Grade 1
Maximum diameter of edema: ≤3 mm
Lesion characteristics: Localized stromal edema, confined to a part of the cornea.

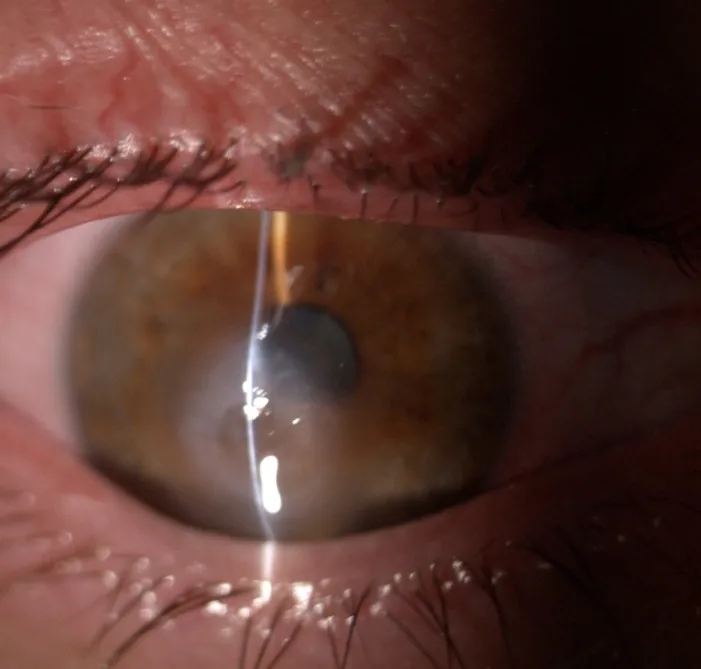
Acute corneal hydrops (ACH) is a rare complication of corneal ectatic disorders. It is a condition in which a tear in Descemet’s membrane and the corneal endothelium allows aqueous humor to rapidly flow into the corneal stroma, causing acute corneal edema.
The most common underlying disease is keratoconus, but it also occurs in keratoglobus and pellucid marginal degeneration (PMD).
Incidence rates are as follows1):
Age of onset ranges from 5 to 59 years, with a peak in the 20s to 40s1). The risk in men is up to twice that in women. Ethnic differences are also observed, with higher prevalence in Pacific Islanders in New Zealand and in South Asians and Black people in the UK.
Recent findings indicate that acute corneal hydrops (ACH) does not occur with a Descemet membrane defect alone; a defect in the posterior corneal stroma is also necessary. A report noted that ACH did not occur in 16 eyes with Descemet membrane defects during DMEK, but occurred in all 5 eyes with posterior stromal perforation 1).
It is reported to occur in approximately 2.4–3% of patients with keratoconus, 6–11.5% of patients with pellucid marginal corneal degeneration, and about 11% of patients with keratoglobus 1). The risk is higher with more severe corneal ectasia, and special attention is needed in patients with a habit of eye rubbing.
The main subjective symptoms of ACH are as follows.
Triggers include actions that temporarily increase intraocular pressure, such as eye rubbing, sneezing, coughing, blowing the nose, and vigorous exercise. It often occurs spontaneously.
Slit-lamp microscopy reveals conjunctival hyperemia, corneal stromal edema, epithelial edema, intrastromal clefts, and epithelial bullae1).
The grade classification based on the extent of edema is shown below1).
Grade 1
Maximum diameter of edema: ≤3 mm
Lesion characteristics: Localized stromal edema, confined to a part of the cornea.
Grade 2
Maximum diameter of edema: 3–5 mm
Lesion characteristics: Moderate extent of edema, may affect the visual axis.
Grade 3
Maximum diameter of edema: >5 mm
Lesion characteristics: Extensive edema, presenting severe opacity including the visual axis. The youngest case (5-year-old girl) reported limbus-to-limbus total corneal edema8).
For corneal transparency assessment, a 5-grade classification from Grade 0 (anterior chamber and iris completely invisible) to Grade 4 (completely transparent) is also used1).
Anterior segment OCT (AS-OCT) can non-invasively visualize Descemet membrane rupture and detachment, intrastromal fluid clefts, and increased corneal thickness1). The size and depth of the tear are predictive indicators of ACH resolution time. Seidel test may be positive, but this is due to aqueous humor leakage from the edematous cornea, not perforation.
There are reports of ACH occurring in cases complicated by Graves ophthalmopathy, and elevated intraocular pressure and ocular surface inflammation may be risk factors7).
Regarding the risk of infection, a report indicates that 2.7% (3/112 cases) of ACH cases were complicated by infectious keratitis 5). All complicated cases were males with severe atopic dermatitis, with an average age of 46 years. Cultures detected MSCNS and MSSA.
Rubbing the eye applies mechanical force to Descemet’s membrane, and in a cornea that is already thinned and ectatic, it can directly trigger a rupture of Descemet’s membrane 1). Patients with atopic dermatitis are particularly at high risk because they tend to rub their eyes due to itching. If itching occurs, it is important to control symptoms with eye drops or other medications.
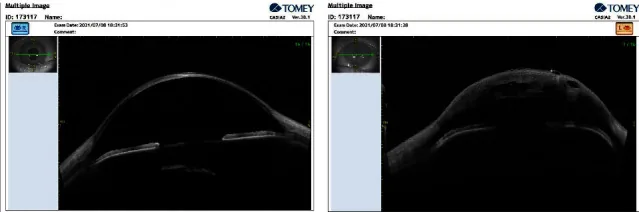
The diagnosis of ACH is based on clinical findings and supplemented by various imaging tests.
This is the basis of diagnosis. Conjunctival injection, stromal edema, epithelial edema, and intrastromal clefts are identified 1). A Seidel test is performed to differentiate from perforation.
The characteristics of the main examination modalities are shown below.
| Examination Method | Main Evaluation Content | Features |
|---|---|---|
| AS-OCT | DM rupture, stromal clefts | Non-contact, quantitative |
| Ultrasound biomicroscopy | Rupture site, corneal thickness | Resolution monitoring |
| In vivo confocal microscopy (IVCM) | Inflammatory cells, edema | Cellular-level assessment |
Treatment for ACH begins with conservative management; surgical treatment is considered when improvement is poor or early resolution is needed.
The following medications are used1)9).
Spontaneous resolution with conservative treatment takes 2–4 months (maximum 5–36 weeks)1). In eyes that have undergone corneal cross-linking (CXL), resolution is significantly faster, with complete resolution reported within 3 weeks7).
Performed to promote early resolution, relieve symptoms, and minimize corneal scarring.
Intracameral Gas Injection
Pneumatic descemetopexy: Air, SF6, or C3F8 is injected into the anterior chamber to unfold and reattach the edge of Descemet’s membrane through a tamponade effect 1).
Resolution time: 2 to 7 weeks (significantly shorter than 64 to 117 days with conservative treatment).
Reinjection rate: Air 77.8%, SF6 66.7%, C3F8 0 to 35.7% require reinjection.
Compression Sutures
Compression sutures: Sutures compress and close the Descemet’s membrane tear. Includes full-thickness sutures (FTS) and partial-thickness sutures (PTS) 2).
Resolution time: Literature review reports 1 hour to 45 days; most within 2 weeks 2).
Combined with intracameral air: Complete resolution within 2 weeks reported with 3 FTS plus air (50% anterior chamber fill) 3).
Corneal Transplantation
Descemet stripping automated endothelial keratoplasty (DSAEK)/Descemet membrane endothelial keratoplasty (DMEK): Considered for large Descemet’s membrane tears 1).
Deep anterior lamellar keratoplasty (pDALK): Can achieve DM closure, corneal curvature recovery, and visual recovery in a single surgery.
Penetrating keratoplasty (PKP): Performed for visually significant scarring after ACH. Long-term graft survival after ACH is lower than in non-ACH cases 3).
A representative postoperative eye drop regimen after compression sutures is shown 3).
| Medication | Dose/Frequency |
|---|---|
| moxifloxacin 0.5% | 4 times/day |
| prednisolone acetate 1% | taper as appropriate |
| NaCl 5% | once at bedtime |
Many cases resolve within 2 to 4 months with conservative treatment alone 1). However, corneal scarring and neovascularization may persist after resolution, leading to permanent visual impairment. Intracameral gas injection or compression sutures can shorten resolution to about 2 weeks and minimize scar formation. It is important to discuss treatment options with your doctor.
This is a surgery in which air or a special gas (SF6, C3F8, etc.) is injected into the anterior chamber, and the tamponade effect supports the torn Descemet’s membrane from the inside to promote reattachment 1). It can shorten the resolution time from 64–117 days with conservative treatment to 2–7 weeks. However, pupillary block glaucoma or the need for reinjection may occur.
Conventionally, the following pathway has been assumed: microtrauma such as eye rubbing → Descemet’s membrane tear → aqueous humor penetrates into the stroma → separation of lamellar layers → edema 1).
Recent studies have shown that Descemet’s membrane defect alone does not cause ACH; a defect in the posterior corneal stroma is also essential 1). This finding is important for explaining why ACH occurs only in cases of severe corneal ectasia.
In full-thickness corneal transplant (PKP) specimens of ACH corneas, loosely packed stromal lamellae are observed. Laminin and type IV collagen are localized adjacent to the rolled-up Descemet’s membrane, and this is thought to play an important role in endothelial cell migration and re-endothelialization 1).
The anterior one-third of the corneal stroma remains mildly swollen due to lamellar interweaving. The posterior two-thirds consist of non-interwoven unidirectional fibrils and can swell up to three times 7).
Adjacent corneal endothelial cells migrate to the Descemet’s membrane tear, restore endothelial pump function, leading to corneal dehydration, and finally repair is completed by scar formation 1). Compression sutures physically approximate the edges of Descemet’s membrane, promoting endothelial cell migration.
Studies using in vivo confocal microscopy have shown that cases where inflammatory cells persist for more than 4 weeks have a high risk of corneal neovascularization 1).
Varshney et al. (2021) reported a case of ACH complicating keratitis after CXL 7). In the CXL-treated cornea, resolution occurred rapidly in 21 days, significantly shorter than the usual 2–4 months. The mechanism was suggested to be enhanced collagen crosslinking by CXL → increased interlamellar cohesive strength in the anterior stroma → increased resistance to swelling. Crosslinked corneas have higher resistance to enzymatic digestion, which is thought to contribute to early resolution.
A technique using intraoperative OCT (iOCT) to adjust suture depth and tension in real time has been reported.
Kaur et al. (2025) performed iOCT-guided partial-thickness compression sutures (PTS, target depth 50–60%) in 7 cases (median age 16 years, preoperative corneal thickness 1120–2363 μm)2). Suturing was performed while confirming stromal compaction with a “caterpillar” appearance via iOCT; all cases resolved within 2 weeks, and sutures were removed after 8–12 weeks. PTS showed efficacy comparable to full-thickness sutures (FTS) with a lower risk of endothelial damage.
A literature review (19 studies) reported that resolution time for compression sutures ranged from 1 hour to 45 days, with most resolving within 2 weeks2).
Liu et al. reported a two-stage procedure: TKP-assisted epikeratophakia (first stage for Descemet membrane repair) followed by DALK using the same graft (second stage) after 2.1±0.7 months to achieve visual recovery2).
Petrelli et al. performed lamellar wedge resection for corneal perforation after ACH when cyanoacrylate adhesion was unsuccessful2). This achieved refractive improvement while avoiding corneal transplantation.
Alio et al. performed an anterior chamber injection of 0.3 ml of Enriched Platelet-Rich Plasma (EPRP) in cases unresponsive to SF6, achieving resolution within one week 1). The mechanism was presumed to be promotion of endothelial cell regeneration by growth factors.
For cases with large Descemet membrane tears, a method using a 5 mm diameter small DMEK patch to temporarily seal the tear has been reported 1). Cases have been reported where the cornea naturally achieved deturgescence even after partial graft detachment.
Ontiveros-Holguín & Pacheco-Padrón (2022) reported a case of ACH and ICRS migration after intracorneal ring segment (ICRS) implantation 8). It resolved after 8 weeks with conservative treatment alone. This is considered the first report of ACH after ICRS implantation recovering with only conservative management.
This is a compression suture performed while confirming suture depth and tension in real time using intraoperative OCT (iOCT) 3). Since it allows objective control of suture depth, which previously relied on experience, effective corneal compression can be achieved while reducing the risk of endothelial damage. In the report by Kaur et al., all cases resolved within two weeks.