Choroidal Osteoma
1. What is choroidal osteoma?
Section titled “1. What is choroidal osteoma?”Choroidal osteoma is a benign tumor in which heterotopic bone tissue forms within the choroid. It commonly occurs in the posterior pole of the fundus and appears as a flat, yellowish-white lesion. The cause is unknown; theories include osseous choristoma (heterotopic bone tissue) and inflammatory involvement.
During the course, pigmentation, retinal pigment epithelium (RPE) atrophy, and decalcification may occur, so fundus findings change depending on the disease stage. It tends to slowly enlarge but eventually stops.
Epidemiology
Section titled “Epidemiology”- It is a relatively rare benign tumor, accounting for a small portion of all intraocular tumors.
- It is more common in young women, and hormonal involvement is suspected, but the cause is unknown.
- It can occur bilaterally.
- It commonly occurs in the posterior pole, especially around the optic disc.
Stages of Disease Progression
Section titled “Stages of Disease Progression”Although there is no clear classification system, the following stages of disease progression are distinguished.
| Stage | Characteristics |
|---|---|
| Early | Yellow-white or yellow-orange patchy, flat lesion in the posterior pole. Almost no elevation. |
| Progressive | Decalcification, degeneration and atrophy of the RPE and outer retina occur, changing the fundus appearance. |
| CNV Complicated | Choroidal neovascularization (CNV) develops, leading to retinal hemorrhage and vision loss. |
It is a benign tumor in which ectopic bone tissue forms in the choroid. It commonly occurs in the posterior pole and is slightly more frequent in young women. There are no reports of malignant transformation or metastasis, but if it extends to the macula, it can cause vision loss. There is no fundamental treatment, and observation is the basic approach.
2. Main Symptoms and Clinical Findings
Section titled “2. Main Symptoms and Clinical Findings”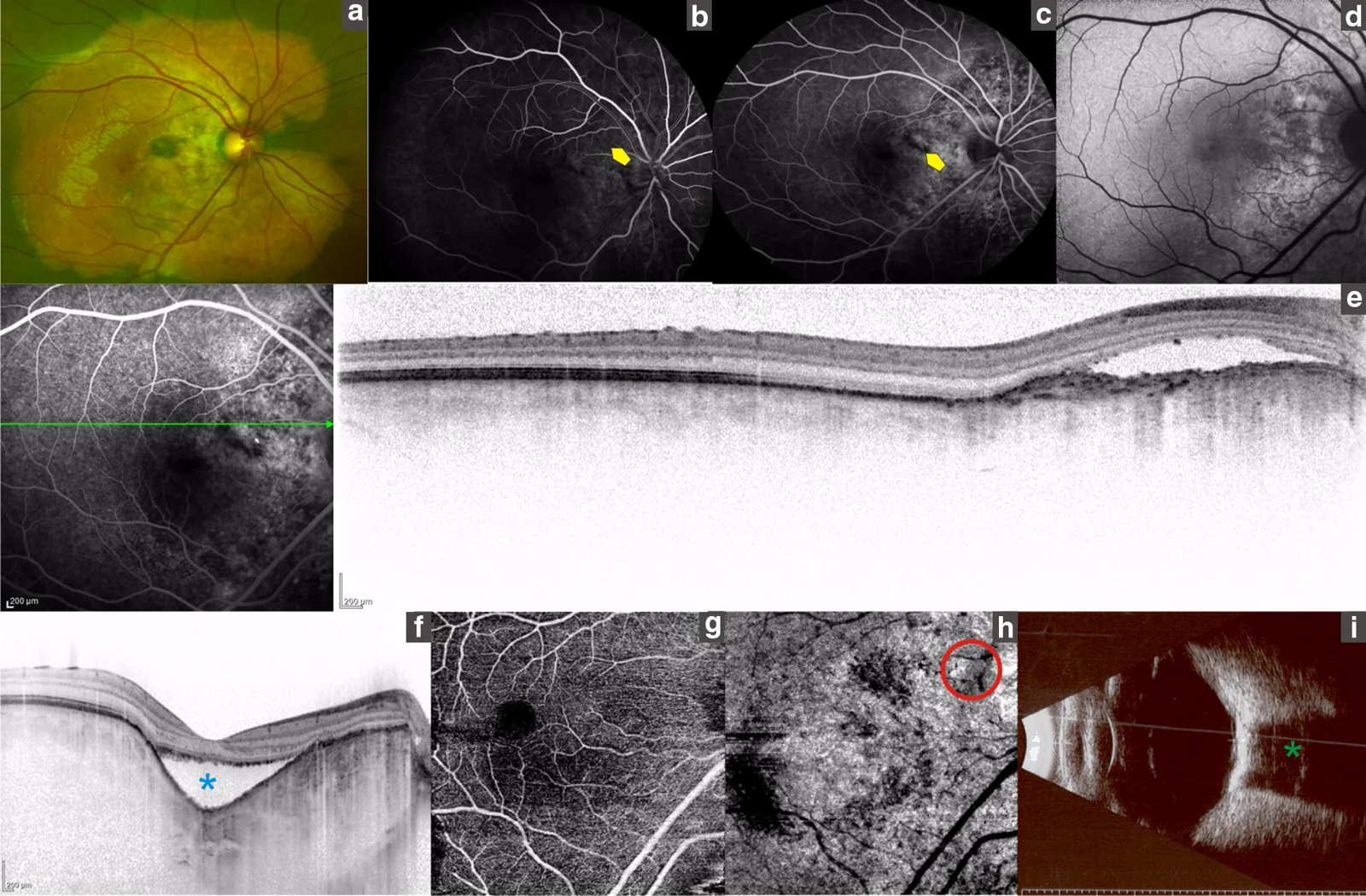
Subjective Symptoms
Section titled “Subjective Symptoms”- Visual field defect: Occurs corresponding to the tumor location. It is often difficult to notice in the early stages.
- Vision loss: When the lesion involves the macula, significant vision loss occurs.
- Retinal hemorrhage and vitreous hemorrhage: Occur when CNV develops. They cause sudden vision loss.
- If the tumor does not involve the macula, subjective symptoms may be minimal.
Fundus Findings
Section titled “Fundus Findings”The following findings are observed in the posterior pole, especially around the optic disc.
- Yellow-white to yellow-orange patchy lesion with almost no elevation. It may be accompanied by irregularities.
- Observed as a well-defined, flat, yellowish-white elevated lesion. Often accompanied by fine vessels on the tumor surface.
- As it progresses, degeneration and atrophy occur in the adjacent RPE and outer retina, and the fundus findings also change.
Imaging Findings
Section titled “Imaging Findings”Fluorescein angiography (FA):
- From the early phase, patchy or granular hyperfluorescence corresponding to the tumor is observed.
- In the late phase, it becomes diffuse hyperfluorescence.
- When CNV is present, fluorescein leakage is observed.
- A plate-like high-reflectivity lesion corresponding to the tumor and posterior acoustic shadowing (loss or attenuation of reflection) are observed.
CT (definitive diagnosis):
- The tumor area shows high attenuation (high CT value) similar to bone.
OCT:
- Useful for evaluating tumor height, CNV, and subretinal fluid.
3. Causes and Risk Factors
Section titled “3. Causes and Risk Factors”The pathogenesis of choroidal osteoma is unknown. Currently, the following hypotheses have been proposed.
- Choristoma theory: Assumes a congenital abnormality in which ectopic bone tissue forms in the choroid.
- Inflammatory theory: Suggests that ossification occurs triggered by inflammation.
- Hormonal hypothesis: Since it is relatively common in young women, an association with hormone secretion is suspected, but the specific mechanism has not been elucidated.
There have been no reports of malignancy or metastasis, and the tumor itself follows a benign course.
4. Diagnosis and Examination Methods
Section titled “4. Diagnosis and Examination Methods”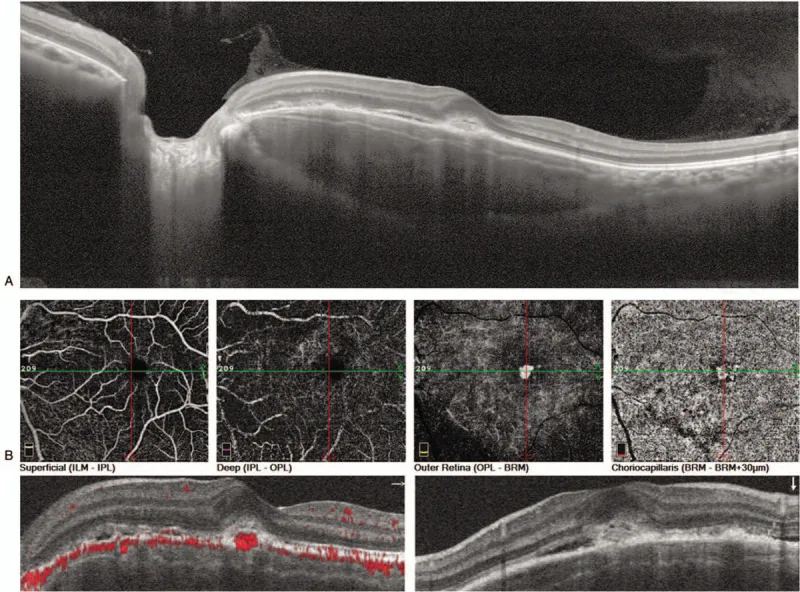
Diagnostic Points
Section titled “Diagnostic Points”A yellowish-white flat lesion in the posterior pole (especially around the optic disc) is observed on fundus examination, and definitive diagnosis is made by CT and ultrasonography.
- Fundus examination: Yellowish-white to yellow-orange flat lesion in the posterior pole. Evaluate lesion enlargement, RPE changes, and presence of CNV.
- CT (definitive diagnosis): High attenuation values (high CT values) equivalent to bone at the tumor site are the key to definitive diagnosis.
- Ultrasonography: Characteristic plate-like hyperreflective image with posterior acoustic shadowing supports the diagnosis.
- Fluorescein angiography (FA): Shows patchy/granular hyperfluorescence in the early phase and diffuse hyperfluorescence in the late phase. When CNV is present, fluorescein leakage is added.
- OCT: Allows detailed evaluation of changes in the RPE and neurosensory retina, subretinal fluid, and presence of CNV.
Differential Diagnosis
Section titled “Differential Diagnosis”Differentiation from diseases involving calcification is important.
| Disease | Key differentiating features |
|---|---|
| Retinoblastoma | Common in children. White mass with calcification. Assessed by CT and ultrasound. |
| Astrocytic hamartoma (associated with tuberous sclerosis) | White lesion around the optic disc and posterior pole. Calcification present. |
| Choroidal malignant melanoma | Dome-shaped elevation, pigmented. Double circulation pattern on FA. Low internal reflectivity on ultrasound. |
| Choroidal metastatic tumor | History of systemic malignancy. Flat, multifocal lesions. Early hyperfluorescence on FA. |
| Choroidal hemangioma | Orange-red color. Characteristic pattern on FA and ICGA. High reflectivity on ultrasound. |
The definitive diagnostic clue is a high-attenuation area on CT, similar to bone. Diagnosis is made in conjunction with acoustic shadowing on ultrasound. If fundus examination reveals a yellowish-white flat lesion at the posterior pole, suspect this disease and perform detailed examination with CT and ultrasound.
5. Standard treatment
Section titled “5. Standard treatment”Basic Policy: Observation
Section titled “Basic Policy: Observation”Since no curative treatment has been established, the basic approach is observation. It is important to regularly evaluate tumor enlargement and the presence of new blood vessels. As long as the macula is not involved, visual function is often minimally affected.
Treatment for CNV Onset
Section titled “Treatment for CNV Onset”If choroidal neovascularization (CNV) is confirmed, the following symptomatic treatments are considered to preserve vision.
- Photocoagulation (laser photocoagulation): Performed for CNV. Lesions outside the macula are more suitable.
- Photodynamic therapy (PDT): Photodynamic therapy using verteporfin. Although not covered by insurance, some efficacy has been reported.
- Anti-VEGF intravitreal injection: Ranibizumab, aflibercept, bevacizumab, etc. Not covered by insurance, but expected to induce CNV regression.
The indication and choice of treatment are determined individually based on the location of CNV, visual acuity, and patient background.
There is no curative treatment, and the basic approach is observation. If choroidal neovascularization develops and causes vision loss, symptomatic treatment with PDT or anti-VEGF intravitreal injection may be performed. Since these are not covered by insurance, the treatment plan should be decided after thorough discussion with the attending physician.
6. Pathophysiology and Detailed Mechanism of Onset
Section titled “6. Pathophysiology and Detailed Mechanism of Onset”Mechanism of Ectopic Bone Formation
Section titled “Mechanism of Ectopic Bone Formation”The essence of choroidal osteoma is ectopic bone formation within the choroid. The mechanism by which bone tissue arises in normal choroidal tissue is unknown, but the following points are known.
- The lesion gradually expands but tends to eventually stop.
- Since it is relatively common in young women, hormonal involvement is suspected, but the molecular mechanism is unknown.
- The choristoma theory assumes ectopic bone tissue due to embryonic abnormalities.
- The inflammation theory assumes ossification induced after inflammation, but a clear history of inflammation is often not confirmed.
Decalcification
Section titled “Decalcification”Decalcification of the tumor may occur during the course. When decalcification occurs, the border of the choroidal osteoma becomes indistinct and its color changes. There are reports that CNV is more likely to develop after decalcification. The frequency of decalcification and its quantitative impact on visual prognosis are still being studied through case accumulation.
Mechanism of Visual Impairment
Section titled “Mechanism of Visual Impairment”- The RPE and photoreceptors located directly above the tumor undergo degeneration and atrophy over time.
- When CNV develops, leakage from the vessels causes subretinal fluid and retinal hemorrhage, leading to acute vision loss.
- If the macula is involved, the foveal photoreceptors are damaged, resulting in severe vision loss. Once vision loss occurs, recovery is difficult.
Prognosis
Section titled “Prognosis”- Unless the macula is involved, there is no significant visual impairment.
- If the lesion extends to the macula, severe vision loss occurs and recovery is not expected.
- In bilateral cases, even if progression in one eye precedes, attention must be paid to the course of the other eye.
- There are no reports of malignant transformation or metastasis.
7. Latest Research and Future Prospects
Section titled “7. Latest Research and Future Prospects”Application of Anti-VEGF Therapy to CNV
Section titled “Application of Anti-VEGF Therapy to CNV”Although not covered by insurance, anti-VEGF intravitreal injections of ranibizumab or aflibercept have been applied to cases with CNV. Case reports and small series have reported CNV regression and visual improvement, but there are currently no randomized controlled trials regarding the administration protocol (interval, number of injections, combination with other treatments). Accumulation of evidence in the future is expected.
Detailed Evaluation with OCT and OCT-A
Section titled “Detailed Evaluation with OCT and OCT-A”Advances in OCT angiography (OCT-A) are enabling non-invasive detailed evaluation of the vascular structure of CNV. Quantitative assessment of CNV using OCT-A in choroidal osteoma and its application to follow-up are future challenges.
Elucidation of Decalcification Mechanism
Section titled “Elucidation of Decalcification Mechanism”The molecular mechanism of why decalcification occurs and whether CNV is more likely to develop after decalcification remains unknown. Research investigating the association with bone metabolism-related factors (such as RANKL and OPG) is ongoing.
8. References
Section titled “8. References”- Shields CL, Sun H, Demirci H, et al. Factors predictive of tumor growth, tumor decalcification, choroidal neovascularization, and visual outcome in 74 eyes with choroidal osteoma. Arch Ophthalmol. 2005;123(12):1658-1666.
- Shields JA, Shields CL. Intraocular Tumors: An Atlas and Textbook. 3rd ed. Philadelphia: Wolters Kluwer; 2015.
- Tsui I, Gillies MC, Barlow R, et al. Photodynamic therapy for choroidal neovascularization complicating choroidal osteoma. Retina. 2006;26(7):804-810.
- Nadarajah S, Blumenkranz MS. Intravitreal bevacizumab for choroidal neovascularization secondary to choroidal osteoma. Retina. 2009;29(6):888-890.
