Ocular surface evaluation
Location of filaments: Directly linked to estimating the underlying disease
Meibomian gland status: Check for obstruction or atrophy
Eyelid closure and opening: Assess for incomplete blink or lagophthalmos
Filamentary keratitis is a chronic, recurrent corneal disease in which filamentous structures (corneal filaments) adhere to the corneal surface. The filaments consist of degenerated corneal epithelial cells as a core, with mucin attached, and also include inflammatory cells and conjunctival epithelial cells as components.
This disease rarely occurs alone and almost always has underlying ocular surface, eyelid, or systemic diseases. Representative background diseases include aqueous-deficient dry eye, superior limbic keratoconjunctivitis (SLK), lagophthalmos, ptosis, neurotrophic keratopathy, post-ocular surgery, conjunctivochalasis, fixed strabismus, diabetes, and prolonged eyelid closure (after impaired consciousness or stroke).
Traditionally, the mechanism was thought to be impaired corneal wound healing, but in recent years, increased friction on the ocular surface has been placed at the center of the pathology. The Dry Eye Diagnostic Guidelines of the Japanese Dry Eye Society 1) positions filamentary keratitis together with lid-wiper epitheliopathy (LWE), SLK, and conjunctivochalasis as dry eye-related diseases with a common mechanism of increased friction during blinking, and a framework for understanding and treating them as a group has been proposed.
Epidemiologically, it often complicates moderate to severe aqueous-deficient dry eye and after corneal transplantation, and is frequent in patients with Sjögren’s syndrome 1). Although epidemiological data on prevalence in Japan are limited, the guidelines of the Japanese Ophthalmological Society emphasize that in refractory cases, attention should be paid to complications such as ptosis and entropion 1). In necrotizing keratitis due to herpes, epithelial instability based on stromal edema and trigeminal nerve palsy can lead to filamentous lesions in the upper cornea, and it is important that the onset involves multiple factors rather than a single cause.
Historically, it has long been described as an ocular surface disease with corneal filaments, and associations with keratoconjunctivitis sicca, lagophthalmos, and post-ocular surgery have been repeatedly reported. In Japan, since the 1980s, with advances in tear dynamics analysis, the association with aqueous-deficient dry eye became clear, and after the 2000s, immunohistochemical analysis of filament components and the concept of ocular surface friction-related diseases including increased friction during blinking have organized the modern understanding of the pathology 5,7).
The most common is dry eye, especially aqueous-deficient dry eye with low tear secretion. In addition, it tends to occur in people with incomplete blinking (ptosis, facial nerve palsy, prolonged eyelid closure due to impaired consciousness), after eye surgery, those with systemic diseases such as Sjögren’s syndrome or graft-versus-host disease, and those who have used eye drops containing preservatives for a long time.
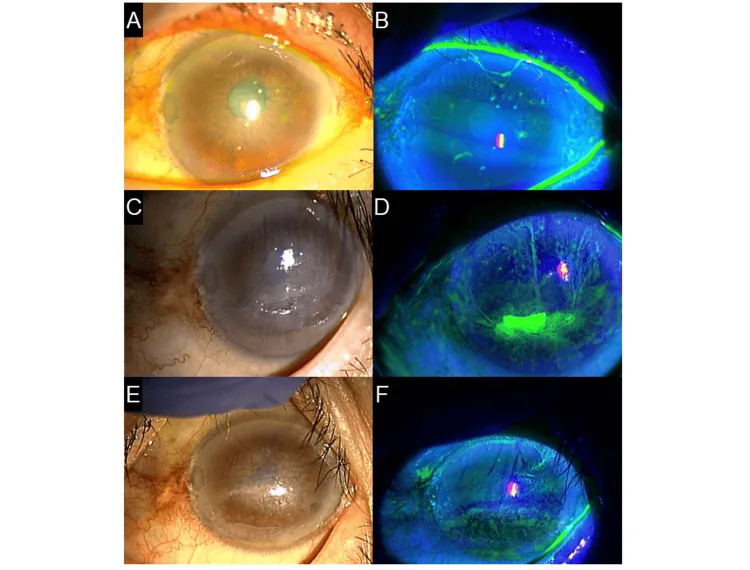
Because the filament moves with friction during each blink, severe foreign body sensation and eye pain are characteristic. Symptoms are mild when the eyes are closed, and pain increases with opening of the eyelids or blinking, so patients may also develop photophobia and blepharospasm. Stimulation causes tearing, and in severe cases, the patient may be almost unable to open the eyes.
Corneal filaments are usually 2 mm or less, but rarely they can form long filaments up to 10 mm. They stain with fluorescein, rose bengal, or lissamine green. The base of the filament may be accompanied by a gray epithelial opacity (foot plaque), and stimulation causes conjunctival injection and edema. The cornea often remains transparent except for the filament 1). Depending on the cause, filaments may occur over the entire cornea, along the eyelid margin, or at corneal irregularities, and their distribution pattern is directly linked to the suspected underlying disease.
The movement of the filament during blinking is easily observed under a slit lamp, and sometimes the filament is seen elongated and hanging from the central cornea to the periphery. In severe cases, epithelial defects may be present at the base of the filament, and the background epithelium often shows diffuse punctate keratopathy (SPK). In an example figure from the Dry Eye Clinical Practice Guidelines, fluorescein staining shows severe epithelial damage with corneal filaments over the entire cornea 1).
Since the distribution pattern of filaments is directly linked to the suspected underlying disease, detailed observation with a slit lamp is important.
| Location of filaments | Suspected underlying disease |
|---|---|
| Entire cornea | Aqueous tear-deficient dry eye, epithelial instability |
| Along the eyelid margin | Ptosis, entropion, lagophthalmos |
| Corneal irregularity | Post-surgery, post-trauma, recurrent corneal erosion |
| Superior cornea | Superior limbic keratoconjunctivitis (SLK) |
| Central localization | Refractory (consider PTK) |
The following six items are standard observations during examination.
Ocular surface evaluation
Location of filaments: Directly linked to estimating the underlying disease
Meibomian gland status: Check for obstruction or atrophy
Eyelid closure and opening: Assess for incomplete blink or lagophthalmos
Tear film, nerve, and eye position evaluation
Tear volume: Measure BUT and tear meniscus height
Corneal sensation: Essential to rule out neurotrophic keratopathy
Eye movement: Evaluate resting fixation position (fixed strabismus)
Each blink pulls on the filaments, causing a gritty foreign body sensation or stabbing pain. It is relatively comfortable with eyes closed, but pain intensifies with blinking or opening the eyes. Pain may cause tearing, photophobia, or inability to keep the eyelids open.
Filamentary keratitis is always a secondary disease following some underlying condition; identifying and managing the underlying disease is key to treatment.
| Underlying disease | Mechanism |
|---|---|
| Aqueous tear-deficient dry eye | Decreased aqueous layer of tears / turnover abnormality |
| Superior limbic keratoconjunctivitis (SLK) | Increased friction and keratinization of superior conjunctiva |
| Neurotrophic keratopathy | Delayed epithelial healing due to trigeminal nerve palsy |
| Lagophthalmos / ptosis | Corneal dryness, exposure, incomplete eyelid closure |
| Post-ocular surgery | Epithelial damage, hypoesthesia, delayed wound healing |
| Recurrent corneal erosion | Instability of epithelial basement membrane |
| Diabetes mellitus | Delayed epithelial healing, hypoesthesia |
| Fixed strabismus | Lagophthalmos and persistent exposure of part of the cornea |
Systemic diseases associated include Sjögren’s syndrome, ocular graft-versus-host disease (oGVHD), thyroid eye disease, cerebrovascular disease (prolonged eyelid closure), Parkinson’s disease (decreased blink rate), and myasthenia gravis (ptosis). Filamentary keratitis due to brain lesions is known to be the most treatment-resistant. In Sjögren’s syndrome, autoimmune destruction of the lacrimal gland reduces tear production; in oGVHD, chronic inflammation of the conjunctiva and lacrimal gland combined with meibomian gland atrophy significantly increases the risk of developing this condition6).
Surgery-related triggers include cataract surgery, corneal transplantation, vitrectomy, and refractive surgery. Intraoperative corneal epithelial damage, postoperative use of multiple eye drops containing preservatives, postoperative dry eye, and temporary reduction in corneal sensation act in combination. In particular, after penetrating keratoplasty (PKP), corneal hypoesthesia persists for a long time, making the condition more difficult to treat.
Iatrogenic factors include long-term use of anticholinergic drugs, epithelial toxicity from long-term use of eye drops containing preservatives (benzalkonium chloride), and the washout effect from multiple eye drop instillations. The Japanese Dry Eye Guidelines state that when encountering refractory filamentary keratitis, attention should be paid to the presence of eyelid abnormalities (especially ptosis and entropion)1).
Diagnosis is easily made by slit-lamp examination. The presence of filamentous material attached to the corneal surface that moves with blinking confirms the diagnosis1). Vital staining should always be used during observation. Fluorescein staining most sensitively visualizes the filaments and surrounding epithelial damage, and is further enhanced with a blue-free filter. Rose bengal and lissamine green are excellent for staining degenerated epithelium and facilitate evaluation of the underlying epithelial defect.
Since most cases of filamentary keratitis involve tear film abnormalities, the following tests are combined.
If Sjögren’s syndrome is suspected, perform anti-SS-A/SS-B antibodies, antinuclear antibodies, salivary gland scintigraphy, and labial minor salivary gland biopsy. If thyroid eye disease is suspected, evaluate thyroid function (TSH, FT3, FT4) and autoantibodies (TRAb, TSAb).
The following differential diagnoses are important for diseases that present with findings similar to filamentary keratitis or that are complicated by filamentary keratitis.
In clinical practice, stratification is performed based on the number, size, and distribution of filaments, severity of underlying disease, and intensity of subjective symptoms, as follows.
The Japanese Ophthalmological Society’s dry eye clinical practice guidelines recommend including the presence of filamentary keratitis, superior limbic keratoconjunctivitis, and conjunctivochalasis as dry eye-related findings in the evaluation items 1).
Treatment of filamentary keratitis has two main pillars: removal of filaments and management of the underlying disease. Since recurrence is inevitable if only one is performed, both should be carried out in parallel.
After topical anesthesia (e.g., 0.4% oxybuprocaine), remove the filaments from the base using non-toothed forceps. Small filaments can also be scraped off with a cotton swab, MQA® (micro-absorbent sponge), or spring scissors. Removal provides immediate symptom relief, but if the underlying disease remains unresolved, recurrence occurs within days to weeks; therefore, this is positioned as the starting point of treatment 1).
The first-line drug is rebamipide ophthalmic suspension (Mucosta® UD 2%). It is instilled four times daily. Rebamipide increases both secreted and membrane-bound mucins, improving tear film stability, and also has anti-inflammatory effects and promotes corneal epithelial wound healing. The increase in mucins reduces ocular surface friction during blinking, which is considered the mechanism of action for this disease. The efficacy of rebamipide is explained as “improvement of tear film stability through increased secreted and membrane-bound mucins, and corneal epithelial wound healing including anti-inflammatory effects,” and it is positioned as a relatively new treatment option for filamentary keratitis.
| Treatment Category | Drug/Intervention | Dosage/Method | Positioning |
|---|---|---|---|
| Mucin regulation | Rebamipide ophthalmic suspension (Mucosta® UD 2%) | 4 times daily | First-line |
| Artificial tears | Preservative-free artificial tears (Soft Santear®, Hyalein Mini®, etc.) | As needed, 4-6 times or more per day | Basic combination |
| Anti-inflammatory | Fluorometholone ophthalmic solution 0.1% | 2-4 times per day | Short-term |
| Immunomodulation | Cyclosporine ophthalmic solution 0.1% (Papilock Mini®, etc.) | 2 times per day | Chronic cases |
| Mucolytic | N-acetylcysteine ophthalmic solution 5-10% | 4 times per day | Refractory cases overseas4) |
| Oral anti-inflammatory | Minocycline 100 mg/day, Doxycycline | Oral | Complicated with severe MGD |
| Tear volume maintenance | Punctal plug | Silicone / Absorbable | Aqueous tear-deficient type |
| Physical protection | Therapeutic soft contact lens | Continuous wear for 1 week / Replace once a week | Prevention of recurrence |
N-acetylcysteine eye drops (5–10%) have been used as a mucolytic agent, but since there is no commercially available formulation, their use is limited to facilities that prepare them as in-hospital formulations. Avisar et al. reported in a randomized controlled trial that 0.1% diclofenac eye drops were effective in improving filamentary keratitis compared to 5% saline (NaCl 5%), but the Dry Eye Clinical Practice Guideline takes a cautious stance on the general efficacy of NSAID eye drops1,4).
Therapeutic soft contact lenses are an option that mechanically protects the ocular surface, aiming to reduce pain and stabilize the epithelium11). Because continuous wear carries a risk of infectious keratitis, the lenses should be used for a short period with strict management and replacement, while assessing whether they can be discontinued. During wear, frequent instillation of preservative-free artificial tears should be combined to ensure tear volume.
For refractory cases, consider the following stepwise approaches.
The clinical treatment flow is as follows1).
If the underlying disease is properly managed, the prognosis is generally good, but recurrence is not uncommon. In particular, filamentary keratitis caused by brain lesions is the most treatment-resistant and requires long-term ocular surface management.
If the underlying causes (such as dry eye, eyelid abnormalities, or nerve palsy) are appropriately treated, symptoms often improve. However, it is a disease prone to recurrence, and long-term ocular surface management is necessary. Since removal of filaments alone leads to repeated recurrence, it is important to combine basic treatments such as rebamipide eye drops with management of the underlying disease. Cases caused by brain disease or nerve damage tend to be more difficult to treat.
The mechanism of filamentary keratitis is a multi-step process starting from corneal epithelial damage 5).
First, damage to the basal epithelial cells, epithelial basement membrane, or Bowman’s layer causes local detachment of the epithelial basement membrane. When there is an underlying abnormality in tear turnover or tear film components, the damaged area is difficult to repair and becomes chronic. Shear forces from blinking lift the detached area, inducing irritation and inflammation. Mucin adheres to the epithelial cell components as a core, and inflammatory cells and conjunctival epithelial cells become involved, forming filaments.
In an immunohistological study by Tanioka et al. (2009, IOVS), corneal filaments in filamentary keratitis were shown to have a structure with a core derived from corneal epithelial cells, surrounded by conjunctival epithelial cells and mucin. The study concluded that chronic corneal epithelial damage and increased friction during blinking are involved in filament formation 5). This immunohistological finding supported the previously empirically known pathology at the molecular level.
The most important concept in recent years is increased ocular surface friction. The Japanese Dry Eye Society’s clinical practice guidelines (Journal of Japanese Ophthalmological Society, 2019) categorize the pathophysiology of dry eye into two vicious cycles: decreased tear film stability during open-eye maintenance (core mechanism 1) and increased friction during blinking (core mechanism 2). Filamentary keratitis, along with LWE, SLK, and conjunctivochalasis, is positioned as a dry eye-related disease sharing the latter mechanism 1). The area of the palpebral conjunctiva that causes friction with the ocular surface during blinking was named the lid wiper by Korb et al., and increased friction between this area and the corneal surface leads to lid-wiper epitheliopathy and corresponding corneal epithelial damage 8).
Changes in the composition of the tear film are also involved. An imbalance in the ratio of mucin to aqueous layer, typically based on reduced aqueous tear production, but increased mucin production or accumulation may also contribute. This alteration in tear film composition provides a substrate for filament formation 7). The clinical fact that rebamipide ophthalmic solution is effective for this disease by increasing mucin and reducing friction is consistent with this mechanistic model.
Historically, the concept of “epithelial crack line” proposed by Ohashi et al. in 1992 is known as a pioneering study suggesting a close relationship between corneal epithelial layer structure and tear dynamics 9). Additionally, the Meibomian Gland Dysfunction (MGD) clinical practice guidelines point out that abnormalities in the meibomian oil layer can contribute to filamentary keratitis through reduced tear film stability 10).
The involvement of inflammation has also gained attention in recent years. While inflammation is seen as a result of the two vicious cycles of reduced tear stability and increased friction, there is also a theory that inflammation itself is the core mechanism of dry eye, and it is important to understand both in an integrated manner 1). Molecular mechanisms underlying filamentary keratitis, such as CD4-positive T cell infiltration, reduction of conjunctival goblet cells by interferon-gamma, and promotion of apoptosis of corneal and conjunctival epithelial cells, are being elucidated. Cyclosporine ophthalmic solution and diquafosol ophthalmic solution are positioned as drugs that intervene in these pathways as anti-inflammatory and mucin secretagogue agents.
The TFOS DEWS III (2025) Management and Treatment Report mentions botulinum toxin injection as one option for refractory filamentary keratitis 2). Multiple randomized trials have shown improvements in TBUT, Schirmer test values, corneal fluorescein staining scores, and OSDI scores after local injection into the medial lower eyelid. This may be a reasonable choice especially for filamentary keratitis complicated by blepharospasm. However, the effect lasts about 3 months, and repeated administration and cost are challenges.
Development of novel mucin secretagogues and mucin mimetics is also underway, exploring therapeutic strategies that reinforce the mucin layer of the tear film through mechanisms different from rebamipide. Visualization technologies for the pathology, such as friction meters that quantitatively measure ocular surface friction and in vivo confocal microscopy for real-time observation of filaments, are also advancing 6).
Expansion of indications for blood-derived products such as autologous serum eye drops, allogeneic serum eye drops, and platelet-rich plasma (PRP) eye drops is also being considered. These contain growth factors, vitamin A, fibronectin, etc., and are expected to promote corneal epithelial healing.
The treatment algorithm based on the concept of “friction enhancement” is becoming widespread, and personalized treatment combining rebamipide, diquafosol, artificial tears, punctal plugs, therapeutic contact lenses, and eyelid surgery is being implemented clinically. In the future, refinement of the disease concept itself is required, including severity classification specific to filamentary keratitis, predictive models of treatment response, and exploration of biomarkers to prevent recurrence of filaments.
Evidence reviews of acetylcysteine eye drops for refractory filamentary keratitis and reports of long-term management using therapeutic scleral lenses (PROSE, scleral lens) are also accumulating 6,11). Scleral lenses form a fluid reservoir between the cornea and the back surface of the lens, providing continuous ocular surface wetting and physical protection, suggesting their usefulness in refractory cases associated with severe dry eye or neurotrophic keratopathy.
日本ドライアイ研究会. ドライアイ診療ガイドライン. 日眼会誌. 2019;123(5):489-592. URL: https://dryeye.ne.jp/wp/wp-content/themes/dryeye/file/dryeye_guideline.pdf
Bhattacharya P, Jones L, Craig JP, et al. TFOS DEWS III Management and Therapy Report. Am J Ophthalmol. 2025.
北澤耕司, 横井則彦, 渡辺彰英, 荒木美治, 小室青, 稲垣香代子, ほか. 難治性糸状角膜炎に対する眼瞼手術の検討. 日眼会誌. 2011;115(8):693-698. URL: https://www.nichigan.or.jp/Portals/0/JJOS_PDF/115_693.pdf
Avisar R, Robinson A, Appel I, Yassur Y, Weinberger D. Diclofenac sodium, 0.1% (Voltaren Ophtha), versus sodium chloride, 5%, in the treatment of filamentary keratitis. Cornea. 2000;19(2):145-147.
Tanioka H, Yokoi N, Komuro A, Shimamoto T, Kawasaki S, Matsuda A, et al. Investigation of the corneal filament in filamentary keratitis. Invest Ophthalmol Vis Sci. 2009;50(8):3696-3702.
Albietz JM, Sanfilippo PG, Troutbeck R, Lenton LM. Management of filamentary keratitis associated with aqueous-deficient dry eye. Optom Vis Sci. 2003;80(6):420-430.
山口昌彦. 眼表面摩擦関連疾患の診断と治療. 臨床眼科. 2018;72(11):84-90. doi:10.11477/mf.1410212883.
Korb DR, Herman JP, Blackie CA, Scaffidi RC, Greiner JV, Exford JM, et al. Prevalence of lid wiper epitheliopathy in subjects with dry eye signs and symptoms. Cornea. 2010;29(4):377-383.
大橋裕一, 木下茂, 細谷比左志, 李三榮, 荒木かおる, 切通彰, ほか. 角膜上皮障害の新しい病態—Epithelial Crack Line. 臨床眼科. 1992;46:1539-1543.
マイボーム腺機能不全診療ガイドライン作成委員会. マイボーム腺機能不全診療ガイドライン. 日眼会誌. 2023;127:109-228. URL: https://www.nichigan.or.jp/Portals/0/resources/member/guideline/MGD.pdf
Jain S, Sukhija J, Saini JS. Contact lenses for the treatment of ocular surface diseases. Indian J Ophthalmol. 2023;71:1962-1970. PMCID: PMC10276751. doi:10.4103/ijo.IJO_3004_22.