Sporadic (most common)
Inheritance: Non-hereditary (sporadic)
Lesion distribution: Unilateral, peripheral retina
Clinical course: Usually non-progressive, asymptomatic
Systemic complications: None
Retinal cavernous hemangioma is a vascular malformation of the retina formed by a cluster of low-flow dilated veins. It is usually non-hereditary, unilateral, non-progressive, and occurs sporadically in the peripheral retina. Cases involving the optic disc or macula, as well as autosomal dominant forms associated with skin, central nervous system, and hepatic hemangiomas, have also been reported.
Strictly speaking, this condition is not a tumor but is classified as a vascular malformation. It is a conglomeration of blood vessels composed of vascular endothelial cells, smooth muscle cells, and stromal cells, and is not a proliferation of a single cell type. According to the International Society for the Study of Vascular Anomalies (ISSVA) classification, it is categorized as a low-flow venous malformation and is also a target disease in the guidelines for the management of hemangiomas and vascular malformations.
Sporadic (most common)
Inheritance: Non-hereditary (sporadic)
Lesion distribution: Unilateral, peripheral retina
Clinical course: Usually non-progressive, asymptomatic
Systemic complications: None
Familial (autosomal dominant)
Inheritance: Autosomal dominant
Lesion distribution: Unilateral or bilateral, multiple
Clinical course: May have brain, skin, liver complications
Systemic complications: Cerebral cavernous malformation (CCM), cutaneous hemangioma, hepatic hemangioma
When the lesion is confined to the periphery, it is usually asymptomatic and is often discovered incidentally during fundus examination. If the lesion extends to the optic disc or macula, visual impairment and visual field defects may occur. When a fibrovascular membrane forms over the tumor and causes exudation or hemorrhage due to traction, the impact on visual function becomes apparent.
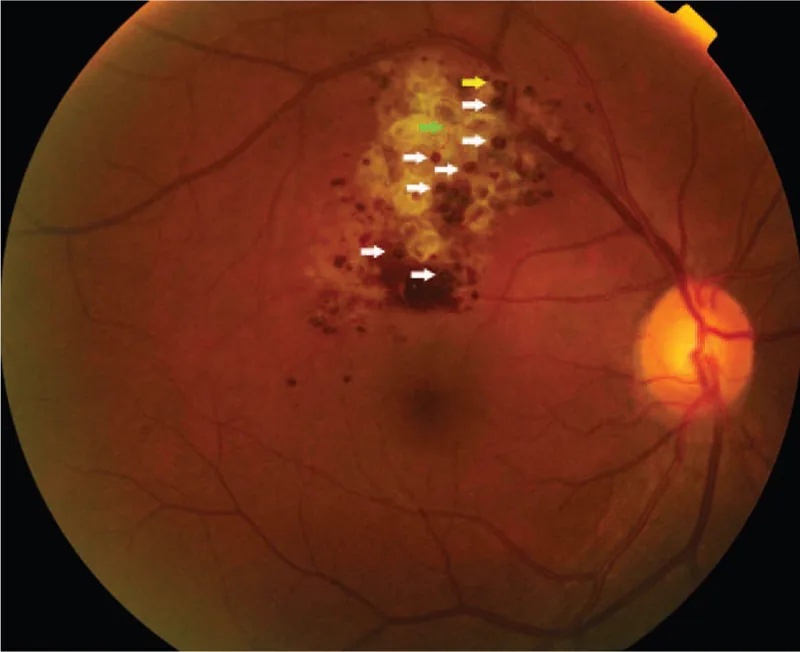
Fundus findings are most important for the diagnosis of this disease. Characteristic findings are shown below.
Fluorescein angiography is the most useful examination for differential diagnosis. Characteristic findings are shown below.
On optical coherence tomography (OCT), it appears as a cystic, protruding, hyperreflective mass arising from the inner retinal layers. Optical coherence tomography angiography (OCT-A) shows poor internal blood flow, confirming its characteristics as a low-flow vascular malformation.
The exact mechanism of retinal cavernous hemangioma is not fully understood. It is thought to be caused by abnormal vascular development during the embryonic period, specifically a localized hamartomatous change in the venous system. No environmental risk factors for sporadic cases have been identified to date.
Familial cases (autosomal dominant inheritance) share a common genetic background with cerebral cavernous malformation (CCM). The following three genes have been identified as causative for cerebral cavernous malformation, and their association with systemic vascular malformations including ocular lesions has been reported1).
| Gene | Alias | Encoded Protein | Main Function |
|---|---|---|---|
| CCM1 | KRIT1 | Krev interaction trapped 1 | Cell adhesion and vascular endothelial homeostasis |
| CCM2 | MGC4607 | Malcavernin | Binding to CCM1 and signal transduction |
| CCM3 | PDCD10 | Programmed cell death 10 | Regulation of apoptosis and vascular permeability |
These gene mutations are involved in cell adhesion, signal transduction, and permeability control of vascular endothelial cells, leading to venous dilation and weakening of the vessel wall2).
Accurate incidence data are limited, but retinal cavernous hemangioma is considered a rare disease3). The population-based prevalence of cerebral cavernous malformations is 0.1–0.5%, but the proportion with ocular involvement is even smaller. Clear data on age of onset and sex differences are lacking. Familial cases are rare, but several families with combined brain, skin, and liver hemangiomas have been reported.
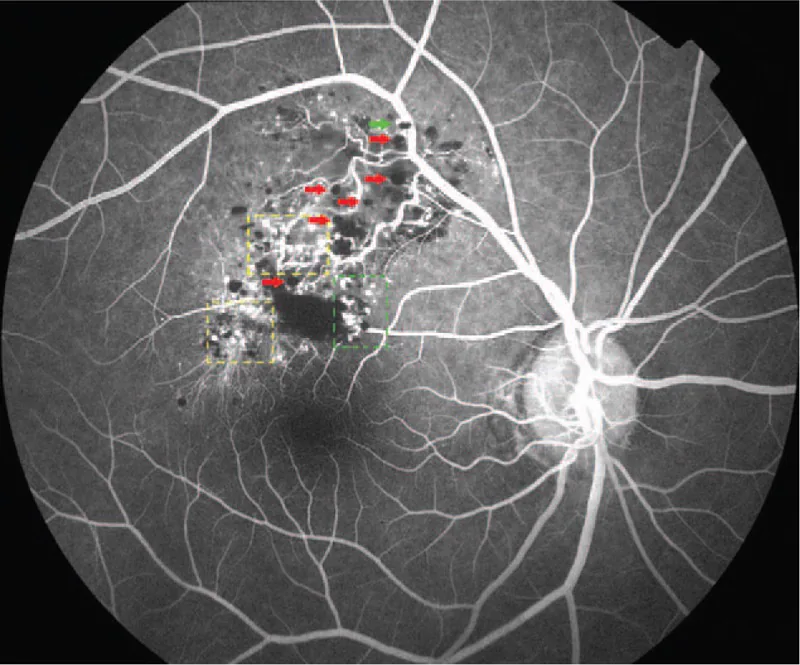
Diagnosis is primarily based on characteristic fundus findings (cluster of grapes-like multilobular dark red mass) and fluorescein angiography findings (fluorescent cap sign, venous filling pattern with little leakage). In typical cases, biopsy is not required and clinical diagnosis is possible.
In familial (autosomal dominant) retinal cavernous hemangioma, there is a genetic association with cerebral cavernous malformations (CCM). Mutations in CCM1, CCM2, and CCM3 genes are common causes, leading to multiple vascular malformations in the retina, brain, skin, and liver. In familial cases, contrast-enhanced brain MRI is essential to rule out undetected brain lesions (which can cause epilepsy or cerebral hemorrhage). In sporadic cases, no association with brain lesions has been demonstrated.
The most important differential diagnosis for retinal cavernous hemangioma is retinal capillary hemangioma (VHL disease). Fluorescein angiography findings are the key point to distinguish between the two diseases.
| Disease | Fundus Findings | FA Findings | Systemic Complications | Treatment |
|---|---|---|---|---|
| Retinal Cavernous Hemangioma | Multilobular dark red mass (grape-like clusters) | Slow venous filling, fluorescent cap sign, minimal leakage | CCM (familial) | Usually observation |
| Retinal capillary hemangioma (VHL disease) | Orange-red semitransparent tumor + dilated feeding and draining vessels | Rapid filling in arterial phase, intense fluorescein leakage | VHL tumors (e.g., renal cell carcinoma) | Laser, surgery, anti-VEGF |
| Coats disease | Exudative retinal detachment, capillary aneurysms | Intense leakage from capillary aneurysms | None (common in children) | Laser, surgery |
| Retinal vasoproliferative tumor | Peripheral yellowish-white mass | Progressive filling and leakage | None | Laser, PDT |
Both diseases are vascular lesions of the retina, but they can be clearly distinguished by fluorescein angiography findings. Retinal capillary hemangioma (VHL disease) is characterized by an orange-red tumor with dilated feeding and draining vessels, showing rapid filling from the arterial phase and intense fluorescein leakage on FA. In contrast, retinal cavernous hemangioma is characterized by slow filling during the venous filling phase and a fluorescent cap sign, with little fluorescein leakage even in the late phase. On fundus examination, retinal cavernous hemangioma appears as a grape-like multilocular dark red mass, and differentiation is usually possible.
Retinal cavernous hemangioma is non-progressive and usually does not require treatment. Fibrous proliferative membranes may form on the lesion, which can cause exudation or hemorrhage due to traction. In asymptomatic cases without complications, regular fundus examinations are performed for follow-up.
Treatment is considered when the following complications occur.
Vitrectomy: Vitrectomy is considered when tractional retinal detachment due to fibrous proliferative membrane or vitreous hemorrhage associated with traction occurs. Since it is a low-flow vascular malformation, the risk of intraoperative and postoperative bleeding is considered similar to that of general retinal surgery. Most surgical outcomes are based on case reports, and large-scale evidence is lacking4).
Laser photocoagulation: Because it is a low-flow vascular malformation, the response to photocoagulation is poor. It is not actively recommended at present.
In familial cases, not only management of ocular lesions but also neurological management for cerebral cavernous malformations is important. For brain lesions, observation is the basic approach if asymptomatic, but if epileptic seizures or neurological symptoms appear, pharmacotherapy or surgical intervention should be considered 5).
Usually, treatment is not required. It is a non-progressive vascular malformation, and in asymptomatic cases, regular fundus examination for follow-up is the basic policy. Vitreous surgery is indicated only when tractional complications (tractional retinal detachment, vitreous hemorrhage) due to fibroproliferative membranes occur. Laser photocoagulation is generally not performed due to low flow and poor efficacy. In familial cases, systemic management of cerebral vascular malformations is necessary in addition to ophthalmic follow-up.
Retinal cavernous hemangioma is not a tumor but a vascular malformation. It is a cluster of blood vessels composed of vascular endothelial cells, smooth muscle cells, and stromal cells, and is essentially characterized by not being a proliferation of a single cell (i.e., neoplastic proliferation). In the 2018 revised classification of the International Society for the Study of Vascular Anomalies (ISSVA), it is classified as a low-flow venous malformation, distinguished from arteriovenous malformations (high-flow) 6).
In familial (autosomal dominant) cases, loss-of-function mutations in CCM genes play a central role. CCM1 (KRIT1) regulates the integrin pathway involved in cell-cell adhesion, CCM2 (malcavernin) functions as a scaffold protein for CCM1, and CCM3 (PDCD10) is involved in apoptosis regulation and vascular permeability control 2). These mutations cause disruption of endothelial cell-cell adhesion, leading to endothelial-to-mesenchymal transition, vasodilation, and increased permeability.
In sporadic (non-hereditary) cases, local vascular developmental abnormalities due to somatic mutations are suggested. It is thought that abnormalities during embryonic retinal vascular development form cluster-of-grapes-like multilocular venous dilations, but the detailed mechanism remains largely unknown.
Being a low-flow venous malformation defines the clinical characteristics of this disease. Due to low blood flow, the following features arise:
On the surface (vitreous side) of the vascular malformation, gliotic cap or fibrovascular membrane may form. Contraction of this membrane can cause tractional retinal detachment or vitreous hemorrhage. The fibrovascular membrane is not neoplastic but a secondary reactive proliferation.
Elucidation of molecular mechanisms due to CCM gene mutations is progressing. In animal experiments, Rho/ROCK signaling inhibitors and PI3K/Akt/mTOR pathway inhibitors have shown promising results in cerebral cavernous malformation models 7). Application to ocular lesions requires further research.
Multi-gene panel testing using next-generation sequencing (NGS) has become widespread and is used for definitive diagnosis of familial cases, carrier diagnosis, and genetic counseling for families. Interpretation of pathogenic variants of CCM gene mutations is also accumulating 1).
Regarding management of tractional complications by vitrectomy, case reports and case series literature are accumulating 4,8). There are no large-scale randomized controlled trials, and prospective studies are awaited.
Fischer A, Zalvide J, Faurobert E, et al. Cerebral cavernous malformations: from CCM genes to endothelial cell homeostasis. Trends Mol Med. 2013;19(5):302-308.
Couteulx SL, Jung HH, Labauge P, et al. Truncating mutations in CCM1, encoding KRIT1, cause hereditary cavernous angiomas. Nat Genet. 1999;23(2):189-193.
Gass JDM. Cavernous hemangioma of the retina. A neuro-oculocutaneous syndrome. Am J Ophthalmol. 1971;71(4):799-814.
Messmer EP, Font RL, Laqua H, et al. Cavernous hemangioma of the retina. Immunohistochemical and ultrastructural observations. Arch Ophthalmol. 1984;102(3):413-418.
Haller JA Jr, Dortz J, Goldberg MF. Familial retinal cavernous hemangiomas. Arch Ophthalmol. 1979;97(5):879-883.
ISSVA Classification of Vascular Anomalies. International Society for the Study of Vascular Anomalies. 2018 update. Available at: https://www.issva.org.
Lewis RA, Cohen BH, Wise GN. Cavernous haemangioma of the retina and optic disc. A report of three cases and a review of the literature. Br J Ophthalmol. 1975;59(8):422-434.
Shields JA, Shields CL, Timmers E, et al. Spectrum of vitreoretinal surgery. Retina. 1992;12(1):1-11.