Seasonal and Perennial AC

Allergic Conjunctivitis (Including Hay Fever)
Key Points at a Glance
Section titled “Key Points at a Glance”1. What Is Allergic Conjunctivitis?
Section titled “1. What Is Allergic Conjunctivitis?”Allergic conjunctival disease (ACD) is defined as “an inflammatory disease of the conjunctiva primarily involving a type I allergic reaction, accompanied by subjective symptoms and objective findings triggered by antigens”1). A mere allergic predisposition is not sufficient for diagnosis; ACD is diagnosed only when subjective symptoms such as itching, foreign body sensation, eye discharge, and tearing are present together with inflammatory changes of the conjunctiva.
The Japanese Society of Ophthalmic Allergy’s Clinical Practice Guidelines for Allergic Conjunctival Disease, Third Edition (hereafter referred to as the Guidelines) classifies disease subtypes based on the presence or absence of proliferative conjunctival changes (papillary hypertrophy of the palpebral conjunctiva, giant papillae, swelling or gelatinous limbal elevation), complication with atopic dermatitis, and mechanical irritation from foreign bodies1).
- Allergic conjunctivitis (AC): A subtype without proliferative conjunctival changes. Seasonal cases are called seasonal allergic conjunctivitis (SAC), and perennial cases are called perennial allergic conjunctivitis (PAC). Among SAC, cases caused by pollen are also called “pollen conjunctivitis,” with major regional pollens such as Japanese cedar pollen serving as representative antigens1).
- Atopic keratoconjunctivitis (AKC): A chronic ACD complicating facial atopic dermatitis. It is often accompanied by conjunctival fibrosis and corneal neovascularization or opacification1). The first report is attributed to Hogan et al., who described five cases of atopic eczema with bilateral keratoconjunctivitis in 19522).
- Vernal keratoconjunctivitis (VKC): A severe type with proliferative conjunctival changes. It may be accompanied by various corneal lesions including corneal epithelial disorders, persistent corneal epithelial defects, shield ulcers, and corneal plaques1).
- Giant papillary conjunctivitis (GPC): A subtype caused by mechanical irritation from contact lenses, artificial eyes, or surgical sutures, with proliferative changes in the upper palpebral conjunctiva. In the Guidelines, it is positioned as the most severe form of contact lens-related papillary conjunctivitis, with papillae measuring 1 mm or more in diameter1).
Epidemiology in Japan
Section titled “Epidemiology in Japan”Nationwide surveys led by ophthalmologists include the 1993–1995 Japan Ophthalmologists Association Allergic Eye Disease Survey, the 1993 Ministry of Health and Welfare field survey, and the 2017 Japanese Society of Ophthalmic Allergy prevalence survey1, 4). The 2017 prevalence survey reported the prevalence, subtype distribution, and age distribution as follows.
| Indicator | Survey result |
|---|---|
| Overall ACD prevalence | 48.7% (marked increase from 15–20% in 1993) |
| SAC due to Japanese cedar and cypress | 37.4% |
| Perennial allergic conjunctivitis (PAC) | 14.0% |
| SAC due to allergens other than Japanese cedar and cypress | 8.0% |
| Atopic keratoconjunctivitis (AKC) | 5.3% |
| Vernal keratoconjunctivitis (VKC) | 1.2% |
| Giant papillary conjunctivitis (GPC) | 0.6% |
| Age distribution of all ACD | Peak in 40s, secondary peak in teens |
| Regional variation of SAC | Higher in the Tokyo metropolitan area and Chubu region |
Source: Japanese Society of Ophthalmic Allergy Clinical Guideline Committee “Clinical Guidelines for Allergic Conjunctival Diseases, 3rd Edition” 1) and Okamoto et al. “2017 Japanese Society of Ophthalmic Allergy Survey of Allergic Conjunctival Diseases” 4).
PAC has a bimodal peak in the teens and 40s, while SAC prevalence increases with age from childhood. In SAC, the complication rate of allergic rhinitis is as high as 65–70% 1). AKC is estimated to occur concurrently in 25–40% of patients with atopic dermatitis 5).
The 3rd edition of the Japanese Society of Ophthalmic Allergy clinical guidelines classifies allergic conjunctival diseases into four clinical types based on the presence or absence of conjunctival proliferative changes, concurrent atopic dermatitis, and mechanical stimulation. Cases without proliferative changes are classified as allergic conjunctivitis (subdivided into seasonal SAC and perennial PAC), those with facial atopic dermatitis as atopic keratoconjunctivitis (AKC), those with proliferative changes and severe corneal lesions as vernal keratoconjunctivitis (VKC), and those caused by mechanical stimulation such as contact lenses or ocular prostheses as giant papillary conjunctivitis (GPC).
2. Main Symptoms and Clinical Findings
Section titled “2. Main Symptoms and Clinical Findings”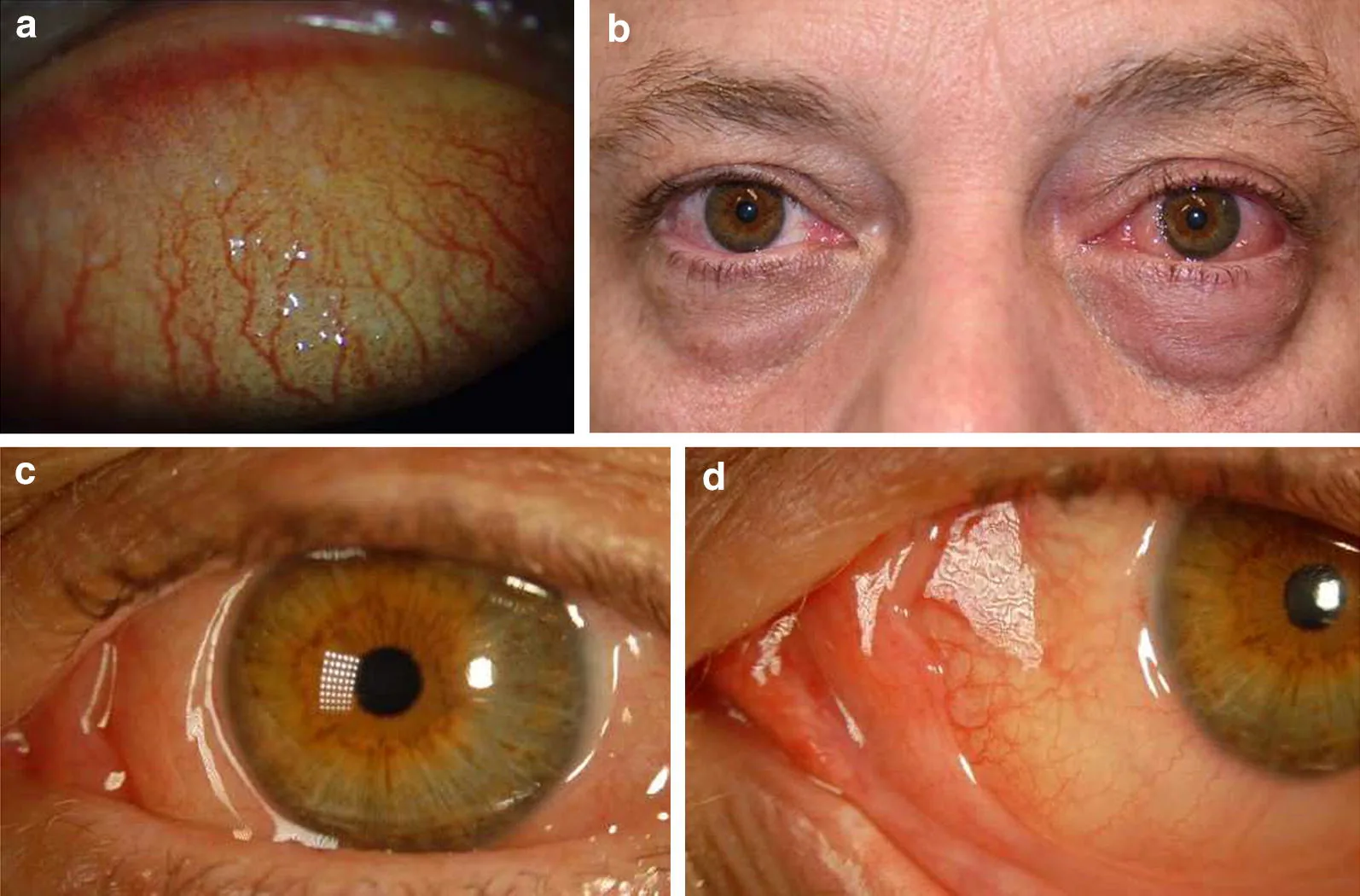
Subjective Symptoms
Section titled “Subjective Symptoms”The typical subjective symptoms of ACD are itching, foreign body sensation, and eye discharge, among which ocular itching is the most specific symptom 1).
- Ocular itching: The most characteristic symptom of ACD. It occurs when histamine released from mast cells stimulates sensory nerve endings. In children and the elderly, it may not be reported as itching but rather as a foreign body sensation or discomfort 1).
- Foreign body sensation: Described as a “gritty” feeling. It is often caused by numerous conjunctival papillae coming into contact with the cornea during blinking 1).
- Eye discharge: Primarily serous or mucous, characterized by white or translucent stringy, viscous discharge. Its properties differ from neutrophil-dominant bacterial discharge 1). In VKC, yellow viscous discharge may be observed.
- Tearing and injection: High frequency but low specificity.
- Eye pain, photophobia, and blurred vision: Observed when corneal lesions are present, correlating with disease severity 1).
- GPC-specific complaints: Starting with mild foreign body sensation during contact lens wear, patients often sequentially notice lens displacement, lens soiling, increased discharge, and blurred vision.
Clinical Findings and Severity Assessment
Section titled “Clinical Findings and Severity Assessment”This guideline establishes severity assessment criteria for each site: palpebral conjunctiva, bulbar conjunctiva, limbus, and cornea 1). Representative items are shown below.
| Site | Item | Severity Criteria |
|---|---|---|
| Palpebral conjunctiva | Hyperemia | Mild: a few vessels; Moderate: many vessels; Severe: vessels indistinguishable |
| Palpebral conjunctiva | Papillae | Mild 0.1–0.2 mm, moderate 0.3–0.5 mm, severe ≥0.6 mm |
| Palpebral conjunctiva | Giant papillae | ≥1 mm in diameter; moderate if occupying <1/2 of the upper tarsal conjunctiva, severe if ≥1/2 |
| Bulbar conjunctiva | Edema | Partial = mild, diffuse = moderate, vesicular = severe |
| Limbus | Trantas dots | 1–4 = mild, 5–8 = moderate, ≥9 = severe |
| Cornea | Epithelial damage | Superficial punctate keratitis → exfoliative superficial punctate keratitis → shield ulcer |
Source: Created based on Table 2-2 of this guideline1).
Characteristic Findings by Type
Section titled “Characteristic Findings by Type”Vernal Keratoconjunctivitis, Palpebral Type
Cobblestone giant papillae: Hard, cobblestone-like papillae lined up on the upper tarsal conjunctiva1).
Mucoid discharge: Copious stringy, mucoid discharge.
Corneal complications: Superficial punctate keratitis, exfoliative superficial punctate keratitis, shield ulcer, and corneal plaque1, 14).
Vernal Keratoconjunctivitis, Limbal Type
Gelatinous limbal elevation: Gelatinous elevations at the corneal limbus.
Trantas dots: Small white elevations composed of degenerated conjunctival epithelium and eosinophil aggregates1).
Pseudogerontoxon: Arcus senilis-like opacity remaining after severe inflammation of the limbal type1).
Atopic Keratoconjunctivitis
Eczematous blepharitis: Continuous with facial atopic dermatitis. Accompanied by eyelid thickening, trichiasis, and madarosis.
Conjunctival pigmentation and symblepharon: Causes conjunctival sac shortening and symblepharon over a long course1, 12).
Corneal complications: May progress from superficial punctate keratitis to persistent epithelial defects and corneal ulcers.
Giant Papillary Conjunctivitis
Characteristic papillae: Round, well-demarcated, smooth-surfaced, non-confluent, and low elevation. Morphologically distinct from the giant papillae of vernal keratoconjunctivitis1).
Corneal complications are rare: Mechanical irritation is the main cause, and inflammation is often mild.
Low IgE positivity rate: The involvement of type I allergy is not always evident1).
3. Causes and Risk Factors
Section titled “3. Causes and Risk Factors”Causative Allergens
Section titled “Causative Allergens”The distribution of causative allergens differs significantly by disease type. These guidelines list the “PAC set,” which includes mites, house dust, alder, Japanese cedar, cypress, sweet vernal grass, orchard grass, mugwort, ragweed, Candida, Alternaria, cat epithelium, and dog epithelium, as items covered by insurance for serum allergen-specific IgE antibody testing1).
| Disease Type | Main Causative Allergens | Seasonality |
|---|---|---|
| Seasonal Allergic Conjunctivitis (SAC) | Japanese cedar, cypress (spring); orchard grass, timothy grass, etc. (early summer); ragweed, mugwort, etc. (autumn) | Yes |
| Perennial Allergic Conjunctivitis (PAC) | Mites, house dust, mold, pet dander | No |
| Atopic Keratoconjunctivitis (AKC) | Polysensitization (mites, house dust, pollen, food, etc.), atopic predisposition | Chronic |
| Vernal Keratoconjunctivitis (VKC) | House dust and mites are common, but reacts to multiple antigens including pollen and animal dander | Exacerbations present |
| Giant Papillary Conjunctivitis (GPC) | Contact lens material, lens deposits, ocular prostheses, exposed sutures | None |
Risk Factors and Background
Section titled “Risk Factors and Background”- Atopic diathesis and family history: A family history of bronchial asthma, allergic rhinitis, or atopic dermatitis increases the risk. In AKC, skin barrier dysfunction including filaggrin gene abnormalities is involved5).
- Urban living and environmental factors: Air pollution, smoking, and dry environments can be exacerbating factors for ACD6).
- Dupilumab: Dupilumab, an IL-4 receptor alpha chain antibody used for treating severe atopic dermatitis, has been reported to have a risk ratio of 2.64 for concurrent conjunctivitis, and caution is also required during the treatment course of AKC1).
- Eye rubbing habit: Chronic eye rubbing due to itching is a risk factor for keratoconus and atopic cataract5, 12). In AKC in particular, chronic mechanical stimulation is noted to promote weakening of corneal structure and has been associated with early-onset keratoconus5).
The Japanese Society of Ophthalmic Allergy guidelines recommend wearing goggles-style or regular glasses, discontinuing contact lens use and switching to glasses if possible, rinsing with preservative-free artificial tears (tap water should not be used as it damages the corneal epithelium), wearing a smooth-fabric jacket when going out and removing it at the entrance, and washing the face and gargling after returning home. Furthermore, “initiation therapy”—starting anti-allergic eye drops approximately two weeks before the predicted pollen dispersal date—is effective.
4. Diagnosis and Testing
Section titled “4. Diagnosis and Testing”Diagnostic Criteria (This Guideline)
Section titled “Diagnostic Criteria (This Guideline)”Diagnosis is made in three stages based on three elements: clinical symptoms, type I allergic predisposition, and local type I allergic reaction in the eye1).
- Clinical diagnosis (A only): Clinical symptoms characteristic of ACD are present.
- Clinically confirmed diagnosis (A+B): In addition to clinical symptoms, positive tear total IgE antibody, positive serum antigen-specific IgE antibody, or a positive skin reaction matching the suspected antigen.
- Confirmed diagnosis (A+B+C, or A+C): In addition to the above, eosinophils in conjunctival scrapings are positive.
A: Clinical symptoms present / B: Type I allergic predisposition present / C: Type I allergic reaction in the conjunctiva present.
This guideline categorizes subjective symptoms and objective findings by “specificity: high/medium/low,” and severe ocular itching, giant papillae, limbal proliferation, and shield ulcer are considered the findings with the highest specificity1).
Local Ocular Examination
Section titled “Local Ocular Examination”- Eosinophil test in conjunctival scrapings: After topical anesthetic instillation, evert the upper eyelid, gently massage the palpebral conjunctiva with a glass rod, collect mucus with a spatula, and smear it onto a slide. Perform Hansel or Giemsa staining, and if even one eosinophil is identified under light microscopy, the result is considered positive1, 3).
- Tear total IgE antibody test (Allerwatch®): A rapid diagnostic kit from Wakamoto Pharmaceutical / Minaris Medical that tests tear samples using immunochromatography. It has been reported to have a sensitivity of 73.6% and specificity of 100% for ACD1). Positive rates by disease type are reported as SAC 61.9%, PAC 65.4%, AKC 80.5%, VKC 94.7%, GPC 75.0%, with an overall rate of 72.2%3).
- Conjunctival provocation test: A method that involves instilling a known antigen solution to confirm the induction of conjunctivitis symptoms; however, it is not covered by insurance in Japan and has not been standardized1).
Systemic allergic predisposition testing
Section titled “Systemic allergic predisposition testing”- Serum antigen-specific IgE antibody testing: Select items mainly from the PAC set, including dust mites, house dust, Japanese cedar, Japanese cypress, orchard grass, ragweed, mugwort, and dog/cat epithelium. Up to 13 items are covered by insurance1, 3). For screening purposes, multi-item simultaneous measurement systems such as View Allergy 39 (Thermo Fisher Diagnostics) and MAST Immunosystems V (Minalis Medical) can also be used under insurance coverage3).
- Skin reaction (prick test / scratch test): A positive result is determined when the wheal diameter is 3 mm or more, or at least half the diameter of the positive control wheal. A positive control (to confirm absence of false negatives) and a negative control (to confirm absence of dermographism) are used together. Histamine H₁ receptor antagonists and tricyclic antidepressants can cause false negatives, so attention to the drug withdrawal period before testing is necessary3). Use with caution in patients with severe bronchial asthma, a history of anaphylaxis, or significant cardiovascular disease.
Diagnostic flowchart
Section titled “Diagnostic flowchart”In the clinical diagnostic flowchart of Figure 2-40 in this guideline, cases presenting with ocular itching and hyperemia are first stratified by the presence or absence of conjunctival proliferative changes. In the absence of proliferative changes, SAC/PAC is determined based on seasonality. When proliferative changes are present, GPC is considered if contact lenses are worn; if not, AKC/VKC is differentiated based on the presence or absence of atopic dermatitis1).
Differential diagnosis
Section titled “Differential diagnosis”The following diseases are listed as differential diagnoses in this guideline1).
- Viral conjunctivitis: Adenovirus (epidemic keratoconjunctivitis, pharyngoconjunctival fever), herpes simplex virus, varicella-zoster virus, enterovirus, SARS-CoV-2, etc. Acute onset, unilateral involvement, and preauricular lymphadenopathy are suggestive findings. For adenovirus, antigen is confirmed using a rapid diagnostic kit.
- Bacterial conjunctivitis: Causative organisms include Staphylococcus aureus, Streptococcus pneumoniae, Haemophilus influenzae, etc. It is differentiated by mucopurulent yellow to yellow-green discharge and polymorphonuclear leukocytes in conjunctival scrapings.
- Chlamydial conjunctivitis: In adults, it presents as unilateral acute follicular conjunctivitis, characterized by giant follicles in the lower tarsal conjunctival fornix.
- Conjunctival folliculosis: Millet-sized follicles seen in the lower tarsal conjunctival fornix of children, asymptomatic and of little pathological significance.
- Dry eye disease: Accompanied by shortened BUT and often coexists with ACD1).
When ocular itching is severe, the condition is bilateral, and the discharge is predominantly serous or mucoid white, with allergic predisposition (comorbid allergic rhinitis or dermatitis, family history), allergic conjunctivitis is suspected. On the other hand, when the onset is acute and unilateral, with mucopurulent or yellow discharge, preauricular lymphadenopathy, fever, or sore throat, infectious conjunctivitis should be considered, and adenovirus rapid testing or conjunctival scraping examination should be performed. The diagnostic criteria for both are detailed in the 3rd edition of the Japanese Society of Ophthalmic Allergy guidelines.
5. Standard treatment
Section titled “5. Standard treatment”According to this guideline, the first-line treatment for all disease types is anti-allergic eye drops, with the strategy of adding steroid eye drops or immunosuppressive eye drops depending on severity1). For refractory severe cases, oral steroids, subconjunctival steroid injection, and surgical treatments such as conjunctival papillectomy are also options.
Anti-allergic eye drops
Section titled “Anti-allergic eye drops”Anti-allergic eye drops are broadly classified into two categories based on their mechanism of action1, 13).
- Mediator release inhibitors: Inhibit mast cell degranulation and suppress the release of histamine, leukotrienes, thromboxane A₂, etc., thereby reducing the immediate-phase response of type I allergic reactions. They also act on the late phase by suppressing local conjunctival infiltration of inflammatory cells.
- Histamine H₁ receptor antagonists: Competitively inhibit the binding of histamine released from mast cells to H₁ receptors, suppressing hyperemia and ocular itching. They are reported to have a faster onset of action for itching compared to mediator release inhibitors.
Representative drugs are summarized in the table below.
| Classification | Generic name | Representative brand name | Concentration and dosage |
|---|---|---|---|
| Mediator release inhibitor | Pemirolast potassium | Alegysal ophthalmic solution | 0.1% Twice daily |
| Mediator release inhibitors | Tranilast | Rizaben ophthalmic solution | 0.5% 4 times daily |
| Mediator release inhibitors | Ibudilast | Ketas ophthalmic solution | 0.01% 4 times daily |
| Mediator release inhibitors | Acitazanolast hydrate | Zepelin ophthalmic solution | 0.1% 4 times daily |
| H₁ receptor antagonists | Ketotifen fumarate | Zaditen ophthalmic solution | 0.05% 4 times daily |
| H₁ receptor antagonists | Levocabastine hydrochloride | Livostin ophthalmic solution | 0.025% 4 times daily |
| H₁ receptor antagonists | Olopatadine hydrochloride | Patanol ophthalmic solution | 0.1% 4 times daily |
| H₁ receptor antagonists | Epinastine hydrochloride | Alesion ophthalmic solution / Alesion LX ophthalmic solution | 0.05% 4 times daily / 0.1% 2 times daily |
Source: Table 2-6 of this guideline1).
Ketotifen, olopatadine, and epinastine have been shown to have mediator release inhibitory effects in vitro in addition to their histamine H₁ receptor antagonist activity. Alesion LX does not contain preservatives, making it easier to prescribe for contact lens wearers.
Initial Therapy (Seasonal Allergic Conjunctivitis)
Section titled “Initial Therapy (Seasonal Allergic Conjunctivitis)”This guideline recommends initial therapy, starting anti-allergic eye drops approximately two weeks before the predicted pollen dispersal date or at the first appearance of any symptoms1). This approach reduces symptoms during the peak pollen season, and some reports indicate it suppresses the onset of hay fever symptoms altogether in approximately 30% of patients. In perennial allergic conjunctivitis as well, mediator release inhibitors are effective for maintaining mast cell membrane stabilization throughout the year.
Treatment Strategies by Disease Type
Section titled “Treatment Strategies by Disease Type”1) Seasonal and Perennial Allergic Conjunctivitis (SAC / PAC)
Section titled “1) Seasonal and Perennial Allergic Conjunctivitis (SAC / PAC)”- First-line: Anti-allergic eye drops (mediator release inhibitors or H₁ antagonists)1).
- During periods of severe symptoms or when accompanied by rhinitis: Oral anti-allergic medications, with NSAID eye drops as needed1).
- If control cannot be achieved with anti-allergic eye drops: add steroid eye drops for short-term use. In CQ1 of this guideline, the use of steroid eye drops for SAC/PAC is weakly recommended1). Steroid eye drops should not be used as first-line therapy and should only be used short-term with regular intraocular pressure monitoring.
- For contact lens wearers or patients with concurrent dry eye, choose preservative-free formulations.
2) Atopic Keratoconjunctivitis (AKC)
Section titled “2) Atopic Keratoconjunctivitis (AKC)”- First-line treatment is anti-allergic eye drops. In cases with insufficient response, combine with steroid eye drops or immunosuppressive eye drops1).
- In CQ3 of this guideline, the use of steroid eye drops for AKC is strongly recommended1).
- CQ7 strongly recommends the use of tacrolimus eye drops for AKC (however, insurance coverage in Japan is limited to vernal keratoconjunctivitis only)1, 8). Cyclosporine eye drops are also conditionally recommended for AKC1).
- Treatment of atopic blepharitis must be performed concurrently, and collaboration with a dermatologist or allergist is essential5).
- When prescribing oral steroids, collaborate with an internist or dermatologist1).
- In cases receiving systemic treatment including biologic agents such as dupilumab or oral cyclosporine, be aware of the risk ratio of 2.64 for developing dupilumab-related conjunctivitis1).
3) Vernal Keratoconjunctivitis (VKC)
Section titled “3) Vernal Keratoconjunctivitis (VKC)”Treatment is escalated in stages according to the treatment flowchart in this guideline (Figure 2-42)1, 7).
- First, prescribe anti-allergic eye drops as the foundation.
- For moderate to severe cases, add immunosuppressive eye drops. Tacrolimus eye drops are strongly recommended in CQ7 (evidence-based efficacy established), and cyclosporine eye drops are conditionally recommended in CQ41, 8, 9).
- In severe cases where improvement is not achieved with the two agents, additionally administer steroid eye drops (CQ2: strongly recommended)1).
- Cyclosporine plus steroid combination for proliferative changes (CQ6) and tacrolimus plus steroid combination for severe cases (CQ9) are both conditionally recommended1).
- In the most severe cases not controlled by eye drops, consider oral steroids, subconjunctival steroid injection, conjunctival papillae resection, and corneal plaque debridement1). Triamcinolone acetonide or betamethasone sodium phosphate suspension is used for subconjunctival injection, but care should be taken to monitor intraocular pressure elevation, and repeated use or administration in children under 10 years of age should be avoided1).
- After symptom improvement, switch steroid eye drops to a lower potency followed by tapering and discontinuation, and maintain control with a regimen of anti-allergic eye drops and immunosuppressive eye drops.
Proactive therapy: Once a longer remission period is achieved, gradually reduce the immunosuppressive eye drops from twice daily to once daily to twice weekly, and ultimately continue with a small maintenance dose. Adjust the dosage according to symptoms to prevent recurrence1).
4) Giant Papillary Conjunctivitis (GPC)
Section titled “4) Giant Papillary Conjunctivitis (GPC)”- Removing the cause is of utmost importance. If contact lenses are the cause, discontinue use in principle. When resuming use, consider switching to daily disposable soft contact lenses, changing lens material or design, providing instructions on rubbing and cleaning, and changing care products1).
- First-line treatment is anti-allergic eye drops (mainly mediator release inhibitors); in severe cases, short-term concomitant use of steroid eye drops (e.g., 0.1% fluorometholone 4 times daily). Monitor intraocular pressure during eye drop treatment.
- If an ocular prosthesis is the cause, consider replacing or changing the type of prosthesis1).
Immunosuppressive Eye Drops (Covered by Insurance for VKC)
Section titled “Immunosuppressive Eye Drops (Covered by Insurance for VKC)”Cyclosporine Eye Drops
Brand name: Papilock Mini Ophthalmic Solution 0.1%1)
Dosage: 3 times daily
Characteristics: An immunosuppressant that inhibits the calcineurin pathway in T cells. Concomitant use with anti-allergic eye drops and steroid eye drops enables steroid tapering1, 9). Effective for limbal VKC. Onset of effect is somewhat delayed.
Recommendation in this guideline: Weakly recommended for VKC in CQ4, and conditionally recommended for concomitant use in conjunctival proliferative changes in CQ61).
Tacrolimus Eye Drops
Brand name: Talymus Ophthalmic Solution 0.1%1)
Dosage: 2 times daily
Characteristics: Has a stronger immunosuppressive effect than cyclosporine, and is effective as monotherapy even in steroid-resistant severe cases1, 8). Also useful in cases complicated by atopic dermatitis.
Recommendation in this guideline: Strongly recommended for VKC and AKC in CQ7 (AKC is not covered by insurance), and conditionally recommended for concomitant use in severe conjunctival proliferative changes in CQ91).
Both agents may cause stinging or burning sensation upon instillation, and attention should be paid to the development of infections including herpetic keratitis during use. Both children and adults have indications for VKC1).
Other Precautions
Section titled “Other Precautions”Surgical Treatment for AKC Complications
Section titled “Surgical Treatment for AKC Complications”Complications arising during the course of AKC may each require surgical treatment5).
- Amniotic membrane transplantation: Performed for persistent corneal epithelial defects.
- Corneal transplantation: Performed for corneal opacity, severe ulcers, and thinning. However, rejection rates are high in AKC patients, and strict management of inflammation is required postoperatively.
- Cataract surgery: Performed for atopic cataract presenting with anterior subcapsular and posterior subcapsular opacities.
- Eyelid surgery: Correction of trichiasis, ectropion, and entropion.
The third edition of this guideline explains that in addition to type I allergic reactions, Th2 cells (type 2 helper T cells) are deeply involved in the pathogenesis of vernal keratoconjunctivitis and atopic keratoconjunctivitis. There are cases that are difficult to control with conventional anti-allergic eye drops alone, which cannot regulate T cells. Immunosuppressive eye drops such as cyclosporine and tacrolimus suppress T cell activation and improve eosinophil infiltration, proliferative changes, and corneal epithelial disorders. In CQ7, the use of tacrolimus eye drops is “strongly recommended.”
This guideline recommends that steroid eye drops be used short-term in combination with anti-allergic eye drops only for cases that cannot be controlled with anti-allergic eye drops alone, and states that they should not be first-line treatment. There are risks of increased intraocular pressure, cataract formation, and infection induction; especially in children, the frequency of increased intraocular pressure is high, so intraocular pressure must be measured regularly during use. In vernal keratoconjunctivitis, steroids can be tapered by combining with immunosuppressive eye drops, so the principle is to avoid long-term continuous use of steroids alone.
6. Pathophysiology and Detailed Pathogenesis
Section titled “6. Pathophysiology and Detailed Pathogenesis”Immediate and Late Phases of Type I Allergy
Section titled “Immediate and Late Phases of Type I Allergy”The basic pathogenesis of allergic conjunctivitis is a type I (immediate) allergic reaction.
Immediate phase (within approximately 15 minutes after antigen exposure): When allergens reaching the conjunctiva cross-link with IgE antibodies on the surface of sensitized conjunctival mast cells, the mast cells undergo degranulation, releasing chemical mediators such as histamine, tryptase, leukotrienes, and prostaglandins all at once10, 11). The released histamine acts via H₁ receptors to cause capillary dilation, increased vascular permeability, sensory nerve stimulation, and increased mucus secretion, manifesting as ocular itching, conjunctival hyperemia, conjunctival edema, and tearing.
Late phase (several hours later): Expression of cytokines (IL-4, IL-5, IL-13, etc.) and adhesion molecules (ICAM-1, VCAM-1, etc.) leads to infiltration of eosinophils, neutrophils, lymphocytes, and basophils into the local conjunctival tissue10, 11). Tissue-damaging proteins released from eosinophils, such as major basic protein (MBP) and eosinophil cationic protein (ECP), damage the corneal and conjunctival epithelium, causing corneal lesions such as superficial punctate keratitis, persistent corneal epithelial defects, and shield ulcers14).
Th2 Cells and the Special Pathophysiology of VKC and AKC
Section titled “Th2 Cells and the Special Pathophysiology of VKC and AKC”While mild SAC/PAC are primarily based on the type I immediate phase, in vernal keratoconjunctivitis and atopic keratoconjunctivitis, Th2 cells (type 2 helper T cells) play a central role in pathogenesis1, 13). In animal models, type I allergic reactions alone do not induce strong conjunctival eosinophil infiltration; marked eosinophil infiltration is only induced when Th2 cells are involved. Histopathology of giant papillae in VKC shows eosinophil infiltration, fibroblast proliferation, extracellular matrix deposition, and numerous infiltrating T cells. Tear fluid eosinophil count has been reported to correlate with the severity of corneal damage13).
In the conjunctival tissue of AKC, in addition to type I hypersensitivity, type IV (delayed-type) hypersensitivity characterized by infiltration of T cells, macrophages, and dendritic cells is also involved. Histological features include goblet cell proliferation, eosinophil and mast cell infiltration into the epithelium, and mononuclear cell infiltration of the substantia propria; decreased corneal sensation and reduced conjunctival goblet cell density have also been reported5, 14).
Pollen Hatch-Out Mechanism
Section titled “Pollen Hatch-Out Mechanism”Cedar pollen has a large particle diameter and cannot directly pass through the conjunctival epithelium. However, when pollen absorbs tear fluid and ruptures (hatches out), antigen proteins (Cry j 1, Cry j 2, etc.) are eluted, pass through the conjunctival epithelium, reach deep mast cells, and initiate the allergic reaction. The reason artificial tears are effective for prevention through tear dilution and pollen washout is due to this mechanism.
Mechanism of Giant Papillary Conjunctivitis
Section titled “Mechanism of Giant Papillary Conjunctivitis”The involvement of type I allergy in GPC is not always clear, and the main causes are thought to be repeated mechanical stimulation from contact lenses, their deposits, or exposed sutures, as well as immune reactions to lens materials and deposited proteins1). Although infiltration of eosinophils, mast cells, and basophils is observed in the conjunctival tissue, the rates of serum antigen-specific IgE antibody positivity and eosinophil positivity are lower than in other disease types1).
Mechanism of AKC Complications
Section titled “Mechanism of AKC Complications”The pathogenesis of ocular complications characteristic of AKC is summarized as follows5, 12).
- Keratoconus: Repeated mechanical damage to the cornea from chronic eye rubbing and weakening of corneal structure due to chronic inflammation are involved.
- Atopic cataract: Characterized by anterior and posterior subcapsular opacities, developing as lens opacification independent of steroid use.
- Herpes simplex keratitis: Patients with AKC have a high risk of bilateral herpetic keratitis, requiring particular caution during immunosuppressive treatment.
- Retinal detachment: Reported to be more frequent in patients with atopic dermatitis, and an association with eye rubbing behavior has been suggested.
7. Recent Research and Future Perspectives
Section titled “7. Recent Research and Future Perspectives”Biologic Agents
Section titled “Biologic Agents”Omalizumab (anti-IgE monoclonal antibody) is a biologic agent used for bronchial asthma and chronic urticaria. International case reports and small observational studies have reported efficacy in refractory vernal keratoconjunctivitis and severe atopic keratoconjunctivitis. It is not covered by insurance for ocular diseases in Japan and is currently positioned as a research-stage option11).
JAK inhibitors (upadacitinib, etc.): By selectively inhibiting the JAK-STAT pathway, these agents suppress IL-4/IL-13 signaling and are being investigated for their potential to improve both skin and ocular symptoms while avoiding the conjunctivitis associated with dupilumab.
Eye Drops with Novel Mechanisms of Action
Section titled “Eye Drops with Novel Mechanisms of Action”Novel molecules that simultaneously inhibit multiple targets, such as an eye drop (rVA576) that concurrently blocks complement C5 and leukotriene B4, are in early-stage development, including Phase I clinical trials.
Primary Prevention and Lifestyle
Section titled “Primary Prevention and Lifestyle”Probiotics: Prenatal and postnatal probiotic intake has been reported to potentially reduce the incidence of atopic diseases by up to 30%, but conclusive evidence has not been established. Antibiotic use has also been suggested to increase the risk of atopic diseases through alterations in gut microbiota.
Breastfeeding: Breastfeeding, particularly during the first three months of life, has been suggested as a possible protective factor against atopic diseases. No evidence of benefit has been found for vitamin or mineral supplements.
Disclaimer
Section titled “Disclaimer”8. References
Section titled “8. References”- 日本眼科アレルギー学会診療ガイドライン作成委員会. アレルギー性結膜疾患診療ガイドライン(第3版). 日眼会誌. 2021;125(8):741-785. https://www.nichigan.or.jp/member/journal/guideline/detail.html?dispmid=909&itemid=429
- Hogan MJ. Atopic keratoconjunctivitis. Trans Am Ophthalmol Soc. 1952;50:265-281.
- 庄司純, ほか. アレルギー性結膜疾患診断における自覚症状、他覚所見および涙液総IgE検査キットの有用性の検討. 日眼会誌. 2012;116(6):485-493. CiNii: https://cir.nii.ac.jp/crid/1520290883483009152
- 岡本茂樹, ほか. 2017年度日本眼科アレルギー学会アレルギー性結膜疾患実態調査. 日眼会誌. 2022;126(7):625-635. https://journal.nichigan.or.jp/Disp?mag=0&number=7&start=625&style=abst&vol=126&year=2022
- Chen JJ, Applebaum DS, Sun GS, Pflugfelder SC. Atopic keratoconjunctivitis: A review. J Am Acad Dermatol. 2014;70(3):569-575.
- Leonardi A, Castegnaro A, Valerio AL, Lazzarini D. Epidemiology of allergic conjunctivitis: clinical appearance and treatment patterns in a population-based study. Curr Opin Allergy Clin Immunol. 2015;15(5):482-488.
- 福島敦樹. 春季カタルのパターン治療のためのプロトコール. あたらしい眼科. 2013;30(4):491-496.
- Ohashi Y, Ebihara N, Fujishima H, et al. A randomized, placebo-controlled clinical trial of tacrolimus ophthalmic suspension 0.1% in severe allergic conjunctivitis. J Ocul Pharmacol Ther. 2010;26(2):165-174.
- Takamura E, Uchio E, Ebihara N, et al. A prospective, observational, all-prescribed-patients study of cyclosporine 0.1% ophthalmic solution in the treatment of vernal keratoconjunctivitis. Nippon Ganka Gakkai Zasshi. 2011;115(6):508-515.
- Sánchez-García S, Habernau Mena A, Quirce S. Biomarkers in inflammatory allergic conjunctivitis: a systematic review for biomarkers. Clin Transl Allergy. 2019;9:27.
- Leonardi A, Bogacka E, Fauquert JL, et al. Ocular allergy: recognizing and diagnosing hypersensitivity disorders of the ocular surface. Allergy. 2012;67(11):1327-1337.
- 内尾英一. 免疫抑制点眼薬の使用法. 日眼会誌. 2015;119(3):136-141.
- Kumar S. Vernal keratoconjunctivitis: a major review. Acta Ophthalmol. 2009;87(2):133-147.
- Wakamatsu TH, Satake Y, Igarashi A, et al. IgE and eosinophil cationic protein (ECP) as markers of severity in the diagnosis of atopic keratoconjunctivitis. Br J Ophthalmol. 2012;96(8):1127-1131.
