Pupillary Block
Iris morphology: Peripheral iris bulging forward (iris bombé)
Anterior chamber depth: Particularly shallow in the periphery
PI: Occluded or not performed
Miotics: Effective

Malignant glaucoma is a condition of elevated intraocular pressure with a markedly shallow anterior chamber, typically occurring after filtration surgery in eyes with angle-closure glaucoma. It is thought to result from angle closure caused by anterior displacement of the vitreous due to forward rotation of the ciliary body and abnormal inflow of aqueous humor into the vitreous cavity. It is also called aqueous misdirection syndrome, ciliary block glaucoma, or direct lens block glaucoma, and multiple terms are used interchangeably. The ICD-10 code is H40.8.
First reported by Von Graefe in 1869 as a refractory shallow anterior chamber after filtration surgery, this is a historic disease concept 3). Subsequently, the pathophysiological concept that aqueous humor is misdirected posteriorly (into the vitreous cavity) rather than following the normal forward pathway was established, and the term “aqueous misdirection” became widely used. It is most commonly encountered in eyes with angle-closure glaucoma after filtration surgery, but it is clinically important that it can occur after any intraocular surgery, including cataract surgery.
The Glaucoma Clinical Practice Guidelines (5th edition) classify the mechanisms of intraocular pressure elevation in secondary angle-closure glaucoma into the following four categories 6).
Malignant glaucoma is classified under the third category, “anterior movement of tissues posterior to the lens.” It corresponds to the “posterior pushing mechanism” in the EGS 5th edition 5).
The incidence after filtration surgery is reported to be 0.6–4% 5). It is more common in women and usually occurs unilaterally. It is most frequent after filtration surgery in eyes with primary angle-closure glaucoma, but it can occur after any intraocular surgery, including cataract surgery 5). Risk factors include short axial length (<21 mm), high hyperopia (≥+6 D), and a history of primary angle-closure glaucoma 5). Rare spontaneous cases without prior surgery have also been reported. Trabeculectomy for angle-closure glaucoma is known to have a relatively high risk of causing malignant glaucoma postoperatively, along with shallow anterior chamber and choroidal detachment 6).
In pupillary block, obstruction of aqueous flow at the pupillary margin causes the peripheral iris to bulge forward (iris bombé). In contrast, in aqueous misdirection, aqueous humor accumulates in the vitreous cavity from behind the lens, resulting in a uniformly shallow anterior chamber. Miotics are effective for pupillary block, but they are contraindicated in aqueous misdirection.
In acute onset, the following symptoms occur.
Symptoms are fluctuant and may become apparent weeks to years after cataract surgery 3). In early stages, intraocular pressure may remain within normal range, leading to delayed diagnosis 5). When intraocular pressure rises rapidly, the clinical picture resembles acute glaucoma attack, with rapid progression of vision loss.
Diagnosis is made by a combination of the following findings. In addition to elevated intraocular pressure, the pattern of anterior chamber depth change is most important for differential diagnosis.
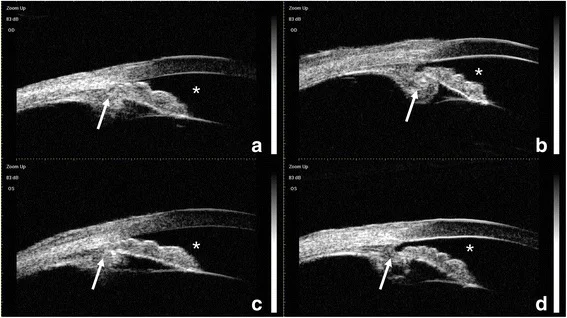
Anterior segment optical coherence tomography (AS-OCT) has been reported to show plateau iris-like findings (flat central iris but occluded angle) associated with anterior displacement of the CTR (capsular tension ring)-IOL complex 2). Ultrasound biomicroscopy (UBM) can directly confirm anterior displacement of the lens capsule-IOL complex and anterior rotation or positional abnormalities of the ciliary body 4). UBM has a high resolution of 20–60 μm and can provide detailed imaging of anterior segment structures such as the iris and ciliary body, so it should be actively performed in cases suspected of malignant glaucoma.
UBM and anterior segment OCT are useful for elucidating the pathophysiology. In particular, while confirming the presence or absence of surgical history, it is important to evaluate whether there is any positional abnormality of the ciliary body.
The most common trigger is intraocular surgery 5).
Smaller eyes have a higher risk 5).
If it occurs in one eye, the risk of developing it in the fellow eye also increases 3). Since the risk is elevated regardless of a history of glaucoma, regular follow-up of the fellow eye is important.
The main risk factors are summarized below.
| Risk factor | Specific example |
|---|---|
| Ocular morphology | Short axial length (<21 mm), high hyperopia (>+6 D), shallow anterior chamber |
| Surgery | Filtering surgery, cataract surgery, CTR placement |
| Medication | Topiramate, SSRIs |
Topiramate increases vascular permeability of the ciliary body and choroid, causing ciliary body swelling and forward rotation 1). In high-risk eyes such as those with microphthalmia, it can trigger aqueous misdirection. SSRIs may also induce angle closure in eyes with narrow angles through a mydriatic effect.
Malignant glaucoma is a diagnosis of exclusion, established only after systematically ruling out pupillary block, choroidal hemorrhage/detachment, suprachoroidal hemorrhage, and other mechanisms of angle closure 4). It is not a disease that can be confirmed by specific laboratory values or imaging findings alone; a comprehensive assessment combining clinical course and multiple examination findings is required.
Suspect this condition when extremely shallow anterior chamber and high intraocular pressure occur early after surgery for angle-closure glaucoma. Hemorrhagic choroidal detachment shows similar slit-lamp findings and can also cause high intraocular pressure, but can be differentiated by fundus examination. In idiopathic cases, diagnosis is more difficult due to the absence of surgery history, and confirmation of abnormal ciliary body position by UBM is key to diagnosis.
The differentiating points between pupillary block and aqueous misdirection are summarized below.
Pupillary Block
Iris morphology: Peripheral iris bulging forward (iris bombé)
Anterior chamber depth: Particularly shallow in the periphery
PI: Occluded or not performed
Miotics: Effective
Aqueous misdirection
Iris morphology: Iris is displaced forward overall
Anterior chamber depth: Uniformly shallow to absent5)
PI: Patent
Miotics: Contraindicated (worsens condition)
Other differential diagnoses include the following. All can cause shallow anterior chamber and high intraocular pressure, but the treatment strategy differs because the pathogenesis is different.
A stepwise approach of medication → laser therapy → surgery is fundamental. First, medication is used to attempt to relieve ciliary block; if ineffective, laser therapy is performed; if still ineffective, surgery is performed. The ultimate goal of treatment is to create a state where no block occurs between the posterior and anterior chambers (unicameral eye) and to prevent recurrence.
Combination therapy with a parasympatholytic agent (mydriatic / cycloplegic), an aqueous humor suppressant (eye drops and oral), and a hyperosmotic agent (intravenous) is recommended 5).
| Drug | Dosage | Mechanism of Action |
|---|---|---|
| Atropine ophthalmic solution 1% | 1–3 times daily | Cycloplegia / mydriasis |
| Timolol ophthalmic solution 0.5% | Twice daily | Aqueous humor suppression (beta-blockade) |
| Diamox tablets 250 mg | 2 tablets, divided into 2 doses after meals | Aqueous humor suppression (oral CAI) |
| Mannitol infusion | Intravenous administration | Reduction of vitreous volume |
Atropine increases tension of the zonules, moving the lens posteriorly, and triggers aqueous humor that has moved into the vitreous cavity to return forward. Cyclopentolate can also be used as an alternative to atropine 5).
Eye drops that suppress aqueous humor production (beta-blockers, carbonic anhydrase inhibitors) are used in combination with intravenous hyperosmotic agents 6). Hyperosmotic agents reduce vitreous volume, but their therapeutic effect is temporary, so they are positioned as preoperative treatment 6).
The response rate to pharmacotherapy is about 50% (within 5 days), but the recurrence rate with medication alone is high 5). Even if the anterior chamber deepens and intraocular pressure normalizes with pharmacotherapy, recurrence often occurs when atropine eye drops are discontinued, so long-term maintenance therapy with atropine eye drops may be necessary. Pharmacotherapy is only an initial measure; definitive treatment often requires laser or surgical intervention.
In pseudophakic eyes, Nd:YAG laser posterior capsulotomy is performed, and then the focus is shifted behind the posterior capsule, delivering 10 to 20 shots at 3-4 mJ to disrupt the anterior vitreous membrane. By creating a passage for aqueous humor between the vitreous cavity and the anterior chamber, the block is relieved. If successful, improvement in anterior chamber depth and reduction of intraocular pressure are obtained immediately to the next day.
In pseudophakic or aphakic eyes, YAG laser or surgical anterior vitreous membrane incision and capsulotomy are selected 6) (evidence level 2C).
Diode laser cyclophotocoagulation may be considered at any stage of treatment 5). It causes coagulation necrosis and atrophy of the ciliary processes, potentially leading to disruption of the ciliary-vitreous interface and posterior rotation of the ciliary body 3). However, the irradiation conditions required to disrupt the ciliary-vitreous interface have not been established.
If pharmacotherapy and laser treatment are insufficient, or in recurrent cases, surgery is performed. It is important to resolve the condition by vitrectomy with anterior vitreous membrane incision 6) (evidence level 2C).
Pars plana vitrectomy (PPV) is performed 5). In some cases, lens extraction is also performed 6) (2C). By combining lens extraction, a unicameral eye is created, completely relieving the block between the posterior and anterior chambers to prevent recurrence.
Zonulo-hyaloido-vitrectomy (irido-zonulo-anterior hyaloidectomy) is effective 5). An approach is made from the peripheral iridectomy site to the anterior chamber, and the zonules around the lens capsule and the anterior hyaloid membrane are excised. This procedure creates a permanent communication between the vitreous cavity and the anterior chamber.
In vitrectomy with anterior hyaloidotomy, some cases require iridectomy to treat the most peripheral anterior hyaloid membrane 6) (2C). If the anterior hyaloid membrane and zonules are not sufficiently treated, a block remains between the vitreous cavity and the anterior chamber, which may lead to recurrence. In phakic eyes, the lens is also removed simultaneously to relieve contact between the ciliary processes and the lens equator, creating a unicameral eye.
There is a report that a combination of CTR removal, anterior vitrectomy, and IOL scleral fixation was effective 2).
Aqueous misdirection is angle closure due to a posterior pushing mechanism of the lens. Miotics contract the ciliary muscle, promoting forward protrusion of the ciliary body and worsening ciliary block, thus they are contraindicated 6). In contrast, mydriatic-cycloplegic agents such as atropine relax the ciliary muscle, increase tension of the zonules, and move the lens posteriorly, which is effective in relieving the block.
Unilateral onset significantly increases the risk in the contralateral eye 3). The risk is elevated regardless of a history of glaucoma, so regular evaluation of intraocular pressure and anterior chamber depth in the contralateral eye is important.
The exact mechanism of malignant glaucoma has not been fully elucidated, but the following two hypotheses have been proposed 5).
Choroidal expansion theory
Main mechanism: Increased choroidal volume creates resistance to fluid movement from posterior to anterior 5)
Result: Aqueous humor accumulates in the vitreous cavity, pushing the iris-lens diaphragm forward
Ciliary block theory
Main mechanism: Abnormal anatomical contact between the ciliary processes and the equator of the lens (or IOL) or the anterior hyaloid membrane
Result: A “malignant cycle” is established, acting as a one-way valve that blocks the forward outflow of aqueous humor
The progression of the condition can be understood through the following cascade. In a normal eye, aqueous humor produced by the ciliary body flows from the posterior chamber through the pupil into the anterior chamber and exits via the trabecular meshwork at the angle. In malignant glaucoma, this pathway is disrupted.
Atropine eye drops relax the ciliary muscle, increasing tension on the zonules and moving the lens posteriorly. This may allow aqueous humor that has moved into the vitreous cavity to return forward.
In contrast, miotic agents such as pilocarpine contract the ciliary muscle. This contraction relaxes the zonules, pushing the lens forward and further promoting anterior rotation of the ciliary body, thereby worsening ciliary block 6). Therefore, miotics are contraindicated in malignant glaucoma.
In pseudophakic eyes, the horizontal diameter of the IOL-capsule complex is larger than that of the natural lens, making it more likely to contact the ciliary processes 4). Some of the aqueous humor produced by the ciliary processes is secreted posteriorly (into the vitreous cavity). If the anterior hyaloid membrane is intact, forward fluid outflow is impeded, creating a pressure gradient and shallowing the anterior chamber.
It has been reported that even in eyes with a history of vitrectomy, the condition can develop if the anterior hyaloid membrane remains 4). In a recurrent case with a myopic shift of 3 D or more, a combination of goniosynechialysis, iridectomy, and zonulo-hyaloidectomy restored refraction and intraocular pressure 4). Therefore, during vitrectomy, thorough removal of the anterior hyaloid membrane, including the most peripheral areas, is essential to prevent recurrence.
Topiramate increases the permeability of ciliochoroidal vessels, causing swelling of the ciliary body and choroid and anterior rotation of the ciliary body 1). In microphthalmia, impaired vortex vein drainage due to scleral thickening further increases the risk of uveal effusion 1).
Placement of a CTR expands and thickens the capsular bag, and the CTR-IOL complex contacts the ciliary body and posterior iris circumferentially 2). This compresses the ciliary sulcus, increasing resistance to the posterior-to-anterior flow of aqueous humor. Because the large diameter of the CTR-IOL complex partially limits anterior displacement, it may present with plateau iris-like angle closure while maintaining anterior chamber depth 2). This atypical finding differs from the classic appearance of malignant glaucoma with uniformly shallow anterior chamber, so detailed evaluation of angle morphology using AS-OCT is key to diagnosis.
Chean et al. (2021) reported a case of aqueous misdirection that developed with myopic surprise as the only initial sign after cataract surgery 3). Intraocular pressure remained within normal range for about three years postoperatively, leading to delayed diagnosis. Even if anterior chamber depth appears normal on biometry, measurement errors can occur in short axial length eyes, so aqueous misdirection should be included in the differential diagnosis when myopic shift is observed.
Stephenson et al. (2023) reported a case of aqueous misdirection with a myopic shift of 3 D or more in an eye with a history of vitrectomy, cataract surgery, and artificial iris implantation 4). They showed that even after vitrectomy, if the anterior hyaloid membrane remains, it can still occur, and refraction and intraocular pressure recovered after goniosynechialysis + iridectomy + zonulo-hyaloidectomy.
Goto et al. (2024) first characterized secondary angle closure due to anterior displacement of the CTR-IOL complex using AS-OCT 2). They reported an atypical plateau iris-like appearance with preserved anterior chamber depth, demonstrating the usefulness of AS-OCT in detecting secondary angle closure after cataract surgery with CTR implantation.
A stepwise approach of medication → Nd:YAG laser → cyclodiode → surgery has been proposed 3). Cyclodiode causes coagulation necrosis and atrophy of the ciliary processes, potentially disrupting the ciliary-vitreous interface and causing posterior rotation of the ciliary body. However, the irradiation conditions required to disrupt the ciliary-vitreous interface have not been established, and when medication and laser alone are insufficient, surgery to create a unicameral eye remains the final treatment option 3).