Non-pathological Myopia
Fundus findings: Mild myopic crescent (atrophic crescent around the optic disc) is observed. Temporal conus and tigroid fundus are characteristic.
Axial length: Often less than 26.5 mm.
Corrected visual acuity: Remains good.
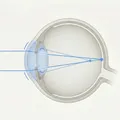
Myopia is a refractive error in which parallel light rays from infinity focus in front of the retina. The refractive power of the eye is excessive relative to the axial length, characterized by reduced distance vision.
Pathological myopia (malignant myopia, degenerative myopia, high myopia) is myopia that causes degeneration in the posterior pole of the fundus, and refers to an axial length that is three or more standard deviations from the mean of the normal distribution curve of emmetropic eyes.
Benign myopia (simple myopia, school myopia) is myopia contrasted with pathological myopia, without organic abnormalities of ocular tissues, and is mostly mild to moderate. School myopia is thought to be related to near work and develops during school age or adolescence.
The definition of high myopia is not fixed, but it often refers to strong myopia of approximately −6D or more. Among these, those accompanied by posterior pole fundus lesions are specifically called pathological myopia.
Severity Classification (Shoji Classification)
| Classification | Spherical Equivalent |
|---|---|
| Mild | −3D or less |
| Moderate | Greater than −3D and up to −6D |
| High | Greater than −6D and up to −10D |
| Very High | Greater than −10D |
Age-Specific Diagnostic Criteria for Pathological Myopia
| Age | Refractive error | Corrected visual acuity |
|---|---|---|
| 5 years or younger | Greater than −4.0 D | 0.4 or less |
| 6–8 years | Greater than −6.0 D | 0.6 or less |
| 9 years or older | Greater than −8.0 D | 0.6 or less |
Classification by etiology
Causes of pseudomyopia include central accommodative spasm due to encephalitis, brain tumor, head trauma, etc.; administration of miotics, acetazolamide, sulfa drugs, steroids, and organophosphates; and excessive accommodation due to intermittent exotropia.
More than one-fifth of the world’s population has myopia, and it is predicted to reach half by 2050 1). In a systematic prediction study by Holden et al. (2016), the global myopic population was estimated to increase from 1.3 billion to 4.9 billion, and high myopia from 160 million to 940 million between 2000 and 2050 12). Productivity loss is estimated at $250 billion annually, and loss due to myopic macular degeneration at $6 billion 1). In some parts of Asia, 80–90% of children have myopia, posing a major public health concern.
There are racial differences in the development of high myopia, particularly common in Asians. Pathological myopia exceeding −8 D accounts for about 1% of the general population and about 5% of all myopia.
According to Japanese school statistics (Ministry of Education, Culture, Sports, Science and Technology), the proportion of high school students with uncorrected visual acuity less than 1.0 is about 63% (FY2014), junior high school students about 53%, and elementary school students about 30%. Recent surveys on myopia by the Ministry suggest that myopia is becoming more severe in younger students, raising concerns about future risks of visual complications 2).
The Hisayama Study confirmed an increasing prevalence of myopic maculopathy in adults, showing that longer axial length is an independent risk factor for its development 14). The 5-year cumulative incidence of myopic maculopathy in high myopia far exceeds that in the general population, providing epidemiological evidence supporting the medical significance of myopia progression control.
More than one-fifth of the world’s population has myopia, and it is predicted that about half will be myopic by 2050 1). The prevalence is particularly high in Asia, with about 80% of children in Taiwan affected. In Japan, about 63% of high school students have uncorrected visual acuity less than 1.0, and in recent years, there are concerns about increasing severity at younger ages 2).
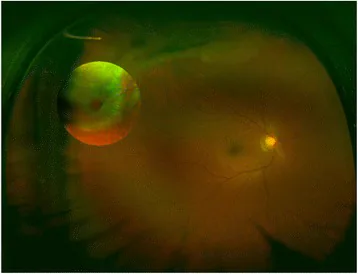
Non-pathological Myopia
Fundus findings: Mild myopic crescent (atrophic crescent around the optic disc) is observed. Temporal conus and tigroid fundus are characteristic.
Axial length: Often less than 26.5 mm.
Corrected visual acuity: Remains good.
Pathologic Myopia
Posterior staphyloma: Localized outward bulging of the posterior pole of the eye. The sclera expands and protrudes posteriorly.
Myopic macular degeneration: Macular lesions including Fuchs spot, choroidal neovascularization (CNV), retinal hemorrhage, and atrophy. Linear yellowish lesions due to Bruch membrane rupture (lacquer crack lesions) are also characteristic.
Myopic retinoschisis (MRS): Found in 9–34% of pathologic myopic eyes with posterior staphyloma 4).
Peripheral changes: White without pressure, lattice degeneration, and holes.
The main complications of high myopia are as follows:
The estimated increase in disease risk per additional diopter is 58% for myopic maculopathy, 20% for open-angle glaucoma, 21% for posterior subcapsular cataract, and 30% for retinal detachment 5). In a meta-analysis by Haarman et al., even mild myopia (−1D to −3D) was associated with a 3-fold increased risk of retinal detachment and a 9-fold increased risk of myopic maculopathy compared to non-myopia 13). This quantification of complication risks provides the medical rationale for interventions to control myopia progression.
In pathologic myopia, axial elongation leads to fundus changes such as posterior staphyloma, Fuchs spot, choroidal neovascularization, lacquer crack lesions, retinal tears and detachment, and optic disc tilt. Myopic retinoschisis (MRS) is found in 9–34% of pathologic myopic eyes with posterior staphyloma and may be an indication for vitrectomy 4).
The etiology of myopia is multifactorial, involving a complex interplay of genetic and environmental factors.
Myopia of Prematurity (MOP) is a distinct disease entity with a different pathogenesis from ordinary myopia. It is primarily caused by anterior segment developmental abnormalities such as increased corneal curvature, lens thickening, and shallow anterior chamber, rather than axial elongation. The severity of retinopathy of prematurity (ROP) and the type of treatment (cryocoagulation > laser photocoagulation > anti-VEGF therapy, in order of increasing myopia risk) greatly affect refractive outcomes.
Other associated diseases: congenital glaucoma, retinopathy of prematurity, retinitis pigmentosa, cataract, congenital stationary night blindness, keratoconus, Stickler syndrome, Marfan syndrome, Weill-Marchesani syndrome.
Reports indicate that increased outdoor activity can reduce the onset of myopia by up to 50%1). It is thought that high-intensity outdoor light stimulates retinal dopamine release, suppressing axial elongation. This is one of the simplest and most side-effect-free interventions for slowing myopia progression, with a 50% reduction effect shown with an increase of 76 minutes per day1).
The diagnosis of high myopia itself is made through refraction testing and axial length measurement. Since the pathology of high myopia is essentially an abnormal elongation of the eye axis, axial length measurement is essential.
School health checkups and vision tests in pediatric clinics are the first opportunities for detection. Detection is possible with photoscreening or autorefractometers, but these are insufficient for determining quantitative refractive error.
| Examination Method | Content/Purpose |
|---|---|
| Cycloplegic Refraction | Gold standard for children. Instill cyclopentolate twice at 10-minute intervals, then perform autorefraction 45–60 minutes after the first instillation2) |
| Axial Length Measurement | Measured with an optical biometer. Important for monitoring myopia progression. |
| Dilated fundus examination | Essential for confirming posterior staphyloma, Fuchs spot, and retinal tears |
| OCT | Useful for early detection of macular retinoschisis and choroidal neovascularization |
| Fluorescein angiography | Useful for differentiating CNV from simple macular hemorrhage |
| Visual field test | Used to evaluate myopic optic neuropathy |
Cycloplegic refraction is the gold standard for children. Without eliminating accommodation, excessive minus correction is likely in children with strong accommodative ability. For infants and young children, 1% cyclopentolate (Cyplegin®) eye drops are the first choice, and management using axial length percentile curves is also useful for monitoring myopia progression 2).
Key points for differential diagnosis: Always perform cycloplegic refraction to differentiate from pseudomyopia (accommodative spasm). If there are findings suggesting rapidly progressing myopia or systemic diseases (such as Stickler syndrome or Marfan syndrome), conduct a systemic evaluation.
When evaluating the indication for low-concentration atropine eye drops, progression of refractive error >0.5D or axial length elongation >0.3mm over 6 months is considered a criterion for progression.
There are two approaches for monitoring myopia progression 2):
In myopia progression management, visually demonstrating treatment effects to patients and parents using these monitoring tools is effective for maintaining motivation to continue treatment2).
Myopia treatment is broadly divided into three categories: ① securing visual acuity through refractive correction, ② suppressing myopia progression, and ③ treating complications of high myopia.
The comparative effects of each intervention for suppressing myopia progression are shown below.
| Intervention | Refractive suppression effect | Axial length suppression effect |
|---|---|---|
| Low-concentration atropine 0.05% | Up to 67%1) | — |
| Orthokeratology | — | 32–59%1) |
| MiSight 1 day (+2.00 D add) | 59%6) | 52%6) |
| DIMS spectacle lens (MiYOSMART®) | 55–59%3) | — |
Low-concentration atropine eye drops are the most evidence-based pharmacotherapy for myopia progression suppression1). Atropine is a reversible antagonist of muscarinic receptors, and it is thought to be involved in scleral remodeling via muscarinic receptors (mainly M1/M4 receptors) present in the retina and sclera, thereby suppressing axial elongation; however, the detailed mechanism remains unclear2).
Comparison of characteristics by concentration (LAMP study)
| Concentration | Myopia progression suppression rate | Side effects |
|---|---|---|
| 0.01% | Approximately 49%1) | Minimal |
| 0.025% | Approximately 62% (moderate) 2) | Mild |
| 0.05% | Up to 67% 1) | Slightly increased photophobia and blurred vision |
Treatment is recommended for children whose parents are myopic, who spend little time outdoors, or who developed myopia at a young age (earlier onset increases the risk of higher future myopia) 2). After starting treatment, the first follow-up is performed within 1 week to 1 month, followed by regular check-ups every 3 to 6 months. It is desirable to continue until the late teens when myopia progression stabilizes. Since rebound may occur after discontinuation, follow-up should continue after stopping 2).
A method in which special rigid lenses are worn during sleep to temporarily flatten the central cornea. Thickening of the mid-peripheral cornea creates peripheral myopic defocus, suppressing axial elongation.
CLs designed to reduce peripheral hyperopic defocus and suppress axial elongation. The AAO Ophthalmic Technology Assessment confirms that 12 RCTs, including 11 Level 1 trials, show significant suppression of myopia progression and axial elongation 6).
Special spectacle lenses with peripheral defocus control design (multi-segment lenses) were positioned as one of the standard treatments in the 2025 guideline (1st edition) by the Japanese Society of Myopia. 3)
The discontinuation criteria for myopia management spectacle lenses are based on stabilization of myopia progression around age 18 ± 2 years. If no changes in refractive error or axial length are observed in two consecutive follow-up visits every 6 months, discontinuation of lens wear may be considered 3). No rebound has been observed after discontinuation.
In the LAMP study, 0.05% was most effective, showing up to 67% progression suppression 1). In Japan, 0.025% Rijusea® Mini ophthalmic solution was approved in December 2024 2). 0.01% may have limited efficacy. The choice of optimal concentration should be individualized based on the balance between efficacy and side effects (photophobia, near vision impairment).
In a Japanese multicenter study (1,438 patients), the incidence of MK was 5.4 per 10,000 patient-years 1). With proper care, it is a relatively safe treatment. However, rinsing lenses with tap water is strictly prohibited as it increases the risk of Acanthamoeba keratitis.
Intravitreal injection of aflibercept or ranibizumab is the first-line treatment. Photodynamic therapy (PDT) and triamcinolone acetonide (off-label) are also effective but inferior to anti-VEGF antibodies.
In the natural course, most patients progress to a corrected visual acuity of 0.1 or less after 5 years, and the prognosis is poor. In patients with only diffuse chorioretinal atrophy without CNV or myopic traction maculopathy, visual acuity is often relatively well preserved.
Cases of MRS progression after intravitreal aflibercept injection (IVA) for myopic choroidal neovascularization associated with pathologic myopia have been reported 4), and attention should be paid to worsening of MRS after anti-VEGF injection.
Vitrectomy (PPV + ILM peeling) to relieve traction is the standard treatment.
Myopic foveoschisis is essentially a progressive disease; although rare cases of spontaneous resolution exist, some cases progress rapidly.
Vitrectomy and gas tamponade are the standard treatment. For refractory cases, macular buckling or scleral shortening is performed. With vitrectomy, final reattachment is achieved in almost all cases, but visual prognosis depends on the condition of the macula before and after surgery.
Intraocular pressure reduction (similar to treatment for glaucomatous optic neuropathy) is performed.
In cataract surgery for highly myopic eyes, reduced accuracy of IOL power calculation is a problem. AI-driven new-generation formulas (Kane, Hill-RBF) have been reported to have significantly lower MAE (0.51D and 0.52D, respectively) compared to the SRK/T formula in cases with axial length ≥30 mm, with refractive errors exceeding ±1.0D occurring in only 7.5% of cases versus 42.5% for SRK/T. Similar issues arise in cases after refractive surgery such as LASIK, so it is important to follow the indications, preoperative evaluation, and postoperative management procedures specified in the guidelines (8th edition) of the Japanese Society of Refractive Surgery 7).
The main pathology of high myopia is axial elongation. An increase of 1 mm in axial length corresponds to approximately 3 diopters of myopia. The detailed mechanism of axial elongation is not fully understood, but studies using experimental myopia models have shown that changes in growth factor expression in the sclera are involved in axial elongation.
Axial elongation is thought to be regulated by optical signals from the retina.
When axial elongation progresses severely, mechanical stretching occurs in the choroid, retina, and sclera.
RLRL therapy using 650 nm red light is a novel intervention that suppresses axial elongation by increasing choroidal thickness 1). In a multicenter RCT by Jiang et al. (2022), the RLRL group showed significant suppression of axial elongation at 0.10 mm/year (control group 0.38 mm/year) 11). A multicenter RCT by Zeng et al. (2023) suppressed spherical equivalent progression by approximately 72%. However, major studies are biased toward East Asia, and long-term safety data are still limited. As of April 2026, this therapy is not approved by Japanese pharmaceutical authorities and is not covered by insurance, being offered as self-pay treatment.
New optical design spectacle lenses such as PLARI, NLARI, and CARE are being developed and investigated 1).
In cataract surgery for high myopia, further improvement in the accuracy of IOL calculation formulas using AI technology is expected. Stable refractive outcomes may be achievable even in extreme axial myopia with axial length of 30 mm or more.
Correlations between saturated fat and cholesterol intake and axial length have been reported, and the possibility of myopia suppression through nutritional intervention is being investigated. 1)