Red Light Therapy (Myopia Progression Control)
1. What is red light therapy (myopia control)?
Section titled “1. What is red light therapy (myopia control)?”Repeated low-level red light therapy (RLRL) is a non-invasive treatment that suppresses myopia progression in children by irradiating the eye with low-intensity red visible light of specific wavelengths (mainly 650–670 nm). The standard protocol uses a dedicated handheld device, with sessions of 3 minutes twice daily, 5 days per week.
Positioning of RLRL therapy
Section titled “Positioning of RLRL therapy”RLRL therapy is a relatively new myopia progression control treatment for which evidence has rapidly accumulated since the 2020s. It is attracting attention as a “fourth option” after the three main conventional therapies (low-dose atropine eye drops, orthokeratology, and multifocal contact lenses), and most research has been conducted in East Asia, particularly in China.
Myopia is primarily caused by elongation of the axial length, with a 1 mm increase in axial length corresponding to approximately 3 diopters of myopia. Suppressing axial elongation is the most important goal of myopia management, and changes in axial length are the most important endpoint in evaluating the efficacy of RLRL therapy1).
Major complications associated with myopia include retinal detachment (30% increased risk per −1 D), myopic macular degeneration (58% increase), and open-angle glaucoma (20% increase)6). RLRL therapy is positioned as a preventive intervention to reduce these long-term risks.
In Asian children, the prevalence of myopia is particularly high, and if left untreated and progresses to high myopia, the risk of serious complications such as retinal detachment and myopic macular degeneration increases significantly. The estimated risk increase per 1 D of myopia is 30% for retinal detachment, 58% for myopic macular degeneration, 20% for open-angle glaucoma, and 21% for posterior subcapsular cataract6).
Since a 1 mm increase in axial length corresponds to approximately 3 D of myopia, suppressing axial elongation is the most important goal of myopia management. A meta-analysis by Haarman et al. (2020)7) showed that the risk of complications increases exponentially with the severity of myopia, highlighting the importance of intervention at an early stage for improving long-term prognosis.
RLRL therapy, as one of the myopia progression control interventions alongside orthokeratology and low-dose atropine eye drops, has rapidly accumulated evidence since the 2020s. In Japan, it is being introduced as a self-funded treatment in some facilities. A systematic review by Yam et al. (2025) summarizes the evidence for multiple myopia progression control interventions, including RLRL therapy5).
On the other hand, Bullimore et al. (2021) examined the risks and benefits of myopia management in general and pointed out that RLRL therapy carries a risk of retinal phototoxicity when irradiation parameters deviate6). Proper device management and adherence to irradiation intensity and duration are prerequisites for safe use.
RLRL therapy is a myopia progression suppression treatment that irradiates the eye with low-intensity (0.3–1 mW/cm²) red visible light in the 650–670 nm band for 3 minutes twice daily. It is thought to increase choroidal thickness and suppress axial elongation through photobiomodulation 3). Currently, it is not approved in Japan and is provided as self-funded treatment under the management of a specialist.
2. Main Symptoms and Clinical Findings
Section titled “2. Main Symptoms and Clinical Findings”Indicators of Treatment Efficacy
Section titled “Indicators of Treatment Efficacy”The efficacy of RLRL therapy is evaluated using the following objective indicators.
- Axial length: Measured using an optical biometer. Changes are tracked before treatment and every 6 months.
- Choroidal thickness: Measurement of subfoveal choroidal thickness (SFCT) using OCT. An increase in choroidal thickness is observed after RLRL irradiation. A 2-year follow-up study by Dong et al. (2023) confirmed an increase in choroidal thickness 2).
- Spherical equivalent refractive error: Objective assessment under cycloplegia.
- Corrected visual acuity: Confirmation of stability of corrected visual acuity before and after treatment.
Safety Evaluation Precautions
Section titled “Safety Evaluation Precautions”RLRL therapy is considered to have few short-term adverse events due to its low intensity, but the following points should be regularly checked.
- Risk of retinal toxicity: Excessive irradiation intensity, duration, or frequency may theoretically cause retinal damage 6). Perform baseline OCT evaluation of retinal and choroidal layers before treatment and compare periodically.
- Changes in visual acuity and color vision: Check for any decrease in visual acuity or color vision abnormalities during treatment.
- Subjective symptoms: Pay attention to complaints such as glare, afterimages, or visual field abnormalities.
3. Causes and Risk Factors
Section titled “3. Causes and Risk Factors”Risk Factors for Myopia Progression
Section titled “Risk Factors for Myopia Progression”The following risk factors are involved in myopia progression, which is the target of RLRL therapy.
- Early onset (especially between ages 6 and 12)
- Both parents are myopic (especially if both are myopic)
- Long near-work time (more than 3 hours per day)
- Insufficient outdoor activity time (less than 1 hour per day)
- Asian ethnicity (highest prevalence in East Asians)
- Holden et al. (2016) predict that by 2050, about 50% of the world population will be myopic8), increasing the social importance of early intervention.
4. Diagnosis and Examination Methods
Section titled “4. Diagnosis and Examination Methods”Pre-treatment Evaluation
Section titled “Pre-treatment Evaluation”Before starting RLRL therapy, record baseline data and confirm indications through the following examinations.
| Examination Item | Purpose | Main Points to Confirm |
|---|---|---|
| Cycloplegic refraction | Objective assessment of myopia degree | Spherical equivalent and astigmatism |
| Axial length measurement | Baseline setting and progression monitoring | Measured by optical biometry |
| Fundus examination | Pre-treatment retinal and choroidal status | Assessment of high myopia-related changes |
| OCT (macula and optic nerve) | Establishing baseline for retinal toxicity | Retinal and choroidal layer thickness |
| Slit-lamp examination | Exclusion of anterior segment diseases | Active inflammation and cataract |
For cycloplegic examination, instillation of cycloplegic agents (e.g., Cyclogyl 1%) is essential in young children. Subjective refraction without cycloplegia may result in overestimation of myopic power.
Monitoring during treatment
Section titled “Monitoring during treatment”The following schedule is recommended to confirm treatment efficacy and safety.
| Timing | Content |
|---|---|
| 1 month | Check compliance with eye drops, subjective symptoms, corrected visual acuity |
| Every 3–6 months | Axial length measurement, cycloplegic refraction, OCT (choroidal thickness, retinal evaluation) |
| Every year | Dilated fundus examination, OCT angiography (optional) |
It is essential to record baseline values with cycloplegic refraction and optical biometry. Additionally, fundus examination and OCT should be performed to document the pre-treatment retinal and choroidal status, which will be used for safety monitoring during treatment. If high myopia is suspected, check for peripheral retinal degeneration or tears.
5. Standard treatment
Section titled “5. Standard treatment”Pre-treatment informed consent and eligibility assessment
Section titled “Pre-treatment informed consent and eligibility assessment”The basic considerations for selecting patients eligible for RLRL therapy are as follows.
Recommended indications:
- Ages 6 to 18 years (most studies target this age group)
- Spherical equivalent myopia of −0.5 D or more with confirmed progression
- Myopia confirmed by cycloplegic examination
- Guardians are able to provide appropriate management
Situations requiring careful consideration:
- Cases with a history or family history of retinal disease
- Photosensitive disorders (e.g., xeroderma pigmentosum, cutaneous photosensitivity)
- Use of photosensitizing medications
Situations where use should be avoided:
- Active retinal disease (e.g., retinal degeneration, neovascularization)
- Patients with a confirmed diagnosis of photosensitivity
Treatment Protocol
Section titled “Treatment Protocol”The standard treatment parameters adopted in major RCTs are shown below.
| Parameter | Standard Value | Notes |
|---|---|---|
| Wavelength | 650 nm (red visible light) | Some studies use 670 nm |
| Irradiance | 0.3–1 mW/cm² | Incoherent light, non-laser |
| Exposure time | 3 minutes/session | Twice daily (morning and evening) |
| Treatment frequency | 5 days/week (school days only) | — |
| Treatment duration | 12–24 months or more | Ongoing maintenance of effect required |
The devices used are mainly Chinese-made dedicated handheld devices (e.g., Suneye) that have been used in research. They are designed for self-irradiation by patients at home, but initial setup and periodic checks are performed by a specialist.
Results of key RCTs
Section titled “Results of key RCTs”In a 1-year RCT by Jiang et al. (2022), the RLRL group showed significant suppression of axial elongation at 0.10 mm/year (control group 0.38 mm/year) 1). A 2-year follow-up by Dong et al. (2023) showed sustained suppression of axial elongation and confirmed an increase in choroidal thickness 2).
In a multicenter RCT by Zeng et al. (2023), the 1-year spherical equivalent progression was −0.20 D in the treatment group (control group −0.71 D), suppressing myopia progression by approximately 72% 4).
Comparison of RLRL therapy with other myopia progression suppression methods:
| Intervention | Refractive suppression rate | Axial suppression rate | Key evidence |
|---|---|---|---|
| RLRL therapy | 72% (Zeng 2023) | 74% (Jiang 2022) | Multiple RCTs |
| Low-concentration atropine 0.05% | Up to 67% | — | LAMP study |
| Orthokeratology | — | 43% | Si 2015 meta-analysis |
| MiSight (multifocal CL) | 59% | 52% | Chamberlain 2019 |
| DIMS spectacles | 55–59% | 37–38% | Lam 2020 RCT |
However, direct comparison requires caution due to differences in study design, target age, and treatment duration 5). Low-concentration atropine eye drops (Rijusea® Mini 0.025%) are the only approved drug in Japan as of December 2024 9), and myopia management spectacle lenses (e.g., DIMS) 10) are also recommended by the Japanese Myopia Society guidelines. RLRL therapy is being positioned as an option to combine with these approved interventions.
Compared with data from the 3-year RCT of MiSight (multifocal CL) 11), the 2-year RCT of DIMS spectacles 12), meta-analysis of OK 13), and the RCT of OK + atropine combination 14), the short-term inhibition rate of RLRL therapy is favorable, but the major challenge is the lack of data beyond 5 years. The increasing trend of myopic maculopathy in Japanese individuals shown by the Hisayama Study 15) further supports the medical importance of myopia management interventions including RLRL therapy.
Combination with Other Therapies
Section titled “Combination with Other Therapies”- RLRL + low-concentration atropine: Since the mechanisms of action differ, additive or synergistic effects are expected, but verification in large-scale RCTs is ongoing 5).
- RLRL + orthokeratology: Reported as a combination selected for rapidly progressing cases.
- When monotherapy is insufficient, consider combination with other myopia progression control therapies.
Informed Consent
Section titled “Informed Consent”As of April 2026, RLRL therapy is not approved or covered by insurance in Japan and is provided as out-of-pocket treatment. It is necessary to fully explain the following to patients and guardians and obtain consent.
- This treatment is not approved by the Japanese regulatory authorities.
- Current evidence is mainly from 1-2 year observational studies, and long-term safety has not been established.
- There is a theoretical risk of retinal toxicity if irradiation parameters are deviated 6).
- This is not a “treatment” for myopia but “progression control.”
- Regular ophthalmological visits and safety monitoring are mandatory.
As of April 2026, RLRL therapy is not approved as a pharmaceutical or covered by insurance in Japan. It is offered as a self-funded treatment at some ophthalmology clinics. When visiting a clinic, it is important to thoroughly confirm with a specialist regarding indications, safety, and costs. Low-dose atropine eye drops (Rijusea® Mini 0.025%) were approved in December 2024 as the first domestic drug for myopia progression control and are an insured treatment option.
Position of RLRL in the mechanism of myopia progression control
Section titled “Position of RLRL in the mechanism of myopia progression control”While the main optical interventions for myopia progression control (orthokeratology, DIMS spectacles, multifocal CL) share the common mechanism of the “peripheral myopic defocus hypothesis,” RLRL therapy acts through a completely different pathway: photobiomodulation (PBM) → mitochondrial activation → improved choroidal blood flow → choroidal thickening → suppression of axial elongation. This different mechanism of action leads to expectations of synergistic effects when combined with other therapies 3).
Additionally, a unique feature of RLRL therapy is the observation of an acute physiological response: choroidal thickness increases immediately after treatment 2). This characteristic is not seen with other myopia progression control therapies, and measurement of choroidal thickness is considered useful as an immediate effect indicator of treatment.
6. Pathophysiology and detailed pathogenesis
Section titled “6. Pathophysiology and detailed pathogenesis”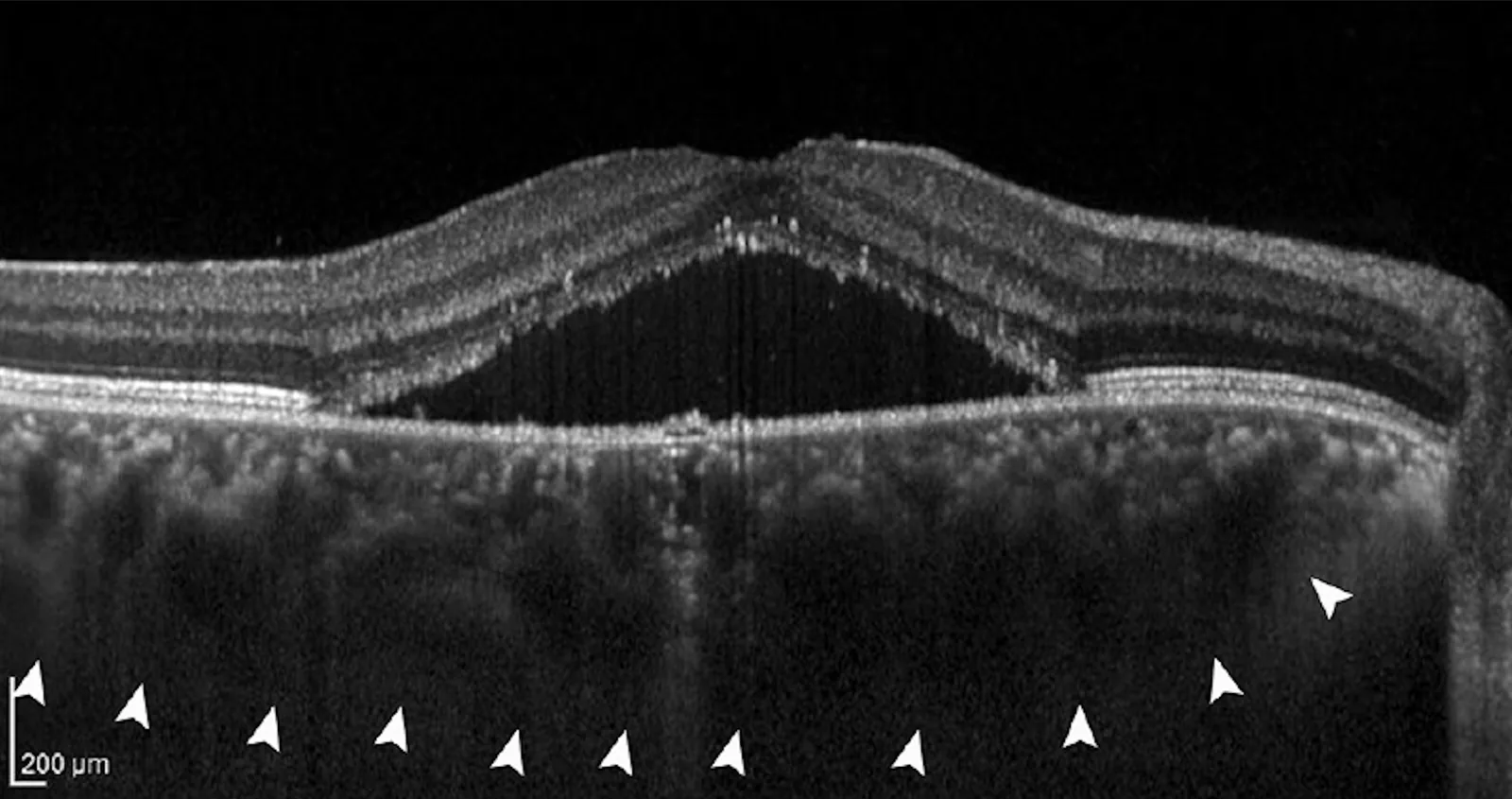
Photobiomodulation (PBM)
Section titled “Photobiomodulation (PBM)”The main mechanism of RLRL therapy is proposed to be photobiomodulation (PBM) 3). Red light in the 650–670 nm band is specifically absorbed by cytochrome c oxidase (Complex IV) in mitochondria, inducing the following biological responses.
- Promotion of ATP production
- Activation of cell signaling through moderate production of reactive oxygen species (ROS)
- Vasodilation and increased blood flow via nitric oxide (NO) release
- Improved choroidal circulation and thickening
Choroidal thickening and suppression of axial elongation
Section titled “Choroidal thickening and suppression of axial elongation”Consistent observations have shown that choroidal thickness increases after RLRL irradiation2)3). This increase in choroidal thickness is thought to affect the rigidity of the eye wall and contribute to suppressing axial elongation. Increases in choroidal thickness have also been reported with orthokeratology and low-concentration atropine, drawing attention to the possibility that the choroid may be a common pathway for myopia progression control.
Basic mechanisms of myopia progression
Section titled “Basic mechanisms of myopia progression”Myopia progression is a biological process in which eye growth is regulated by signal transduction from the retina to the sclera, involving the following steps:
- Peripheral hyperopic defocus serves as a driving signal for axial elongation
- Dopamine-mediated growth-inhibitory signals brake axial elongation
- Extracellular matrix remodeling of the sclera determines axial length
- Choroidal thickening and thinning serve as biomarkers for axial length changes
RLRL therapy intervenes in these signaling pathways and brakes eye growth at the choroidal and scleral levels, but the full picture remains largely unclear3). The quantitative risk of myopia complications shown in the meta-analysis by Haarman et al. (2020) is widely cited as the medical rationale for therapeutic intervention7).
7. Latest research and future perspectives
Section titled “7. Latest research and future perspectives”Establishment of long-term efficacy and safety data
Section titled “Establishment of long-term efficacy and safety data”Current major RCTs have observation periods of 1–2 years, and long-term data over 5 years are scarce1)2). The persistence of effect after treatment discontinuation (carryover effect) is also not established, and studies tracking changes in myopia progression rate after treatment cessation are ongoing.
In the 2-year follow-up study by Dong et al. (2023), the RLRL treatment group showed sustained axial elongation suppression and choroidal thickening compared to the control group, and the effect was maintained with continuous use for 1 year2). However, caution is needed in direct comparisons because study designs, irradiation parameters, and target ages differ between studies.
Quantification of retinal toxicity risk
Section titled “Quantification of retinal toxicity risk”Theoretically, excessive irradiation intensity, duration, or frequency could cause retinal damage. In the risk-benefit assessment of myopia management by Bullimore et al. (2021), the overall safety balance of optical and pharmacological interventions including RLRL therapy is discussed, emphasizing the importance of appropriate parameter management6).
Studies aimed at setting safe irradiation limits and risk assessment are being conducted. In clinical practice, it is necessary to strictly adhere to irradiation intensity (0.3–1 mW/cm²), duration (3 minutes/session), and frequency (2 times/day, 5 days/week), and to perform regular calibration checks of the device.
From the perspective of phototoxicity, cumulative light exposure to photoreceptors and RPE is important, and caution is needed with irradiation exceeding recommended levels (especially high-intensity, prolonged off-label use). As indicated by the meta-analysis of myopia complications by Haarman et al. (2020)7), it is important to individually assess the risk-benefit of RLRL therapy under the premise that appropriate myopia management can reduce the risk of serious complications.
Direct comparison with low-concentration atropine and orthokeratology
Section titled “Direct comparison with low-concentration atropine and orthokeratology”Direct comparison RCTs of RLRL therapy with orthokeratology and low-concentration atropine are being conducted. Establishment of personalized myopia management protocols considering the advantages and disadvantages of each therapy is expected5).
Comparison of characteristics of the three major interventions (RLRL, OK, low-concentration atropine):
| Intervention | Axial length suppression rate | Invasiveness | Convenience | Regulatory status in Japan |
|---|---|---|---|---|
| RLRL therapy | Approximately 60–72% | Non-invasive | Can be performed at home | Not approved (out-of-pocket) |
| Orthokeratology | 30–50% | Contact lens wear | Overnight wear | Not approved (out-of-pocket) |
| Low-concentration atropine 0.025% | Approximately 62% | Eye drops | Instilled before bedtime | Approved |
AI-Powered Prediction of Myopia Progression
Section titled “AI-Powered Prediction of Myopia Progression”Models that integrate axial length data, risk factors, and treatment response using AI to propose optimal treatment strategies for each patient are under development. They may also be applied to predict responders to RLRL therapy.
Trends Toward Domestic Approval
Section titled “Trends Toward Domestic Approval”With accumulating evidence from China and Southeast Asia, dialogue with regulatory authorities in Japan may begin. The progress of domestic multicenter RCTs and regulatory approval is attracting attention. Against the backdrop of the global increase in myopia (Holden 2016: 4.9 billion by 20508)), RLRL therapy is becoming established as one of the options for myopia management.
8. References
Section titled “8. References”- Jiang Y, Zhu Z, Tan X, et al. Effect of repeated low-level red-light therapy for myopia control in children: a multicenter randomized controlled trial. Ophthalmology. 2022;129:509-519.
- Dong J, Zhu Z, Xu H, et al. Myopia control effect of repeated low-level red-light therapy in Chinese children: a randomized, double-blind, controlled clinical trial. Ophthalmology. 2023;130:198-204.
- Wang W, Jiang Y, Zhu Z, et al. Chorioidal thickening as a potential therapeutic target in myopia management: a review of repeated low-level red light therapy. Surv Ophthalmol. 2023;68:1-12.
- Zeng J, Liu Y, Wang Z, et al. Efficacy of repeated low-level red-light stimulation on slowing myopia progression and ocular growth in children with myopia: a randomized controlled trial. Transl Vis Sci Technol. 2023;12:22.
- Yam JC, Zhang XJ, Zhang Y, et al. Interventions to reduce incidence and progression of myopia in children and adults. Prog Retin Eye Res. 2025.
- Bullimore MA, Ritchey ER, Shah S, et al. The risks and benefits of myopia control. Ophthalmology. 2021;128:1561-1579.
- Haarman AEG, Enthoven CA, Tideman JWL, et al. The complications of myopia: a review and meta-analysis. Invest Ophthalmol Vis Sci. 2020;61:49.
- Holden BA, Fricke TR, Wilson DA, et al. Global prevalence of myopia and high myopia and temporal trends from 2000 through 2050. Ophthalmology. 2016;123:1036-1042.
- 低濃度アトロピン点眼液を用いた近視進行抑制治療の治療指針作成委員会. 低濃度アトロピン点眼液を用いた近視進行抑制治療の手引き. 日眼会誌. 2025;129(10):851-854.
- 近視管理用眼鏡ガイドライン作成委員会. 近視管理用眼鏡(多分割レンズ)ガイドライン(第1版). 日眼会誌. 2025;129(10):855-860.
- Chamberlain P, Peixoto-de-Matos SC, Logan NS, et al. A 3-year randomized clinical trial of MiSight lenses for myopia control. Optom Vis Sci. 2019;96:556-567.
- Lam CSY, Tang WC, Tse DY, et al. Defocus incorporated multiple segments (DIMS) spectacle lenses slow myopia progression: a 2-year randomised clinical trial. Br J Ophthalmol. 2020;104:363-368.
- Si JK, Tang K, Bi HS, et al. Orthokeratology for myopia control: a meta-analysis. Optom Vis Sci. 2015;92:252-257.
- Kinoshita N, Konno Y, Hamada N, et al. Efficacy of combined orthokeratology and 0.01% atropine solution for slowing axial elongation in children with myopia: a 2-year randomized trial. Sci Rep. 2020;10:12750.
- Ueda E, Yasuda M, Fujiwara K, et al. Trends in the prevalence of myopia and myopic maculopathy in a Japanese population: the Hisayama Study. Invest Ophthalmol Vis Sci. 2019;60:2781-2786.
