Diffuse Choroidal Hemangioma
1. What is diffuse choroidal hemangioma?
Section titled “1. What is diffuse choroidal hemangioma?”Definition and Concept
Section titled “Definition and Concept”There are two types of choroidal hemangioma: circumscribed (solitary) and diffuse. Circumscribed type is well-defined and occurs sporadically, while diffuse type often has extensive tumor involvement and unclear borders. Diffuse choroidal hemangioma almost always occurs in association with Sturge-Weber syndrome (encephalofacial angiomatosis).
Sturge-Weber syndrome is one of the phakomatoses (neurocutaneous syndromes) and is caused by ectopic proliferation of neural crest cells during embryonic development. It is characterized by the formation of hemangiomas on the face, eye, and leptomeninges.
Epidemiology
Section titled “Epidemiology”The incidence of Sturge-Weber syndrome is estimated at 1 in 20,000 to 50,000. It is not hereditary; the main cause is a sporadic somatic mosaic mutation in the GNAQ gene. It is a congenital condition present at birth and is detected in infancy or early childhood. It occurs in the ipsilateral fundus of patients with cutaneous hemangioma (port-wine stain) in the distribution of the first and second branches of the trigeminal nerve. Clear data on gender or racial differences are lacking, but as a sporadic disease, it can occur in all races.
Comparison with Circumscribed Choroidal Hemangioma
Section titled “Comparison with Circumscribed Choroidal Hemangioma”| Feature | Diffuse Choroidal Hemangioma | Circumscribed (Solitary) Choroidal Hemangioma |
|---|---|---|
| Associated Syndrome | Always associated with Sturge-Weber syndrome | No systemic disease (sporadic) |
| Fundus Distribution | Extensive, ill-defined borders | Localized, relatively well-defined borders |
| Color tone | Tomato ketchup fundus | Orange elevated lesion |
| Glaucoma association | More than half | Rare |
| Onset timing | Congenital / infancy | Often in adulthood |
Diffuse choroidal hemangioma almost always occurs in association with Sturge-Weber syndrome (encephalofacial angiomatosis). Sturge-Weber syndrome is characterized by the triad of facial port-wine stain, leptomeningeal angioma, and ocular findings (diffuse choroidal hemangioma, glaucoma). The incidence is 1 in 20,000 to 50,000, and it is not hereditary. The hemangioma occurs in the fundus on the same side as the facial port-wine stain.
2. Main symptoms and clinical findings
Section titled “2. Main symptoms and clinical findings”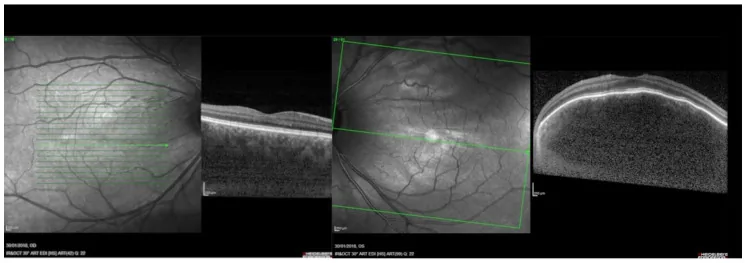
Fundus findings
Section titled “Fundus findings”The most characteristic fundus finding of diffuse choroidal hemangioma is the “tomato ketchup fundus,” where the entire fundus takes on a widespread orange-red hue. The tumor occupies a large area of the choroid, and its borders are indistinct. In ordinary fundus photographs, it can be recognized as a marked difference in redness compared to a normal fundus.
The main fundus and ocular findings are shown below.
- Diffuse choroidal thickening: The choroid is thickened extensively from the posterior pole to the periphery.
- Indistinct borders: Unlike circumscribed hemangioma, the tumor margins are unclear.
- Serous retinal detachment: The main cause of vision loss. Presents with visual field defects and metamorphopsia.
- Hyperopia: Refractive changes occur along the ocular axis due to choroidal elevation.
- Orange elevated lesion: Recognized as an orange elevation in the posterior pole.
Fluorescein Angiography and Imaging Findings
Section titled “Fluorescein Angiography and Imaging Findings”Characteristic findings on various imaging tests are shown below.
- FA (Fluorescein Angiography): Reticular hyperfluorescence within the tumor is seen from the early phase, and dye pooling increases in the late phase (diffuse pattern).
- ICGA (Indocyanine Green Angiography): Better visualization of choroidal vessels than FA, allowing clear identification of tumor vascular structures.
- OCT: Observed as a broad, localized elevation of the posterior choroid. Also useful for evaluating serous retinal detachment.
- Ultrasound: Depicted as a solid elevated lesion. B-scan shows extensive choroidal thickening.
- CT: Similar to ultrasound, depicted as a solid lesion.
Glaucoma Complication
Section titled “Glaucoma Complication”In Sturge-Weber syndrome, glaucoma occurs in more than half of cases. Depending on the age of onset, infantile-onset (congenital glaucoma type) and childhood-onset (acquired type) are each observed in about half of the cases.
- Infantile-onset type: Presents with features of congenital glaucoma such as corneal enlargement, buphthalmos (corneal enlargement), and Haab’s striae. Surgery is often required.
- Childhood-onset type: Intraocular pressure may initially stabilize within the normal range, but an increase in intraocular pressure may occur during follow-up.
- Episcleral hemangioma: Some cases also have hemangioma in the episclera on the affected side.
Glaucoma occurs in more than half of patients with Sturge-Weber syndrome. Infantile-onset and childhood-onset each account for half, and an increase in intraocular pressure may occur later during follow-up. If not properly managed, glaucoma can lead to visual field defects and vision loss, so long-term regular intraocular pressure measurement and fundus examination are essential. Particularly in infancy, buphthalmos and corneal opacity require urgent intervention.
3. Causes and Risk Factors
Section titled “3. Causes and Risk Factors”Pathogenesis
Section titled “Pathogenesis”The development of Sturge-Weber syndrome and diffuse choroidal hemangioma is caused by ectopic proliferation of neural crest cells during the embryonic period. A choroidal hamartoma is formed, and the normal choroidal structure is extensively replaced by vascular tissue.
At the genetic level, mutations in the GNAQ gene (guanine nucleotide-binding protein Gq alpha) (c.548G>A, p.Arg183Gln) are detected in many cases. This mutation occurs as a somatic mosaic mutation, so it is not hereditary and is not transmitted from parent to child in principle. The GNAQ mutation is thought to reduce GTPase activity and constitutively activate angiogenic signaling.
Main Risk Factors and Characteristics
Section titled “Main Risk Factors and Characteristics”- GNAQ somatic mosaic mutation: Most important as a causative mutation. It is not a germline mutation and is not hereditary.
- Congenital condition: Present from birth, mainly due to accidental mutations during the embryonic period rather than genetic background.
- Environmental factors: No established environmental risk factors.
- Family history: In principle, it is not inherited, so familial clustering is usually not observed.
Pathogenesis of glaucoma
Section titled “Pathogenesis of glaucoma”Two main mechanisms are involved in the development of glaucoma in Sturge-Weber syndrome.
- Elevated episcleral venous pressure: Angioma of the episclera increases venous pressure, leading to increased resistance to aqueous outflow. This mechanism is common in glaucoma after childhood.
- Angle abnormality: Developmental abnormality of the anterior chamber angle impairs aqueous outflow. This is often involved in glaucoma that develops in infancy.
4. Diagnosis and examination methods
Section titled “4. Diagnosis and examination methods”Diagnostic approach
Section titled “Diagnostic approach”The combination of facial port-wine stain (cutaneous angioma in the distribution of the trigeminal nerve) and ipsilateral fundus changes strongly supports the diagnosis of diffuse choroidal hemangioma. The following examinations are used for ophthalmic diagnosis.
Fundus examination and imaging:
- Fundus photography (including wide-angle fundus imaging): documentation and follow-up of tomato ketchup fundus
- FA/ICGA: confirmation of early reticular hyperfluorescence (diffuse pattern)
- OCT: evaluation of extensive choroidal thickening and serous retinal detachment
- Ultrasound (A-scan and B-scan): confirmation of solid lesion and measurement of thickness
Systemic evaluation:
- Head MRI: Leptomeningeal angioma and calcification (evaluated by a pediatric neurologist in conjunction with ophthalmic findings)
- Intraocular pressure measurement: Glaucoma screening (under general anesthesia if necessary)
- Gonioscopy: Evaluation of angle abnormalities
Differential Diagnosis
Section titled “Differential Diagnosis”Diseases that require differentiation from diffuse choroidal hemangioma are listed below.
| Differential Diagnosis | Key Differences |
|---|---|
| Circumscribed choroidal hemangioma | Well-defined, localized, no systemic syndrome |
| Choroidal malignant melanoma (diffuse type) | Malignant features, pigment changes, risk of metastasis |
| Posterior uveitis | Inflammatory findings, vitreous opacity |
| Normal fundus color variation | No systemic findings of Sturge-Weber syndrome |
Points to note at diagnosis
Section titled “Points to note at diagnosis”If the fundus appears “somewhat red,” especially in infants with port-wine stains, this disease should be actively suspected. In infants, general anesthesia may be required for examination, and collaboration with anesthesiology and pediatrics is important.
5. Standard treatment
Section titled “5. Standard treatment”Overview of treatment strategy
Section titled “Overview of treatment strategy”Treatment of diffuse choroidal hemangioma is performed stepwise depending on the presence of symptoms and the type of complications. Surgical removal of the choroidal hemangioma itself is not usually performed. The main targets of treatment are serous retinal detachment (the main cause of vision loss) and glaucoma.
1. Observation
Section titled “1. Observation”If asymptomatic and there are no problems with vision or intraocular pressure, continue the following regular observations:
- Intraocular pressure measurement (early detection of glaucoma)
- Fundus photography and OCT to check for serous retinal detachment
- Visual acuity and refraction tests (assessment of hyperopia and amblyopia)
- Follow-up with FA/OCT as needed
2. Treatment for serous retinal detachment and visual dysfunction
Section titled “2. Treatment for serous retinal detachment and visual dysfunction”If vision loss or serous retinal detachment is observed, active treatment is performed.
- Intravenous administration of verteporfin (photosensitizer) followed by irradiation with a 689 nm wavelength laser
- The efficacy of PDT for choroidal hemangioma has been reported
- Not covered by insurance (for diffuse choroidal hemangioma)
- Expected to resolve serous retinal detachment and improve visual acuity
- Efficacy for circumscribed choroidal hemangioma is established, but application to diffuse type has technical limitations due to the large irradiation area
Low-dose radiation therapy:
- Low-dose irradiation of approximately 20 Gy is performed
- May be effective. Expected to resolve retinal detachment and improve visual acuity
- External beam radiation (teletherapy) is often used
Surgical removal is not usually performed. Diffuse choroidal hemangioma extensively involves the entire choroid, making surgical removal technically and functionally difficult. For serous retinal detachment causing visual loss, PDT or low-dose radiation therapy (approximately 20 Gy) is performed and is often effective. For glaucoma, intraocular pressure is controlled with eye drops or surgery.
3. Glaucoma Treatment
Section titled “3. Glaucoma Treatment”Since glaucoma complicates more than half of cases, its management is an important treatment issue.
Pharmacotherapy (eye drops):
- Prostaglandin-related drugs (suppress aqueous humor production, enhance outflow)
- Beta-blockers (suppress aqueous humor production)
- Carbonic anhydrase inhibitors (topical or oral)
- However, in glaucoma due to elevated episcleral venous pressure, eye drops may have limited effectiveness
Surgical treatment:
- For infantile-onset (congenital glaucoma type), goniotomy or trabeculotomy may be attempted
- In adult type, filtering surgery (trabeculectomy or tube shunt surgery) is considered
- When episcleral venous pressure is high, careful postoperative management of trabeculectomy is required
4. Systemic management
Section titled “4. Systemic management”- Epilepsy management: Seizure control with antiepileptic drugs (pediatric neurology)
- Management of intellectual disability: Early intervention and support systems
- Headache and hemiparesis: Monitoring of neurological symptoms and symptomatic treatment
- Multidisciplinary collaboration: Cooperation among ophthalmology, pediatric neurology, dermatology, epilepsy specialists, and habilitation team
6. Pathophysiology and detailed mechanisms
Section titled “6. Pathophysiology and detailed mechanisms”Nature as a vascular hamartoma
Section titled “Nature as a vascular hamartoma”Diffuse choroidal hemangioma is embryologically classified as a vascular hamartoma. A hamartoma is a benign tumor-like lesion characterized by abnormal proliferation of tissue components normally present at that site, distinct from a true neoplasm. In this condition, mature vascular elements proliferate excessively within the choroid, replacing the normal choriocapillaris and medium-to-large vessel structures.
GNAQ Gene Mutation and Molecular Mechanisms
Section titled “GNAQ Gene Mutation and Molecular Mechanisms”As the molecular basis of Sturge-Weber syndrome, somatic mosaic mutations in the GNAQ gene (c.548G>A, p.Arg183Gln) have been identified in many cases.
- Function of GNAQ (Gqα): Gα subunit involved in downstream signaling of G protein-coupled receptors (GPCRs)
- Effect of mutation: The Arg183Gln mutation reduces GTPase activity, maintaining GNAQ in a constitutively active state
- Downstream signaling: Gq-mediated PLC-β activation → IP3/DAG production → PKC activation → constitutive activation of the MAPK cascade (MEK/ERK)
- Impact on angiogenesis: Overproduction of angiogenic factors such as VEGF is promoted, leading to abnormal blood vessel formation
- Significance of somatic mosaic mutation: Since mutations occur only in some cells during early embryogenesis, expression patterns vary among individuals, resulting in symptom diversity
Detailed Pathogenesis of Glaucoma
Section titled “Detailed Pathogenesis of Glaucoma”At least two mechanisms are proposed for the development of glaucoma in Sturge-Weber syndrome.
Mechanism 1: Elevated episcleral venous pressure Hemangiomas in the episclera (sub-Tenon’s capsule) increase episcleral venous pressure, impairing aqueous humor outflow through Schlemm’s canal and the trabecular meshwork. When episcleral venous pressure exceeds the normal value (approximately 10 mmHg), intraocular pressure increases additively. This mechanism is common in acquired glaucoma that develops after childhood.
Mechanism 2: Angle dysgenesis Developmental abnormalities of the anterior chamber angle (angle dysgenesis) result in incomplete development of the trabecular meshwork, causing anatomical dysfunction of the aqueous outflow pathway. This mechanism is often associated with infantile-onset (congenital) glaucoma.
Pathogenesis of Serous Retinal Detachment
Section titled “Pathogenesis of Serous Retinal Detachment”Increased vascular permeability in diffuse choroidal hemangioma leads to extravasation of intravascular components and fluid accumulation in the subretinal space. This causes serous retinal detachment, which is the main cause of vision loss and visual field defects. PDT or radiation therapy reduces tumor vessels, decreasing exudation and allowing retinal reattachment.
Pathophysiology Outside the Eye
Section titled “Pathophysiology Outside the Eye”- Leptomeningeal angioma: Angioma of the cerebral cortex causes focal ischemia and calcification, leading to epilepsy, intellectual disability, and hemiparesis.
- Facial port-wine stain: Present from birth as telangiectasia of the skin.
- Calcification: “Tram-track” calcification in the cerebral cortex beneath the leptomeningeal angioma is a characteristic imaging finding of Sturge-Weber syndrome.
7. Latest Research and Future Perspectives
Section titled “7. Latest Research and Future Perspectives”Molecular targeted therapy targeting downstream of GNAQ
Section titled “Molecular targeted therapy targeting downstream of GNAQ”The MAPK cascade (MEK/ERK pathway) constitutively activated by GNAQ mutation is widely studied as a therapeutic target in uveal melanoma research. Since the same molecular abnormality exists in Sturge-Weber syndrome/diffuse choroidal hemangioma, the application of MEK inhibitors (trametinib, binimetinib, etc.) and GNAQ direct inhibitors (YM-254890, etc.) is being investigated at the basic research level.
In fact, as molecular targeted therapy for Sturge-Weber syndrome as a whole, mTOR inhibitors (sirolimus) and treatments targeting the PI3K-AKT-mTOR pathway have been trialed, with reports of symptom improvement in some patients. However, large-scale studies directly evaluating efficacy for diffuse choroidal hemangioma itself are currently limited.
Optimization of PDT treatment
Section titled “Optimization of PDT treatment”PDT has been shown to be effective for serous retinal detachment in diffuse choroidal hemangioma in multiple case reports, but the protocol (irradiation field, energy, number of sessions) for diffuse extensive lesions has not been standardized. Compared to circumscribed choroidal hemangioma, diffuse lesions require a larger irradiation area, posing technical challenges. Establishment through future multicenter prospective studies is expected.
New developments in glaucoma management
Section titled “New developments in glaucoma management”Conventional filtration surgery for glaucoma caused by elevated episcleral venous pressure carries risks of choroidal effusion and malignant glaucoma under conditions of high episcleral venous pressure. Research on the efficacy of tube shunt surgery (Ahmed valve, Baerveldt tube) is progressing. Additionally, the potential application of selective laser trabeculoplasty (SLT) is being investigated.
Perspectives on GNAQ genetic diagnosis
Section titled “Perspectives on GNAQ genetic diagnosis”Detection of GNAQ mutations in skin biopsy or blood samples is being applied for definitive diagnosis of Sturge-Weber syndrome. In the future, early diagnosis via liquid biopsy (ctDNA) and improved mutation detection rates are expected to enable more reliable diagnosis.
8. References
Section titled “8. References”- Shirley MD, Tang H, Gallione CJ, et al. Sturge-Weber syndrome and port-wine stains caused by somatic mutation in GNAQ. N Engl J Med. 2013;368(21):1971-1979.
- Baselga E, Torrelo A, Mediero IG, et al. Sturge-Weber syndrome: report of 3 cases. Pediatr Dermatol. 2019;36(6):932-934.
- Bhatt A, Bhatt N. Sturge-Weber syndrome: a rare neurocutaneous disorder. J Pediatr Neurosci. 2021;16(1):1-6.
- Zreik O, Elabassy HM, Bakhurji E, Al-Johani SM. Sturge-Weber syndrome: an updated review. Clin Ophthalmol. 2023;17:2369-2381.
- Nassiri N, Rootman DB, Rootman J, Goldberg RA. Orbital and adnexal lymphangiomas: a review of management. Surv Ophthalmol. 2015;60(3):245-257.
