
Developmental Glaucoma (Congenital Glaucoma)
Key Points at a Glance
Section titled “Key Points at a Glance”1. What is Developmental Glaucoma?
Section titled “1. What is Developmental Glaucoma?”Developmental glaucoma is glaucoma caused by abnormal development of the aqueous humor outflow pathway. It is classified into early-onset and late-onset types based on the age of onset 1).
The early-onset type corresponds to primary congenital glaucoma (PCG). The cause of elevated intraocular pressure is limited to trabecular meshwork developmental abnormalities, occurring before 3–4 years of age and accompanied by corneal enlargement (buphthalmos). The late-onset type has milder angle/trabecular meshwork developmental abnormalities, leading to later onset and a course similar to primary open-angle glaucoma. It typically occurs in the teens to twenties.
Classification Systems
Section titled “Classification Systems”Multiple classification systems exist for childhood glaucoma. The Japanese Glaucoma Guideline classifies developmental glaucoma into three types: early-onset, late-onset, and developmental glaucoma with other congenital anomalies 1). Internationally, the classification developed by the Childhood Glaucoma Research Network (CGRN) has been adopted by the World Glaucoma Association (WGA) and the American Board of Ophthalmology (ABO) 2).
| Classification | Japanese Classification | CGRN Classification |
|---|---|---|
| Primary, Early-onset | Early-onset developmental glaucoma | Primary congenital glaucoma (PCG) |
| Primary, Late-onset | Late-onset developmental glaucoma | Juvenile open-angle glaucoma (JOAG) |
| Associated with congenital anomalies | Developmental glaucoma with other congenital anomalies | Secondary: associated with congenital ocular malformations |
| Associated with systemic diseases | (Included above) | Secondary: associated with congenital systemic diseases |
| Acquired factors | (Included above) | Secondary: acquired factors |
| Postoperative | (Included above) | Secondary: post-cataract surgery |
PCG is further subdivided by age of onset into neonatal (0–1 month), infantile (1–24 months), and late-onset (≥2 years) 2). JOAG develops after age 4, does not involve globe enlargement, and presents with a normal-appearing angle 2).
Secondary childhood glaucoma includes glaucoma associated with congenital ocular malformations such as Axenfeld-Rieger anomaly, Peters anomaly, and aniridia; glaucoma associated with congenital systemic diseases such as Sturge-Weber syndrome and Down syndrome; acquired glaucoma secondary to uveitis, trauma, or steroids; and post-cataract surgery glaucoma 2).
Epidemiology
Section titled “Epidemiology”Childhood glaucoma accounts for 5% of causes of blindness in children, affecting more than 300,000 people worldwide 2).
The incidence of early-onset type is 1.8–2.4 per 100,000 live births in Japan, while in Europe and the United States it is reported as 5–10 per 100,000. The highest frequency is found among the Roma people in Slovakia, where consanguineous marriage is common, with an incidence of 80 per 100,000 live births. 75% of cases are bilateral, and 65% occur in boys. 80% of cases develop within the first year of life. Most cases are sporadic, but about 10–40% show heritability, following an autosomal recessive inheritance pattern.
In a large Australasian disease registry (ANZRAG: 660 patients), PCG accounted for 57.6% and JOAG for 19.3% of childhood glaucoma cases 3). The median age at diagnosis was 0.25 years for PCG and 14 years for JOAG 3).
According to the glaucoma clinical practice guidelines, it is classified into three types: early-onset, late-onset, and developmental glaucoma with other congenital anomalies 1). Internationally, the CGRN classification is standard, dividing into primary congenital glaucoma (PCG), juvenile open-angle glaucoma (JOAG), and four types of secondary glaucoma (congenital ocular anomalies, congenital systemic diseases, acquired factors, and post-cataract surgery) 2). PCG is the most common type, accounting for about 60% of childhood glaucoma.
2. Main Symptoms and Clinical Findings
Section titled “2. Main Symptoms and Clinical Findings”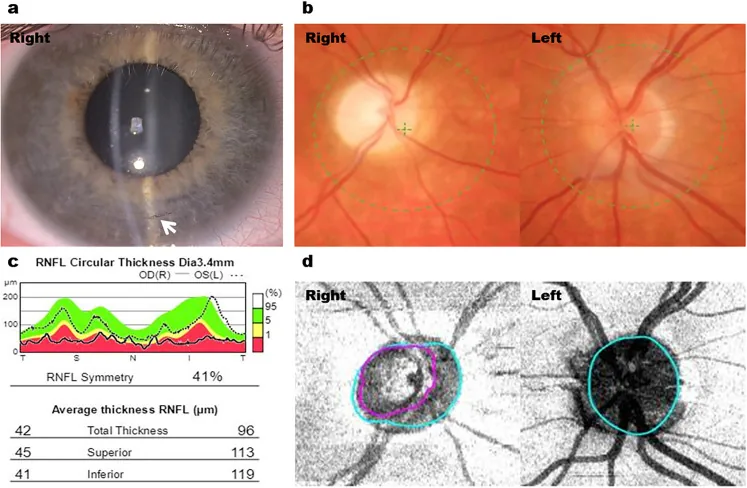
Pediatric Application of MIGS Procedures
Section titled “Pediatric Application of MIGS Procedures”Minimally invasive glaucoma surgery (MIGS) is a conjunctiva-sparing technique, and its application in children is advancing due to the advantage of preserving options for future additional surgeries. However, evidence is limited.
- KDB (Kahook Dual-Blade) goniotomy: Application in pediatric PCG has been reported 7)
- Trab360: Reported as a trabeculotomy-type procedure for childhood glaucoma 8)
- PreserFlo external microshunt: Small-scale reports exist as an adjunctive option for refractory childhood glaucoma 9)
Pharmacotherapy
Section titled “Pharmacotherapy”The choice of medications is similar to that for adult open-angle glaucoma, but there are special considerations for children.
- Prostaglandin (PG) analogs: Many children are non-responders, but they are advantageous in terms of safety and once-daily dosing.
- Beta-blockers: Use the lowest possible concentration. Be cautious of side effects such as bronchial asthma and bradycardia. Apnea has been reported in neonates.
- Carbonic anhydrase inhibitors (CAIs): Oral acetazolamide 5 mg/kg/day divided into three doses. They have a strong intraocular pressure-lowering effect, but long-term use may cause metabolic acidosis and growth retardation. Eye drops have fewer side effects but are less effective.
- Postoperative pilocarpine: Use 1–2% two to three times daily for several weeks. The goal is to prevent peripheral anterior synechiae.
Treatment of late-onset developmental glaucoma
Section titled “Treatment of late-onset developmental glaucoma”For school-age children and older, medication therapy should be attempted first. Since angle abnormalities are milder compared to early-onset type, drug therapy is more effective.
- First-line drugs: PG analogs (e.g., latanoprost)
- Second-line drugs: CAI eye drops or beta-blockers (only in children who can accurately describe subjective side effects)
- If intraocular pressure exceeds 25 mmHg: Start with PG analogs and add beta-blockers or CAIs as needed.
Surgical indications should be carefully considered. Due to large temporal fluctuations in intraocular pressure, surgery is considered when high pressure of 30–40 mmHg or more persists for several weeks with poor response to medication, or when visual field damage is advanced even with pressure around 20 mmHg1).
Target intraocular pressure and long-term management
Section titled “Target intraocular pressure and long-term management”The target intraocular pressure is preferably 19–20 mmHg or lower.
Long-term management is necessary even after intraocular pressure control. To support visual development, regular refraction and visual acuity tests should be performed, and amblyopia treatment or strabismus treatment should be administered as needed. The main cause of poor vision is amblyopia, and early appropriate refractive correction and vision training are important.
For early-onset developmental glaucoma (primary congenital glaucoma), surgery is the first choice, and goniotomy or trabeculotomy is performed 1). The success rate is 65–96%, and it is best in cases with onset between 1 and 24 months of age. Medications are used to reduce corneal edema before surgery or as an adjunct after surgery. In late-onset types, medication is first attempted after school age. In any type, early intervention and long-term amblyopia management are essential for maintaining visual function.
6. Pathophysiology and Detailed Mechanisms
Section titled “6. Pathophysiology and Detailed Mechanisms”Histological Findings of Angle Dysgenesis
Section titled “Histological Findings of Angle Dysgenesis”The pathology of early-onset developmental glaucoma is summarized as follows:
- Abnormal thickening of the juxtacanalicular connective tissue: Because the trabecular meshwork is underdeveloped, the juxtacanalicular connective tissue is abnormally thick.
- Accumulation of extracellular matrix: A non-lamellar compact tissue composed of collagen, elastin-like fibers, and abundant amorphous material exists beneath Schlemm’s canal. It consists of trabecular cells with short cell processes, fibrous components, and basement membrane-like material.
- Abnormal attachment of the ciliary body: The ciliary body attaches to the trabecular meshwork area, and muscle contraction pulls the scleral spur forward, compressing Schlemm’s canal and the trabecular meshwork.
- Ectopic iris root: The iris root is located at the position of the trabecular meshwork.
- Underdevelopment of Schlemm’s canal: In some cases, Schlemm’s canal is absent or incompletely developed.
Embryologically, trabecular cells are derived from the neural crest, whereas the juxtacanalicular connective tissue is derived from vascular endothelial cells. The greatest resistance to aqueous outflow exists at the junction of tissues of different origins.
It is speculated that a trabecular sheet (sheet-like material) is compressed and exists in front of the trabecular meshwork, obstructing aqueous outflow, and also lifts the iris to prevent angle widening, causing the characteristic angle findings in developmental glaucoma.
Mechanism of Ocular Enlargement
Section titled “Mechanism of Ocular Enlargement”In infants, the ocular coat is highly elastic. When high intraocular pressure persists, the ocular coat stretches around the corneoscleral limbus, increasing the corneal diameter (buphthalmos). At the same time, ruptures (Haab striae) occur in Descemet’s membrane, which is less elastic than the corneal stroma, and aqueous humor flows into the corneal stroma and epithelium, rapidly worsening corneal edema and opacity.
If poor intraocular pressure control persists, there is also a risk of lens dislocation due to rupture of the zonules of Zinn, corneal erosion/ulceration, and globe rupture.
Why angle surgery is effective in children
Section titled “Why angle surgery is effective in children”Trabeculotomy is considered highly effective for congenital glaucoma 5). This difference is thought to be due to the abundant elastic fibers in the pediatric angle. Elastic fibers promote opening of the angle structures and maintain postoperative patency of the aqueous outflow pathway 5).
After external trabeculotomy, the patency of Schlemm’s canal is considered important for long-term outcomes 5). The main causes of postoperative failure are fibrous proliferation of the inner wall of Schlemm’s canal and elongation of the Schlemm’s canal endothelium 5).
Genetic abnormalities and angle development
Section titled “Genetic abnormalities and angle development”CYP1B1 is a gene encoding an enzyme belonging to the cytochrome P450 family, and is highly expressed in fetal ocular anterior segment tissues. In cases with CYP1B1 mutations, abnormalities in the tyrosine metabolic pathway are thought to impair normal differentiation and maturation of angle tissues. CYP1B1 mutations have been reported not only in PCG but also in JOAG and glaucoma associated with congenital ocular malformations 3).
Recently, TEK/ANGPT1 mutations have been newly identified 2). TEK encodes a receptor tyrosine kinase involved in the development and maintenance of Schlemm’s canal endothelium, suggesting the involvement of a new molecular pathway in PCG.
7. Latest research and future perspectives
Section titled “7. Latest research and future perspectives”Pediatric application of new MIGS devices
Section titled “Pediatric application of new MIGS devices”The PreserFlo external microshunt is a device that shunts aqueous humor from the anterior chamber to the subconjunctival space. Its use in refractory pediatric glaucoma is limited to small reports, and its position as standard treatment requires future validation 9).
The OMNI surgical system combines TM incision with injection of viscoelastic material into Schlemm’s canal and collector channels (viscocanalostomy), and has been reported for use in a 4-month-old infant with glaucoma associated with Sturge-Weber syndrome.
A paradigm shift from external to internal procedures is underway 5). Internal procedures do not require creation of a conjunctival or scleral flap and preserve the conjunctiva, making them useful as a strategy to leave options for future additional surgeries.
Advances in genetic research
Section titled “Advances in genetic research”TEK/ANGPT1 mutations have been identified as a novel molecular pathway in PCG, advancing the understanding of mechanisms involved in Schlemm’s canal development and maintenance 2). Research on the CYP1B1 gene has attracted the highest academic interest in the field of pediatric glaucoma, and in a bibliometric analysis from 1955 to 2022, molecular genetics accounted for 13 of 25 major research themes 6).
Although the current age-based classification is useful for predicting prognosis, it has been pointed out that it does not consider genetic background 2). With the spread of genetic testing, the construction of a new classification system based on the correspondence between phenotype and genotype is expected.
Trends in QOL Research
Section titled “Trends in QOL Research”In a systematic review by Stingl et al. (2024), 10 patient-reported outcome measures (PROMs) used in pediatric glaucoma cohorts were identified. Seven achieved the highest quality rating of 5/7, but none included the perspective of pediatric glaucoma patients during development 4). The development of PROMs specific to pediatric glaucoma is needed.
The QOL of pediatric glaucoma is influenced not only by visual impairment but also by multifaceted factors such as repeated surgeries, continuous eye drop treatment, anxiety about heredity, and impact on future career choices 4).
8. References
Section titled “8. References”- 日本緑内障学会. 緑内障診療ガイドライン(第5版). 日眼会誌. 2022;126(2):85-177.
- European Glaucoma Society. Terminology and Guidelines for Glaucoma, 5th Edition. PubliComm; 2020.
- Knight LSW, et al. Childhood and Early Onset Glaucoma Classification and Genetic Profile in a Large Australasian Disease Registry. Ophthalmology. 2021;128:1549-1560.
- Stingl JV, et al. Systematic Review of Instruments for the Assessment of Patient-Reported Outcomes and Quality of Life in Patients with Childhood Glaucoma. Ophthalmol Glaucoma. 2024;7:391-400.
- Chihara E, Hamanaka T. Historical and Contemporary Debates in Schlemm’s Canal-Based MIGS. J Clin Med. 2024;13(16):4882.
- Jain D, Dhua S. Bibliometric analysis of pediatric glaucoma. Indian J Ophthalmol. 2023;71(5):2152-2157.
- Elhilali HM, et al. Kahook Dual Blade goniotomy vs conventional goniotomy in primary congenital glaucoma. Am J Ophthalmol. 2020;219:150-158.
- Areaux RG Jr, et al. Trab360 suture trabeculotomy in childhood glaucoma. J AAPOS. 2019;23(6):e52.
- Burgos-Blasco B, et al. PreserFlo microshunt in refractory pediatric glaucoma. J Glaucoma. 2022;31(12):983-988.
