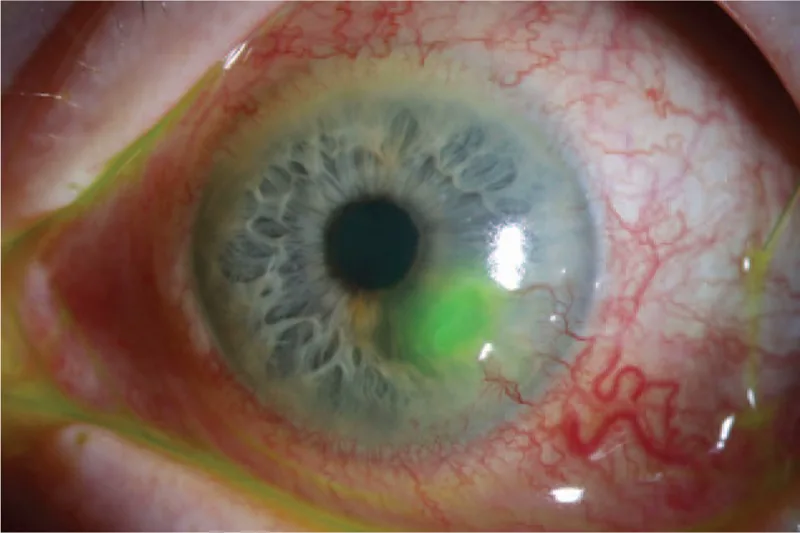
Descemetocele
Descemet’s folds: Folds of DM are observed at the base of the ulcer.
Central clear zone: A transparent area exists in the center of the thinning region.
Anterior bulging: Descemet’s membrane may protrude in a cyst-like manner1).

A descemetocele is a condition in which an intact Descemet membrane (DM) herniates forward through a defect in the corneal stroma and epithelium1). DM is a transparent, elastic, acellular membrane 8–10 μm thick, secreted by endothelial cells1). It is relatively resistant to proteolysis and biomechanical stress, and protects the endothelium from the destructive process of the stroma1).
Descemetoceles are classified by location as follows1):
Classification by size is based on the maximum diameter: small (<3 mm), medium (3–6 mm), and large (>6 mm)1).
When a corneal ulcer extends deep into the stroma and beyond Descemet’s membrane (DM), it leads to corneal perforation. Aqueous humor leaks and the anterior chamber collapses. Causes include infectious, non-infectious, and traumatic factors.
Conservative or surgical treatment is selected based on the size, location, duration, and condition of the cornea at the perforation site.
A descemetocele is a condition where Descemet’s membrane bulges forward while remaining intact, representing a “pre-perforation” state. Corneal perforation occurs when Descemet’s membrane also ruptures, leading to aqueous humor leakage. A descemetocele carries a high risk of perforation and requires urgent intervention.
Descemetocele
Descemet’s folds: Folds of DM are observed at the base of the ulcer.
Central clear zone: A transparent area exists in the center of the thinning region.
Anterior bulging: Descemet’s membrane may protrude in a cyst-like manner1).
Corneal perforation
Uveal prolapse: The iris becomes incarcerated in the defect.
Positive Seidel test: Dilution and outflow of fluorescein are observed.
Shallow anterior chamber or flat anterior chamber: The anterior chamber disappears due to aqueous humor leakage.
Uveal prolapse or a positive Seidel test is a definitive diagnostic finding of corneal perforation. However, if uveal prolapse occludes the defect, the Seidel test may be false negative.
Delayed diagnosis can lead to enlargement of corneal damage, endophthalmitis, secondary glaucoma or cataract, and even loss of the eye.
Corneal ulcers are broadly classified into those occurring in the central area and those in the peripheral area. Central ulcers are often infectious, while peripheral ulcers are often non-infectious.
| Category | Main causative microorganisms |
|---|---|
| Bacterial | Pseudomonas aeruginosa, Staphylococcus, Streptococcus pneumoniae, Moraxella, Neisseria gonorrhoeae |
| Fungal | Fusarium, Aspergillus, Candida |
| Viral | Herpes simplex, Herpes zoster |
Contact lens (CL) wear is the greatest risk factor for microbial keratitis in the United States 7). Overnight wear (including orthokeratology) is a major infection risk 7). Gram-negative bacteria (Pseudomonas, Moraxella, Serratia) are common in CL wearers.
Pseudomonas keratitis progresses alarmingly fast. An initial small round abscess forms a ring abscess within days and perforates from the center. The cornea melts due to proteases produced by the bacteria.
Fungal keratitis caused by filamentous fungi tends to progress into the deep layers, presenting with hypopyon and corneal endothelial plaques 6). As infection spreads, severe melting may eventually lead to perforation 6).
When keratitis complicates conjunctivitis caused by gonococcus, corneal perforation occurs at a high rate, requiring caution 5).
This is an essential test for diagnosing corneal perforation. Apply a sterile fluorescein strip moistened with a small amount of sterile saline to the suspected perforation site and observe under cobalt blue light. If fluorescein is diluted and flows out, the test is positive.
Pressure on the eyeball may enlarge the perforation, so minimize pressure during the test.
This is useful for detailed evaluation of corneal structure1). Even when clinical visualization is hindered by necrotic material or mucoid discharge, it can depict true stromal thickness and DM bulge1). Serial scans allow monitoring of the healing process.
If infectious keratitis is the cause, smear microscopy and culture of lesion scrapings are essential. Ideally, specimens should be collected before antimicrobial treatment.
This is a test to check for aqueous humor leakage from the cornea using fluorescein dye. The appearance of fluorescein diluted by aqueous humor flowing out (positive finding) can be observed under cobalt blue light. It is one of the most important tests for definitive diagnosis of corneal perforation.
The choice of treatment is determined based on the size, extent, location of the perforation, degree of stromal infiltration, visual prognosis, and underlying disease. In many cases, multiple treatments are performed simultaneously or sequentially.
Tseng et al. (2024) reported successful non-surgical management for 7 years with PROSE therapy for a Descemetocele secondary to ocular GVHD, maintaining corrected visual acuity of 20/504). The incidence of corneal perforation associated with oGVHD is estimated at 1-4%4).
If an infectious corneal ulcer is the cause, infection control of the underlying disease is the top priority.
Che Ku Amran et al. (2024) reported managing a corneal perforation due to gonococcal keratoconjunctivitis with cyanoacrylate adhesive + BCL + ceftriaxone 1 g intramuscular injection, achieving infection control after 2 months5).
| Perforation Size | Recommended Treatment |
|---|---|
| <3 mm | Tissue adhesive or AMT |
| >3 mm | PKP or patch graft |
| Total cornea | Corneoscleral transplant1) |
Kusano et al. (2023) reported a case of severe microbial keratitis where the entire cornea became a descemetocele (total corneal descemetocele)1). Anterior segment OCT confirmed a corneal thickness of 37 μm, and corneoscleral transplantation was performed, successfully preserving the eyeball1). This is considered the largest descemetocele reported1).
Shekhawat et al. (2022) reported a technique using a Tenon’s capsule patch graft combined with a vascularized conjunctival flap for a paracentral corneal perforation (1 mm)3). At 4 months postoperatively, uncorrected visual acuity was 20/25 with minimal astigmatism3). The robust wound healing response from Tenon’s capsule fibroblasts and the vascular supply from the conjunctival flap promoted healing3). This procedure can be performed even in settings with limited access to corneal donor tissue3).
Kato et al. (2021) reported good results with one-bite mini-keratoplasty for a paracentral corneal perforation (1 mm) caused by a metallic foreign body, achieving postoperative corrected visual acuity of 180/200 and corneal astigmatism of 0.6 diopters2). A re-perforation at 17 months was also managed with the same technique, maintaining good visual function for over 2 years2).
For perforations less than 3 mm that are not near the limbus and have a well-formed anterior chamber, cyanoacrylate adhesive may be the first choice. Adhesive is also useful as a temporary measure before penetrating keratoplasty (PKP). For perforations larger than 3 mm or with anterior chamber loss, adhesive is difficult to manage and PKP is indicated. The treatment is determined individually based on the size, location, and underlying disease of the perforation.
Corneal perforation is an ophthalmic emergency. If left untreated, it can lead to endophthalmitis, secondary glaucoma, cataract, and blindness. However, not all cases require emergency surgery. Small perforations may be managed with bandage contact lenses or adhesives, and in infectious perforations, antibiotic treatment may be given for 24–48 hours before planning penetrating keratoplasty (PKP).
Corneal ulcer begins with epithelial defect and progresses to the stroma. When stromal melting extends deeply and Descemet’s membrane (DM) is exposed, a descemetocele forms, and further rupture of DM leads to perforation.
In experimental keratitis caused by Pseudomonas aeruginosa, descemetocele formation has been shown to directly correlate with alkaline protease, total protease, and elastase activity 1). High protease-producing strains (102, 115, 118) induced massive stromal destruction, descemetocele formation, and perforation even with few inflammatory cells in the presence of Ca²⁺ and Mg²⁺ 1). Leukocyte proteases also contribute to corneal degeneration, but their presence alone does not necessarily lead to descemetocele formation 1).
DM is resistant to proteolysis and biomechanical stress, so it remains intact for a period even when the surrounding stroma melts 1). This property gives rise to the “pre-perforation stage” known as descemetocele. However, lacking sufficient tensile strength, DM eventually herniates forward 1).
Filamentous fungi tend to progress not only on the corneal surface but also into the deeper layers 6). When hyphae break through DM and reach the posterior corneal surface, they form an endothelial plaque 6). Progression leads to severe melting and perforation.
Neisseria gonorrhoeae has the ability to adhere to and invade corneal epithelium via pili 5). Within one hour of inoculation, it is taken into vacuoles within epithelial cells, and after 24 hours, epithelial thickness is markedly reduced 5). This process progresses through epithelial, stromal, and ulcerative keratitis to perforation 5).
A technique combining Tenon’s capsule patch graft with a vascularized conjunctival flap has been reported 3). The vascular supply from the conjunctival flap resulted in faster healing (complete recovery of corneal stromal thickness at 6 weeks) compared to conventional Tenon’s capsule patch graft 3). It is expected to be useful in low-resource settings where corneal donor tissue is not available 3).
One-bite mini-keratoplasty is a simple technique for small perforations of about 1 mm, inserting a corneal graft with a single 10-0 nylon suture 2). It is reported to induce less astigmatism compared to conventional lamellar keratoplasty 2).
PROSE (Prosthetic Replacement of the Ocular Surface Ecosystem) therapy is gaining attention as an option for long-term management of descemetocele in patients at high risk for surgery 4). The design bridges over the corneal dome, protecting the cornea and providing continuous lubrication and oxygen supply 4). In corneal ectasia, the PROSE group has been reported to be superior to the corneal transplant group in mean visual acuity and speed of visual recovery 4).