Mild (Grade I–II)
Risk of elevated intraocular pressure: Approximately 13.5%.
Visual impairment: Remains mild if the pupillary area is clear.
Fundus visibility: Usually possible.
Traumatic hyphema is a condition in which red blood cells accumulate in the anterior chamber (the space between the cornea and iris) after blunt trauma. A small amount of bleeding that can only be seen with a slit lamp is called microhyphema.
When blunt force is applied to the eye, the intraocular pressure rises sharply and the limbus stretches. The aqueous humor moves posteriorly and toward the angle, damaging the iris and ciliary body, leading to bleeding. The most vulnerable part is the thin attachment of the iris to the ciliary body; a tear here (iridodialysis) displaces the pupil. If a cleft forms in the ciliary body slightly scleral side, angle recession occurs; if the ciliary body detaches from the sclera further scleral side, cyclodialysis results. Both of these are particularly prone to hyphema.
It is classified according to the amount of bleeding as follows.
| Grade | Degree of bleeding |
|---|---|
| 0 | Microhyphema |
| I | Less than 1/3 of the anterior chamber |
| II | 1/3 to 1/2 of the anterior chamber |
| III | 1/2 to less than total filling of the anterior chamber |
| IV | Total hyphema |
Among grade IV, when the anterior chamber is completely filled with bright red blood, it is called total hyphema. When filled with dark red to black blood, it is called 8-ball hyphema (black ball hyphema), suggesting impaired aqueous humor circulation and hypoxia.
Besides trauma, hyphema can occur spontaneously after intraocular surgery (iatrogenic), due to iris neovascularization, ocular tumors, blood disorders (e.g., leukemia, hemophilia), or use of anticoagulants. See the section “Causes and Risk Factors” for details.
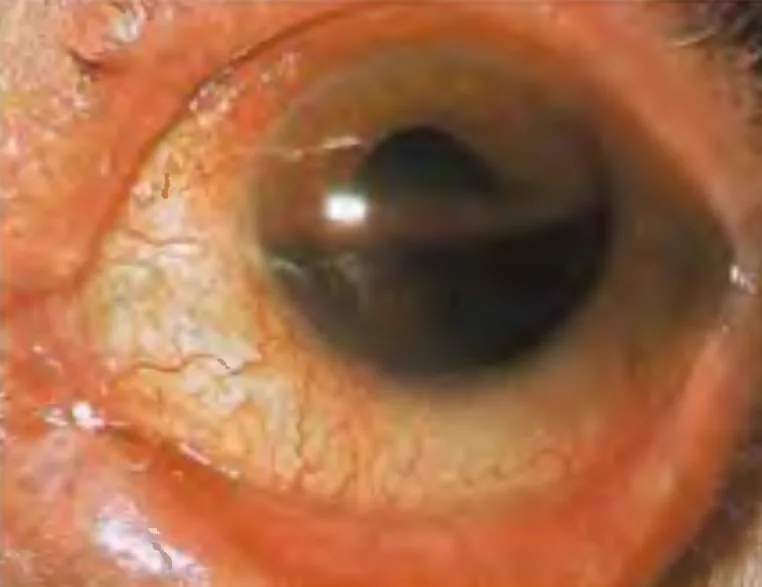
Symptoms of traumatic hyphema vary depending on the amount of bleeding.
Hyphema forms a layer inferiorly (layering), and its color changes from red to black over time. Clotted blood appears darker. In the upright position, the height of the blood layer serves as an indicator of the amount of bleeding and is useful for monitoring absorption or rebleeding. It is important to record the height from the inferior corneal limbus in millimeters.
Mild (Grade I–II)
Risk of elevated intraocular pressure: Approximately 13.5%.
Visual impairment: Remains mild if the pupillary area is clear.
Fundus visibility: Usually possible.
Severe (Grade III–IV)
Risk of elevated intraocular pressure: Increases dramatically to 27% in Grade III and 52% in Grade IV.
Eight-ball hyphema: High risk of pupillary block and secondary angle closure.
Fundus visibility: Often impossible; ultrasound examination is required.
The main complications are as follows.
The following complications are important in children:
Blunt trauma is the most common cause. Typical examples include balls, fists, traffic accidents, and occupational injuries. Compression force to the eye causes rupture of blood vessels in the iris, ciliary body, and trabecular meshwork, leading to accumulation of red blood cells in the anterior chamber.
In hyphema without trauma history, consider the following causes.
Sickle cell disease is a particularly important risk factor. In the hypoxic environment of the anterior chamber, red blood cells sickle and become rigid, making them difficult to pass through the trabecular meshwork. As a result, even a small amount of bleeding can cause severe intraocular pressure elevation. Furthermore, sickled red blood cells within vessels can cause central retinal artery occlusion or ischemic optic neuropathy. Sickle cell trait also poses a risk.
Cataract surgery while continuing warfarin increases bleeding events, but most are self-limited hyphema or subconjunctival hemorrhage, with no adverse effects on postoperative visual acuity5). However, collaboration between the primary care physician and ophthalmologist is important for individual risk assessment.
The diagnosis of hyphema involves the following stepwise examinations. Basic assessments include history taking, visual acuity testing, pupillary light reflex testing, intraocular pressure measurement, and slit-lamp microscopy. In cases of massive hyphema, the consensual light reflex of the fellow eye should also be checked.
| Test | Main Purpose |
|---|---|
| Slit-lamp microscopy | Grade classification and follow-up |
| Gonioscopy (1–2 weeks after injury) | Angle recession, peripheral anterior synechiae |
| UBM / anterior segment OCT (UBM contraindicated in penetrating ocular trauma) | Structural evaluation of ciliary body and angle |
| Ultrasound (B-mode) | Posterior segment evaluation when fundus is not visible |
| CT / MRI (MRI contraindicated for metallic foreign bodies) | Evaluation of globe rupture and intraocular foreign bodies |
The basic principle of treatment is to wait for spontaneous absorption with rest. Strenuous exercise is prohibited, supine position is avoided, and the patient is kept at rest with the head of the bed elevated 30–45 degrees in a sitting or semi-recumbent position. Hospitalization is recommended for children, cases where the hyphema level exceeds 1/3 to 1/2 of the anterior chamber, patients who cannot follow instructions, and those with sickle cell disease accompanied by elevated intraocular pressure.
For elevated intraocular pressure, add the following (4 alone, or 5+6, or a combination of 4–6):
Antifibrinolytic agents (tranexamic acid) are useful for reducing the risk of rebleeding 4). However, their effect on visual prognosis is unclear 4).
Approximately 5% of traumatic hyphema cases require surgery.
Indications for surgery are as follows:
| Patient Group | Surgical Indication Criteria |
|---|---|
| Healthy individuals | IOP ≥50 mmHg for 5 days, or ≥35 mmHg for 7 days |
| Patients with sickle cell disease | IOP ≥25 mmHg for more than 24 hours |
| Signs of corneal blood staining | Indicated without waiting for the above criteria |
| Total hyphema in children | Early intervention is indicated considering the risk of amblyopia due to visual deprivation |
Glaucoma surgery (e.g., filtering surgery) is indicated when high intraocular pressure persists after anterior chamber washout. Lens extraction is necessary if lens dislocation or damage is present. Laser iridotomy should be considered for pupillary block.
In many cases, outpatient management is possible with close follow-up. However, hospitalization is recommended for children, cases with large amounts of bleeding (exceeding 1/3 to 1/2 of the anterior chamber), sickle cell disease with elevated intraocular pressure, or when the patient cannot comply with activity restrictions.
When blunt force is applied to the eye, intraocular pressure rises sharply. The corneal limbus stretches, and aqueous humor moves posteriorly and into the angle. This mechanical change damages the blood vessels of the iris and ciliary body, causing bleeding into the anterior chamber.
Depending on the severity of the injury, the following stepwise structural damage occurs:
Rebleeding occurs 3 to 7 days after injury due to contraction and lysis of the initial clot 4). The incidence is reported to be 5–10% 4). Rebleeding is often more severe and copious than the initial bleed. Elevated intraocular pressure is observed in more than 50% of rebleeding cases.
Risk factors for rebleeding include the following:
Intraocular pressure elevation associated with hyphema occurs through multiple mechanisms.
When severe hyphema is accompanied by sustained high intraocular pressure, the posterior surface of the cornea becomes stained with blood. This may leave visual impairment even after the hyphema resolves, and early anterior chamber washout is necessary.
This is an important chronic complication after blunt trauma. A tear occurs between the circular and longitudinal muscles of the ciliary body, causing recession of the angle. In cases with angle recession of 180 degrees or more, glaucoma develops at a high rate of 6–20% over 10 years. Since onset often occurs several years or more after injury, long-term intraocular pressure follow-up is essential.
The overall incidence of rebleeding is 5–10%, and it often occurs 3 to 7 days after injury4). Since rebleeding tends to be more severe than the initial hemorrhage, it is important to maintain rest and undergo close observation during this period.
The most important long-term complication is angle-recession glaucoma. In cases with angle recession of 180 degrees or more, glaucoma develops in 6–20% within 10 years. Since it often occurs several years or more after injury, regular follow-up of intraocular pressure, visual field, and optic nerve is essential after trauma.
With the widespread use of molecular targeted drugs, drug-related spontaneous hyphema has been reported.
Aldecoa et al. (2023) reported spontaneous hyphema in a 60-year-old woman taking ibrutinib (BTK inhibitor) 420 mg/day for 4 months for chronic lymphocytic leukemia 1). The hyphema completely resolved within 2 weeks after discontinuation of ibrutinib and topical steroid eye drops. Ibrutinib is suggested to reduce platelet adhesion to von Willebrand factor and inhibit collagen-induced platelet aggregation.
Chiang et al. (2022) reported spontaneous hyphema in a 37-year-old man with acute myeloid leukemia and severe COVID-19 pneumonia 2). In addition to severe thrombocytopenia (6×10⁹/L), prolonged prone positioning was presumed to contribute to increased episcleral venous pressure.
Ison et al. (2022) reported spontaneous hyphema from iris microhemangiomas (Cobb’s tufts) in a 56-year-old woman with Eisenmenger syndrome 3). Chronic hypoxemia (resting SpO₂ 78%) and secondary erythrocytosis (Hb 22.5 g/dL) were thought to induce dilation of iris stromal vessels and contribute to microhemangioma formation. Bleeding resolved with topical atropine and dexamethasone.
Zhang et al. (Pictures & Perspectives) reported spontaneous hyphema and posterior chamber hemorrhage after ICL (implantable collamer lens) surgery 6). A 23-year-old woman presented with sudden vision loss without trauma, eye rubbing, or anticoagulant use. UBM examination revealed rupture of an iris-ciliary body cyst associated with the ICL haptics and surrounding hemorrhage. Bleeding resolved with conservative treatment using tobramycin-dexamethasone eye drops (4 times daily) and 1% atropine sulfate gel (twice daily) for 17 days. Emergency ICL removal was not necessarily required.