images1).
It is still at the research stage. Deep learning algorithms for automatic detection of angle closure have shown high accuracy2), but have not yet been widely implemented in clinical practice. Challenges remain, such as insufficient data and lack of unified diagnostic criteria.