Epithelial type
Dendritic keratitis: Characteristic branching epithelial lesions with terminal bulbs
Geographic keratitis: A condition in which epithelial defects enlarge due to prolonged dendritic keratitis

Herpes simplex keratitis is an inflammation of the cornea caused by infection with the herpes simplex virus (HSV). In most cases, the causative virus is type 1 (HSV-1), and cases caused by type 2 (HSV-2) are extremely rare. HSV is a neurotropic virus belonging to the Alphaherpesvirinae subfamily of the Herpesviridae family, which establishes latent infection in the trigeminal ganglion and persists throughout life.
Few cases develop keratitis during primary infection. Typically, HSV latent in the trigeminal ganglion is reactivated by triggers such as fever, cold, stress, or ultraviolet exposure, and reaches the cornea via the trigeminal nerve, causing various lesions. Its most important feature is that it is a recurrent disease, and careful history taking regardless of disease type is the starting point for determining diagnosis and treatment strategy.
Based on population-based estimates from 2016, the overall incidence of HSV keratitis was estimated at 24.0 per 100,000 people per year, with approximately 1.7 million people affected worldwide annually2). Of these, epithelial keratitis accounts for about 16.1 per 100,000 (approximately 1.2 million per year), and stromal keratitis accounts for about 4.9 per 100,000 (approximately 370,000 per year)2). An estimated 230,000 people per year develop new unilateral visual impairment (visual acuity below 6/12) related to HSV keratitis2).
Approximately 67% of the world’s population (4.85 billion people) have been infected with HSV-1. HSV-1 prevalence is particularly high outside the United States and Europe, and treatment access is limited, so the actual disease burden is likely underestimated2). HSV keratitis is the leading cause of corneal blindness in developed countries. In particular, 24–42% of stromal keratitis cases result in corrected visual acuity below 6/12, and 1.5–3.0% progress to severe visual impairment (below 6/60)2).
Japan’s own epidemiological surveys, such as the nationwide surveillance of infectious keratitis, have shown that viral keratitis occupies an important position alongside bacterial, fungal, and Acanthamoeba keratitis. HSV keratitis is positioned as a representative disease in Japan that follows a chronic and recurrent course and affects visual prognosis1). In terms of age distribution, cases are more common in middle-aged and older individuals, but initial onset is observed across a wide range from children to the elderly. Age-related decline in cell-mediated immunity is a factor that increases the incidence rate6).
The disease classification proposed by the Ocular Herpes Infection Study Group (Yuichi Ohashi et al., 1995) has also been adopted in the third edition of the Infectious Keratitis Clinical Practice Guidelines1). This classification is based on the localization and pathological differences of lesions and is an important framework that determines treatment strategy.
Epithelial type
Dendritic keratitis: Characteristic branching epithelial lesions with terminal bulbs
Geographic keratitis: A condition in which epithelial defects enlarge due to prolonged dendritic keratitis
Stromal type
Disciform keratitis: Circular stromal opacity and edema due to delayed-type hypersensitivity reaction
Necrotizing keratitis: Dense stromal opacity with vascular invasion, risk of perforation
Endothelial type
Corneal endotheliitis: Primarily corneal edema and keratic precipitates, with mild stromal opacity
Corneal limbitis: Accompanied by fan-shaped edema with the limbus as the base and elevated intraocular pressure
Secondary Lesions
Persistent epithelial defect: Secondary epithelial defect due to delayed wound healing
Neurotrophic corneal ulcer: Ulcer due to sensory nerve impairment and abnormal wound healing
The epithelial form is a condition caused by direct viral replication in epithelial cells, while the stromal form is primarily driven by immune and inflammatory responses to the virus. Whether the endothelial form is primarily due to direct infection or immune response has not been fully elucidated. Neurotrophic corneal ulcer is a wound-healing abnormality without viral replication and is a secondary complication rather than a direct HSV lesion. These differences in pathophysiology lead to fundamentally distinct treatment strategies.
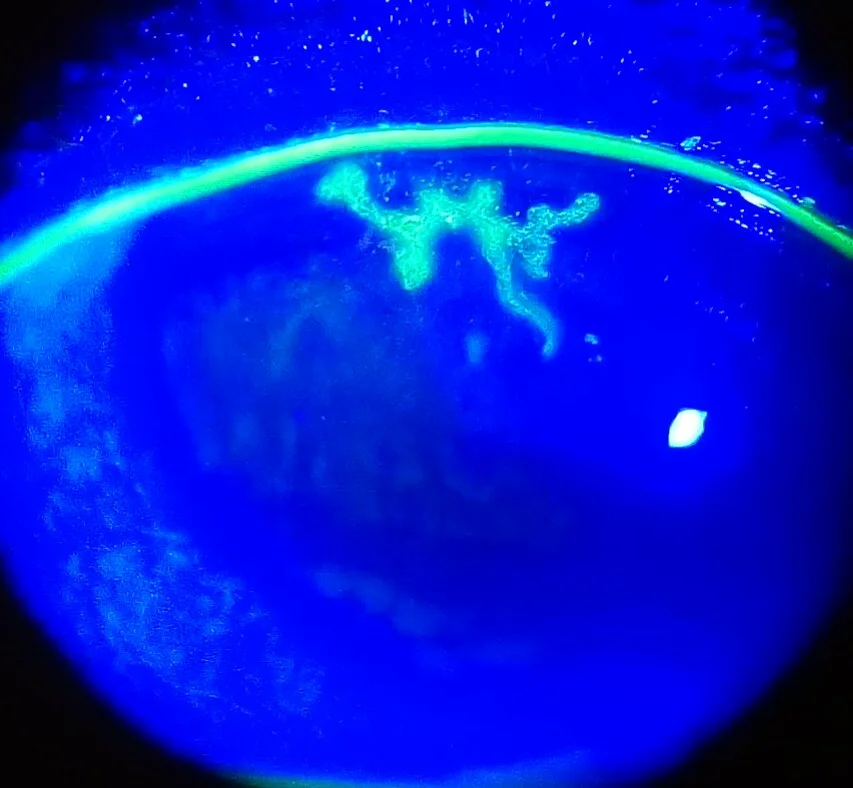
The subjective symptoms of herpes simplex keratitis vary greatly depending on the disease type.
Dendritic keratitis, the representative lesion of epithelial herpes keratitis, has four characteristic features that are key to diagnosis1).
On fluorescein staining, the ulcer base stains positive, and the swollen marginal epithelium stains with rose bengal. When dendritic keratitis persists, the epithelial defect enlarges and takes the form of geographic keratitis. This tends to progress in immunocompromised patients and those using steroids; the marginal findings are preserved, and the presence of dendritic tails (suggesting a dendrite-like pattern) serves as a diagnostic clue.
Herpes simplex keratitis is typically unilateral, but the frequency of bilateral cases is reported to be 1.3–12% in the literature6). Atopic predisposition, rheumatoid arthritis (40% bilateral frequency), immunosuppression, and pediatric cases (26%) are associated with a higher risk of bilaterality6).
Disciform keratitis, the classic form of stromal herpes simplex keratitis, is characterized by the following findings1).
As recurrences repeat, the opacity becomes irregular in shape and extends to deeper layers. In addition to opacity, a wide variety of lesion morphologies can appear, making diagnosis based on slit-lamp findings alone difficult in many cases.
Necrotizing keratitis is a condition in which HSV proliferates within the corneal stroma or an excessive immune response to antigens occurs1). It tends to occur with repeated recurrences, particularly in eyes with pre-existing vascular invasion, scar formation, or fatty degeneration within the stroma. It presents with dense, irregular stromal opacity accompanied by vascular invasion, and in cases with epithelial defects, stromal thinning may progress to perforation. In recent years, severe cases have become rare due to early and appropriate treatment.
Corneal endotheliitis is diagnosed clinically when corneal epithelial and stromal edema with KP corresponding to the lesion are present, but corneal stromal opacity and anterior chamber cells are minimal1). The typical form of HSV-induced endotheliitis is the paracentral edema type, in which corneal edema occurs in a fan shape with the limbus as its base, and KP is observed at its center. It may be accompanied by elevated intraocular pressure due to trabeculitis and limbitis.
The keratouveitis type is a severe form that combines stromal keratitis, endotheliitis, and iritis, presenting with marked conjunctival injection, corneal edema, Descemet’s membrane folds, KP, and anterior chamber cells; in severe cases, hypopyon may develop.
In patients with recurrent episodes, corneal hypoesthesia becomes prominent and serves as an important supportive finding for diagnosis regardless of disease type. Stromal and endothelial types are more difficult to diagnose virologically than the epithelial type, making corneal sensitivity testing more important. Using a Cochet-Bonnet aesthesiometer, the sensory threshold can be evaluated in stages by adjusting the length of the nylon filament, and it is also useful for confirming left-right differences. When there is a history of unilateral recurrent redness and obvious hypoesthesia is found on the affected side, this strongly suggests corneal herpes regardless of the disease type. However, some cases in the early recurrence of the epithelial type do not show clear hypoesthesia, so this disease cannot be ruled out by a negative sensitivity test alone.
According to the literature, the incidence of bilateral HSV keratitis ranges from 1.3% to 12%6). In a 30-year retrospective study, 4% of all patients had bilateral involvement at initial onset, and an additional 1% became bilateral upon recurrence6). A large-scale Korean study reported 12%, and an Indian study reported up to 25% bilateral frequency6). The rate is particularly high in patients with rheumatoid arthritis (40%) and in children (26%), and caution is needed because atopic predisposition and immunosuppressive states also increase the risk of bilaterality6).
Herpes simplex keratitis is caused by HSV, a double-stranded DNA virus. HSV-1 primarily infects the orofacial region, and HSV-2 primarily infects the genital region, but both can cause ocular infection. The majority of corneal herpes cases are caused by HSV-1.
Primary infection often occurs asymptomatically in childhood. The virus travels through sensory nerve axons to the trigeminal ganglion, where it establishes lifelong latent infection. The latent virus reactivates in response to certain triggers, travels via the trigeminal nerve to the cornea, and causes recurrent inflammation.
The risk of bilateral involvement is high in the following conditions6).
HSV keratitis after corneal transplantation has been reported with an incidence of 9.73% in a Chinese retrospective study (411 cases)7). Sixty-five percent of cases occurred within 1 to 3 months postoperatively, and postoperative steroid use is thought to promote viral reactivation7). The distribution of post-transplant HSV keratitis by clinical type was: epithelial 27.5%, necrotizing stromal 20%, mixed 42.5%, and endothelial 10%7). Corneal transplantation in eyes with a history of HSV keratitis carries a significantly higher risk of rejection and graft failure compared to transplantation for keratoconus or Fuchs endothelial corneal dystrophy7).
The Infectious Keratitis Clinical Practice Guideline, 3rd Edition1) presents the following based on the diagnostic criteria of the Ocular Herpes Infection Study Group.
However, since virus isolation and culture are rarely performed in everyday clinical practice, diagnosis is actually made by combining slit-lamp findings, auxiliary diagnostic criteria, and virological testing.
In typical cases, diagnosis is possible based on slit lamp findings alone. In the epithelial form, a dendritic ulcer with terminal bulbs is observed; in the stromal form, discoid superficial opacity and an immune ring are seen; and in endothelitis, sectoral corneal edema and keratic precipitates are characteristic findings. Fluorescein staining and rose bengal staining under a Wood filter are used in combination.
Corneal sensitivity testing is a valuable supplementary examination for diagnosing herpes keratitis. The Cochet-Bonnet aesthesiometer is recommended because it is simple to use and allows stepwise evaluation of the degree of decreased corneal sensation1). In cases with repeated recurrences, marked hypoesthesia is observed; however, this test is not specific and is not necessarily useful in first-episode or mild cases.
The third edition of the Clinical Practice Guidelines for Infectious Keratitis conditionally recommends the following tests1).
It is important to differentiate from pseudodendritic keratitis and other conditions with similar presentations.
| Differential Diagnosis | Key Differentiating Features |
|---|---|
| Herpes zoster keratitis (VZV) | Pseudodendritic lesions: no terminal bulbs, weak fluorescein staining, Hutchinson sign, accompanied by skin rash |
| Acanthamoeba keratitis | Severe pain, radial keratoneuritis, contact lens wear history |
| Recurrent corneal erosion | Acute onset upon waking, severe pain, history of trauma |
| Toxic keratopathy (medication-induced) | History of eye drop use, diffuse punctate keratopathy |
| CMV corneal endotheliitis | Coin-shaped lesions, linear KP, unresponsive to acyclovir |
| Corneal graft rejection | KP localized to the graft posterior surface, Khodadoust line, excellent response to steroids |
Differentiating between endotheliitis and graft rejection after corneal transplantation is particularly important. In post-transplant HSV endotheliitis, KP are distributed beyond the donor–recipient junction and do not improve with steroids alone, requiring the addition of antiviral therapy 7). In contrast, in endothelial rejection, KP are mainly localized to the posterior graft surface, with a Khodadoust line progressing from the periphery toward the center 7).
Corneal sensitivity testing is a valuable adjunctive test for diagnosing herpes keratitis1). Particularly in cases with recurrent episodes, a marked decrease in corneal sensitivity is observed. The Cochet-Bonnet esthesiometer is useful as it is simple and allows for severity grading. However, it is not a specific test and cannot establish a definitive diagnosis on its own. Because virus isolation and PCR have lower sensitivity in stromal and endothelial types than in the epithelial type, obtaining a medical history (history of recurrences) and confirming decreased corneal sensitivity are more important for diagnosis.
Treatment of herpes simplex keratitis differs fundamentally depending on the disease type. The epithelial type is caused by direct viral proliferation, so antiviral agents are the mainstay and steroid eye drops are contraindicated. The stromal and endothelial types are primarily driven by immune reactions, so the principle is to combine antiviral agents with steroid eye drops.
The first-line treatment is acyclovir (ACV, Zovirax®) ophthalmic ointment 3% administered 5 times daily1). Compliance with the dosing frequency is important due to the short half-life, and low doses carry a risk of resistant strains. After the epithelial lesions have resolved, reduce the frequency to 3 times daily and discontinue after 1 to 2 weeks. Concomitant use of antibacterial eye drops (such as levofloxacin 0.5% 3 times daily) is recommended to prevent mixed infection.
| Drug | Dosage | Characteristics |
|---|---|---|
| ACV ophthalmic ointment 3% (Zovirax®) | Instill 5 times daily | First-line treatment. Strict adherence to frequency is important due to short half-life |
| Valacyclovir tablet (Valtrex®) 500mg | 2 tablets, twice daily after meals | Prodrug of ACV. Switch in case of side effects or resistant strains |
| Levofloxacin ophthalmic solution 0.5% (Cravit®) | Instill 3 times daily | To prevent mixed infection |
| Trifluridine ophthalmic solution 1% (TFT) | 8 to 9 times daily | Effective against ACV-resistant strains. Used in the US (not approved in Japan) |
Managing side effects: ACV ophthalmic ointment may cause superficial punctate keratopathy in the central inferior cornea or blepharoconjunctivitis (base reaction). If mild, dose reduction may allow continued use, but in severe cases, switch to oral valacyclovir 500 mg, 2 tablets twice daily after meals. If no improvement after 1 week of continuous ACV ointment, consider ACV-resistant strains or other conditions presenting with pseudodendritic keratitis.
Systemic administration: The Infectious Keratitis Clinical Practice Guidelines, 3rd edition, conditionally recommend systemic administration of antiviral drugs for epithelial keratitis (weak recommendation, evidence level C)1). Oral acyclovir 2,000 mg/day is expected to have equivalent efficacy to ACV ophthalmic ointment and may be useful in cases of ointment toxicity, resistant strains, or poor adherence. However, oral ACV for HSV keratitis is not covered by insurance in Japan1).
Steroid eye drops are contraindicated in epithelial keratitis. Steroids promote viral activation and worsen the condition.
Debridement (epithelial scraping): To reduce the viral load, scraping of dendritic lesions may be performed in combination with antiviral drugs.
Since stromal keratitis is primarily driven by an immune response to the virus, the principle is suppression of the immune response with topical steroids combined with ACV ophthalmic ointment1). If treated with steroids alone without ACV ophthalmic ointment, symptoms may initially improve, but recurrence and reactivation are more likely, and there is a risk of inducing epithelial keratitis during the course1).
In the HEDS (Herpetic Eye Disease Study)-1 SKN (Stromal Keratitis Not on Steroids) trial, topical corticosteroids (prednisolone phosphate in a tapering regimen) were shown to reduce the risk of persistent or progressive inflammation by 68% and shorten healing time compared to placebo in HSV stromal keratitis3). At 6 months, the treatment failure rate was significantly lower in the active drug group, and earlier visual improvement was also demonstrated3). This is the key evidence supporting the use of topical steroids for stromal keratitis, and it has been adopted as the basis of stromal keratitis treatment in the Japanese Infectious Keratitis Clinical Practice Guidelines, 3rd edition1). However, since the HEDS trial design did not demonstrate an additional benefit of oral ACV, the concomitant use of systemic antiviral agents should be determined on a case-by-case basis.
Topical steroid therapy in practice:
Concurrent antiviral therapy: Use ACV ophthalmic ointment 3% 5 times daily in combination1).
Oral steroids: In cases with severe inflammation such as keratouveitis or when epithelial defects are present, add oral betamethasone (Rinderon® tablets 0.5 mg, 2 tablets once daily after breakfast) in addition to eye drops.
Prescription example (severe stromal keratitis):
Prescription example (with epithelial defect):
Endothelial keratitis is treated according to the same protocol as stromal keratitis1). When it coexists with stromal keratitis, the treatment follows that for stromal keratitis. There is no consensus on whether ACV administration for pure endothelial keratitis should be topical or systemic. When corneal edema becomes irreversible, corneal endothelial transplantation is indicated.
For HSV anterior uveitis presenting with unilateral mutton-fat KP, anterior chamber inflammation, and elevated intraocular pressure, a combination of antiviral agents, steroid eye drops, pupil management, and intraocular pressure control is used.
Trophic corneal ulcer is a wound healing disorder that does not involve viral proliferation, so the treatment strategy differs significantly.
The Infectious Keratitis Clinical Practice Guidelines, 3rd edition conditionally recommend systemic antiviral administration for preventing recurrence of epithelial herpes keratitis (weak recommendation, evidence C)1).
In the HEDS-APT trial, 12-month administration of ACV 400 mg twice daily reduced the risk of recurrence of epithelial and stromal HSV disease by nearly half, with 19% in the active drug group versus 32% in the placebo group4). Longer-term administration exceeding 12 months has been shown to further prolong recurrence intervals4). While remaining mindful of the potential emergence of ACV-resistant strains, this is an effective option for cases with frequent recurrence or high-risk cases.
Corneal transplantation is indicated when scarred corneal opacities that do not respond to medical therapy remain. Outcomes have significantly improved in recent years due to advances in surgical techniques and postoperative antiviral prophylaxis5).
Penetrating Keratoplasty (PKP)
Indications: Corneal perforation or full-thickness opacity
Outcomes: Recurrence rate 44%, rejection rate 46% without antiviral prophylaxis5). Relative risk of graft failure reduced to 0.3 with oral ACV5)
Concerns: Immunological rejection is the main cause of graft failure5)
Deep Anterior Lamellar Keratoplasty (DALK)
Indications: Stromal opacity with healthy endothelium
Outcomes: Recurrence rate 6–10%, rejection rate 0–4.5%, 5-year survival rate 96%5). Significantly superior to PKP (5-year survival rate 78.8%)
Advantages: Preserves host endothelium, resulting in fewer immunological rejections5)
Mushroom Keratoplasty (MK)
Indications: Conversion procedure for Descemet membrane perforation during DALK
Outcomes: 10-year graft survival rate 92%, rejection rate 9.7%, recurrence rate 7.8%5)
Features: Two-piece design with 9 mm anterior + 6 mm posterior component. Minimizes endothelial replacement to reduce antigen load5)
Postoperative antiviral prophylaxis: The American Academy of Ophthalmology (AAO) Corneal Edema and Opacification Preferred Practice Pattern and the AAO HSV Keratitis Treatment Guidelines recommend continuing oral ACV 800 mg three times daily or valacyclovir 500 mg twice daily for at least one year as antiviral prophylaxis after corneal transplantation5, 8, 9). The peak period for recurrence is 1–3 months after surgery, and special caution is required during this time7). If HSV keratitis reactivates after transplantation, systemic antiviral therapy should be actively administered, and the steroid dosage should be carefully adjusted according to epithelial status, infection severity, and rejection activity7).
Steroid eye drops are contraindicated in the epithelial type (active epithelial disease). They activate viral replication, leading to progression to geographic ulcers and worsening of the condition. On the other hand, in the stromal type (such as disciform keratitis), the primary mechanism is an immune response to the virus, so the combination of ACV eye ointment and steroid eye drops is the standard treatment1, 3). It is important to taper steroid eye drops gradually over months rather than abruptly discontinuing them1).
In patients with a history of HSV keratitis, it is recommended to start oral antiviral agents (acyclovir 800 mg three times daily or valacyclovir 500 mg twice daily) before surgery and continue for at least one year postoperatively5, 8). The peak recurrence period is 1 to 3 months after surgery, making this period especially important7). Regarding surgical technique, DALK preserves the host endothelium and demonstrates an excellent 5-year survival rate of 96%, compared with PKP (5-year survival rate 78.8%)5). Mushroom keratoplasty also achieves a 10-year graft survival rate of 92%5).
Primary infection: Primary infection usually occurs in childhood through contact with the orofacial area, and most cases are asymptomatic. The virus enters cells via specific receptors on corneal epithelial cells (such as Nectin-1 and HVEM)6). After infection, the virus travels through sensory nerve axons to reach the trigeminal ganglion, where it establishes lifelong latent infection.
Latent infection: HSV-1 maintains its genome within the neuronal nucleus, evading immune surveillance5). During latency, it produces latency-associated transcripts (LAT), which maintain the integrity of the viral genome while suppressing cellular apoptosis6). CD8-positive T cells and cytokines suppress viral gene expression, keeping the virus in a dormant state5).
Reactivation: Various triggers cause latent virus to reactivate and reach the cornea along the trigeminal nerve. In COVID-19 patients, immunosuppression and cytokine storm syndrome occur, and exhaustion of HSV-1-specific CD8-positive T cells is thought to promote reactivation of latent virus6).
In epithelial herpes simplex keratitis, HSV actively replicates in corneal epithelial cells. The characteristic feature is that the virus actively replicates at the margins rather than in the epithelial defect. Dendritic lesions form along the corneal nerve pattern. With repeated recurrences, corneal sensory nerves become damaged and hypoesthesia progresses.
HSV stromal keratitis is primarily driven by an immune response led by CD4-positive T cells. Langerhans cells (antigen-presenting cells of the cornea) that have taken up HSV antigens present them to CD4-positive T cells, and activated T cells release cytokines. This causes neutrophil infiltration into the corneal stroma, leading to tissue destruction. Mouse models have shown that removing CD4-positive T cells from the corneal stroma prevents the development of stromal keratitis, confirming that the immune response is central to the pathogenesis of the stromal form.
In addition, molecular mimicry has been demonstrated between the HSV UL6 protein and human corneal antigens, suggesting that an autoimmune mechanism may be involved in the chronicity and recurrence of stromal keratitis.
Difference between necrotizing and non-necrotizing forms: Non-necrotizing (disciform) keratitis is a pure delayed-type hypersensitivity reaction without active viral proliferation. In contrast, in necrotizing keratitis, neutrophil infiltration through new blood vessels occurs in response to antigen-antibody complexes accumulated in the stroma of corneas with repeated recurrences, leading to more severe tissue destruction.
It remains unclear whether endothelialitis is primarily caused by direct viral infection of endothelial cells or by immunological attack. It is thought that both mechanisms may be involved. Endothelialitis caused by HSV often occurs during the course of stromal or epithelial types, whereas isolated endothelialitis without other forms is more frequently attributed to cytomegalovirus (CMV)1).
Neurotrophic corneal ulcer is a wound healing disorder caused by the accumulation of multiple factors during repeated recurrences. Contributing factors include stromal inflammation, corneal nerve damage, epithelial basement membrane damage, and side effects of antiviral drugs. Corneal nerve damage due to HSV infection causes a state of “neurotrophic keratopathy,” which, combined with decreased tear secretion and reduced blink reflex, delays epithelial healing. Clinically, while geographic keratitis with viral replication shows irregular epithelial defects, neurotrophic corneal ulcer is characterized by oval-shaped epithelial defects with gray-white rounded elevation of the marginal epithelium that appears slightly detached from the stroma. Viruses are not isolated, but residual DNA may be detected by highly sensitive PCR. Since viral replication is not involved, the mainstay of treatment is discontinuation of antiviral eye drops and promotion of epithelial healing.
Acyclovir is monophosphorylated by viral thymidine kinase (TK) in infected cells, and then triphosphorylated by host cell kinases. This active form inhibits viral DNA polymerase, blocking viral DNA synthesis. Since it is not phosphorylated in normal cells, it has high selectivity for herpes viruses and few side effects. ACV-resistant strains arise mainly due to loss or mutation of TK activity that prevents ACV phosphorylation, with particularly high risk in immunocompromised patients6).
To address ACV-resistant strains, research is underway on multiple novel molecules targeting different stages of HSV-1 infection6).
BX795 and OGT 2115 have shown efficacy against ACV-resistant HSV strains and are promising candidates6). Since it is difficult for HSV-1 to simultaneously acquire resistance to three different therapeutic mechanisms, combination therapy with multiple molecules is expected as a future treatment strategy6).
Specific metabolic changes (decreased arginine, altered sphingolipid metabolism) have been identified in the tears of patients with active epithelial HSV keratitis, suggesting their potential as novel diagnostic biomarkers through noninvasive tear sampling.
With the introduction of DALK and mushroom corneal transplantation, outcomes of corneal transplantation for HSV keratitis have dramatically improved5). Large-diameter (9 mm) DALK significantly reduces postoperative astigmatism, with 44% achieving visual acuity of 20/20 or better5). A protocol of high initial dosing and long-term tapering has yielded favorable 10-year outcomes even in scarred eyes with neovascularization5).
For HSV keratitis accompanied by severe inflammatory response, large deep ulcers, and persistent epithelial defects, combined pharmacotherapy and amniotic membrane transplantation has been reported7). Amniotic membrane tissue contributes to promoting epithelial healing, reducing inflammation, suppressing fibroblast proliferation, and inhibiting neovascularization7).
Research on therapeutic vaccines aimed at preventing recurrence of HSV keratitis is ongoing, and animal studies have confirmed reduction of corneal symptoms. Routine HSV testing of donor corneas has also been proposed, with expected improvement in graft survival through detection, treatment, and prevention; however, it has not yet become widespread due to cost and legal constraints7).