Port-Wine Stain Distribution and Risk
V1 area only: Glaucoma risk 6.7%
V2 area only: Glaucoma risk almost none
V1 + V2 areas: Glaucoma risk significantly increased to 31.8%
V1 + V2 + V3 areas: Neurological symptom risk increases 4-fold

Sturge-Weber syndrome (SWS), also called encephalotrigeminal angiomatosis, is a congenital neurocutaneous syndrome (phakomatosis). Its three main features are facial port-wine hemangioma (nevus flammeus) in the trigeminal nerve distribution, ipsilateral leptomeningeal hemangioma, and ocular hemangioma. In 1879, Sturge reported a case of facial hemangioma with buphthalmos, hemiplegia, and epilepsy, and in 1929, Weber established it as a syndrome.
The cause is a somatic mosaic mutation (post-zygotic mutation) of the GNAQ gene, which is not hereditary and mostly occurs sporadically 4)8). It is thought to be due to abnormal vascular development caused by sympathetic nerve dysfunction during the fetal period. The incidence is rare, occurring in 1 in 50,000 births, with no racial or gender predilection 5)7).
SWS is classified into the following three types based on clinical features.
| Classification (Roach) | Features |
|---|---|
| Type I (Classic) | PWS + neurological symptoms + glaucoma |
| Type II | PWS + glaucoma (no neurological involvement) |
| Type III (Rarest) | Meningeal angioma only |
Glaucoma is the most important ocular complication in SWS, and it has the highest incidence of glaucoma among phakomatoses 2). When the eyelid is involved with hemangioma, glaucoma develops at a high frequency of 30–70%. Choroidal hemangioma occurs in about 40% of patients.
Based on the timing of onset, glaucoma is broadly divided into early-onset and late-onset types. About 60% are early-onset, occurring from immediately after birth to 4 years of age, with abnormal angle development as the main cause. The remaining 40% are late-onset, occurring after infancy, and are associated with elevated episcleral venous pressure and choroidal hemangioma. Onset is often before 10 years of age.
Sturge-Weber syndrome is a congenital neurocutaneous disorder characterized by the triad of facial port-wine stain in the trigeminal nerve distribution, ipsilateral leptomeningeal angioma, and ocular angioma. It is caused by a somatic mosaic mutation in the GNAQ gene, is not hereditary, and most cases are sporadic 4)8). Neurological symptoms such as epilepsy (75–90% develop by age 3), intellectual disability, and hemiparesis, along with glaucoma (30–70%), are major complications. There are also type II (glaucoma only) and type III (meningeal angioma only) without the full triad. See the “Causes and Risk Factors” section for details.
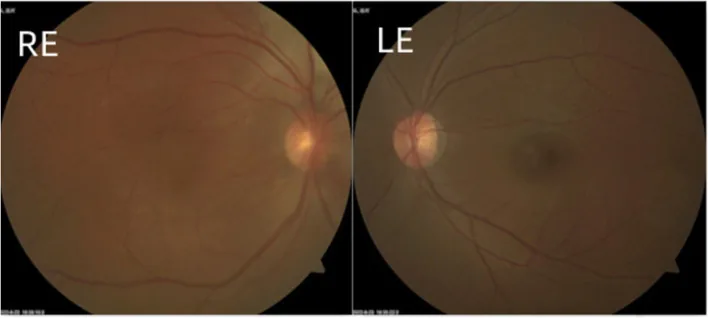
In early-onset type, lacrimation, photophobia, and blepharospasm are initial symptoms. Visual impairment occurs with increased corneal diameter and corneal opacity (buphthalmos). Haab striae, which are breaks in Descemet’s membrane, may be observed.
Late-onset type resembles primary open-angle glaucoma clinically, with few subjective symptoms in the early stage. As it progresses, visual field loss and decreased visual acuity occur.
SWS is caused by a somatic mosaic mutation in the GNAQ gene (chromosome 9q21.2, c.548G→A, p.Arg183Gln)8). This mutation leads to constitutive activation of the Gαq signaling pathway, resulting in uncontrolled proliferation of endothelial cells and vascular malformations4). Since it is a somatic mosaic mutation rather than a germline mutation, it is not hereditary. Molecular diagnosis requires biopsy of affected tissue (usually skin)4).
Detection of GNAQ mutations is influenced by the affected tissue sample and analysis method, so molecular diagnosis should be interpreted in conjunction with clinical findings.
Port-Wine Stain Distribution and Risk
V1 area only: Glaucoma risk 6.7%
V2 area only: Glaucoma risk almost none
V1 + V2 areas: Glaucoma risk significantly increased to 31.8%
V1 + V2 + V3 areas: Neurological symptom risk increases 4-fold
Differences in Onset Timing and Etiology
Early-onset type (approximately 60%): Mainly due to abnormal development of the angle. Presents with buphthalmos and increased corneal diameter 1)
Late-onset type (approximately 40%): Mainly due to increased episcleral venous pressure and involvement of choroidal hemangioma 1)
Eyelid involvement: When the hemangioma extends to the eyelid, the incidence of glaucoma significantly increases
Bilateral PWS: More likely to be associated with SWS compared to unilateral PWS 7)
Diagnosis of glaucoma associated with SWS requires precise intraocular pressure measurement and examination of the anterior segment, angle, and fundus. In children, examination under general anesthesia is often necessary.
Fluorescein angiography is useful. Early phase shows large choroidal vascular patterns, and late phase shows hyperfluorescence of the entire tumor area. Diffuse choroidal hemangioma may be difficult to identify on routine fundus examination.
Head CT detects calcifications in the cerebral cortex. Even in neonates without calcifications, gadolinium-enhanced MRI can detect leptomeningeal angioma. SPECT for cerebral blood flow evaluation is also used as an adjunct.
SWS is one of the phakomatoses and a representative disease causing choroidal hemangioma. Differentiation from other phakomatoses is important.
| Disease | Characteristic ocular findings | Location of hemangioma/tumor |
|---|---|---|
| SWS | Choroidal hemangioma, glaucoma | Face + leptomeninges |
| von Hippel-Lindau disease | Retinal hemangioma (temporal periphery) | Retina, cerebellum, kidney |
| Neurofibromatosis type 1 | Iris Lisch nodules | Cutaneous neurofibroma |
| Tuberous sclerosis | Retinal hamartoma | Brain, skin, kidney, heart |
Klippel-Trenaunay-Weber syndrome also presents with cutaneous hemangiomas similar to SWS, but is distinguished by venous malformations of the limbs and hypertrophy of bone and soft tissue.
In patients with facial port-wine stains, regular ophthalmic examinations including intraocular pressure measurement are essential. In children, intraocular pressure measurement using a rebound tonometer, anterior segment examination (corneal diameter, corneal opacity, presence of Haab striae), gonioscopy (assessment of angle abnormalities), and fundus examination (evaluation of optic disc cupping, detection of choroidal hemangioma) are performed. Examination under general anesthesia is often necessary in children. In late-onset cases, the presence of blood in the angle or dilation of episcleral vessels is useful for differential diagnosis. Fluorescein angiography is essential for evaluating choroidal hemangioma, and head CT and contrast-enhanced MRI are performed for systemic evaluation.
Treatment of glaucoma associated with SWS requires different strategies depending on the age of onset and pathogenic mechanism.
Surgical treatment is necessary for congenital or infantile-onset glaucoma 1). Trabeculotomy or goniotomy is the first-line treatment 1). However, the success rate is lower compared to primary congenital glaucoma, and additional surgery is often required.
Trabeculectomy carries a risk of severe suprachoroidal hemorrhage or expulsive hemorrhage due to bleeding from the hemangioma. In general, the response to glaucoma surgery is poor, and trabeculectomy or tube shunt surgery is often required.
In older patients, because episcleral venous pressure is elevated, medical treatment is the first choice 1). Aqueous humor suppressants (beta-blockers, carbonic anhydrase inhibitors) are considered most effective. The intraocular pressure-lowering effect of prostaglandin analogs has been reported to be inconsistent.
When drug therapy or outflow reconstruction surgery is ineffective, trabeculectomy or tube shunt surgery may be considered1).
Filtration Surgery / Tube Shunt
Trabeculectomy: Outcomes may improve with the use of antimetabolites (mitomycin C). However, in SWS eyes, the risk of choroidal effusion and expulsive hemorrhage is very high1).
Ahmed device: Reported cumulative success rates of 79% at 24 months and 30% at 60 months.
Two-stage Baerveldt device: A report showed that all eyes achieved intraocular pressure below 21 mmHg at a mean follow-up of 35 months.
Pediatric GDD meta-analysis: Analysis of 1,221 eyes showed success rates of 87% (95% CI: 83-91%) at 12 months and 77% (95% CI: 71-83%) at 24 months9).
Management of Intraoperative and Postoperative Complications
Choroidal hemorrhage/effusion: In cases with choroidal hemangioma, rapid intraocular pressure reduction carries a high risk of choroidal detachment and hemorrhage1).
Preventive measures: Preoperative intraocular pressure reduction with hyperosmotic agents, posterior sclerotomy, pre-placed scleral flap sutures, and additional tight sutures.
Device selection: Use of valved (Ahmed) or two-stage (Baerveldt) GDD reduces the risk of hypotony.
Cyclophotocoagulation: For refractory cases, cyclophotocoagulation (CPC) may be considered. A report showed that 10 of 16 eyes (62.5%) maintained intraocular pressure between 6 and 22 mmHg without complications (mean follow-up 8.87 years).
In the two-stage technique, a capsule is formed around the plate several weeks before the tube is inserted into the anterior chamber. This prevents excessive intraocular pressure reduction immediately after surgery and minimizes the risk of choroidal effusion and hemorrhage. This method is particularly useful in cases with choroidal hemangioma, such as SWS.
There are reports that netarsudil effectively lowers intraocular pressure in glaucoma associated with SWS even when added as a fourth- or fifth-line agent. It has a mechanism that promotes aqueous humor outflow from the trabecular meshwork.
Diffuse choroidal hemangioma associated with SWS may require management in parallel with glaucoma treatment.
Although more refractory than developmental glaucoma, if good intraocular pressure control is achieved early, vision can be preserved. However, if the choroidal hemangioma enlarges and causes exudative retinal detachment, cryocoagulation may not be sufficiently effective, leading to severe visual impairment.
Because the lesions involve the cornea, lens, retina, and nerve, achieving good vision is often difficult. The life expectancy of SWS patients is shorter than that of the general population, and bilateral leptomeningeal involvement leads to more severe neurological symptoms and a poorer prognosis.
The most important consideration is surgical complications related to choroidal hemangioma. In SWS, choroidal hemangioma is present in about 40% of cases, and rapid intraocular pressure reduction during glaucoma surgery carries a risk of choroidal effusion, hemorrhage, and retinal detachment 1). Preventive measures include preoperative administration of hyperosmotic agents, performing posterior sclerotomy, tight suturing of the scleral flap, and use of valved or two-stage drainage devices. Also note that alpha-2 receptor agonists (brimonidine) are contraindicated in children under 2 years of age 1). For details, refer to the “Standard Treatment” section.
The underlying cause of SWS is a somatic mosaic mutation in the GNAQ gene (c.548G→A, p.Arg183Gln)8). This mutation constitutively activates the Gαq signaling pathway, leading to uncontrolled proliferation of endothelial cells and vascular malformations4). Because the mutation occurs in somatic cells early after fertilization, the distribution of mutant cells determines the diversity of clinical phenotypes (types I–III).
The Glaucoma Clinical Practice Guidelines (5th edition) list the following five mechanisms for intraocular pressure elevation in SWS1).
Histological examination of enucleated eyes has revealed the following findings, which are similar to those seen in primary congenital glaucoma.
In cases that develop in infancy, developmental abnormalities of the angle are considered the most important factor.
In cases that develop in late adolescence to adulthood, elevated episcleral venous pressure due to hemangioma is the main cause 1). Gonioscopy shows minimal angle abnormalities, but blood is often observed in the angle, correlating with elevated episcleral venous pressure. In this condition, aqueous humor suppressants are considered most effective, and medical therapy is the first-line treatment.
This is because the mechanisms of glaucoma development are fundamentally different between the two. In the early-onset type, congenital angle dysgenesis is the main cause, and there is a problem with the trabecular meshwork or angle structure itself, so angle surgery (trabeculotomy or goniotomy) that physically opens the outflow pathway is effective 1). In contrast, in the late-onset type, elevated episcleral venous pressure is the main cause, and the angle structure itself is relatively normal. When episcleral venous pressure is high, aqueous humor suppressants are most effective, and medical therapy is the first-line treatment 1). If medical therapy is ineffective, filtering surgery or tube shunt surgery that can bypass the episcleral venous system is considered. For details, see the section on “Standard treatment”.
In the management of glaucoma associated with SWS, future progress is expected in the following areas.
日本緑内障学会. 緑内障診療ガイドライン(第5版). 日眼会誌. 2022;126:85-177.
European Glaucoma Society. Terminology and Guidelines for Glaucoma, 5th Edition. PubliComm, Savona. 2020.
European Glaucoma Society. Terminology and Guidelines for Glaucoma, 6th Edition. Br J Ophthalmol. 2025.
Yeom S, Comi AM. Updates on Sturge-Weber Syndrome. Handb Clin Neurol. 2015;132:157-168. doi:10.1016/B978-0-444-62702-5.00011-1.
Yadav PS, Adhikari P, Mehta B, et al. Unmasking Sturge-Weber Syndrome in Adulthood: A Case with Extrafacial Port-Wine Stain and Delayed Neurological Symptoms. Ann Med Surg. 2024;86:3679-3682.
Ainuz BY, Wolfe EM, Wolfe SA. Surgical Management of Facial Port-Wine Stain in Sturge Weber Syndrome. Cureus. 2021;13(1):e12637.
Pathak BD, Sharma S, Adhikari A, et al. Sturge-Weber Syndrome with Bilateral Port-Wine Stain. Case Rep Pediatr. 2022;2022:2191465.
Shirley MD, Tang H, Gallione CJ, et al. Sturge-Weber syndrome and port-wine stains caused by somatic mutation in GNAQ. N Engl J Med. 2013;368:1971-1979.
Stallworth JY, O’Brien KS, Han Y, Oatts JT. Efficacy of Ahmed and Baerveldt glaucoma drainage device implantation in the pediatric population: A systematic review and meta-analysis. Surv Ophthalmol. 2023;68(4):616-629.