Grade I
Diameter less than 5 mm: Superficial and confined to the limbus.
Depth of invasion: Limited to about half of the corneal stroma.

Corneal dermoid is a congenital benign tumor that occurs on the cornea, limbus, or conjunctiva. It is a choristoma in which skin tissue aberrantly proliferates in the corneoconjunctival area, also called limbal dermoid. A choristoma is a tumor composed of normal tissue growing in an ectopic location.
The incidence in newborns is 1–3 per 10,000, accounting for 25% of congenital orbital lesions 2). Limbal dermoid is the most common type, typically occurring at the inferotemporal limbus. It is usually unilateral and occurs sporadically without heredity. Its size generally does not change after birth. Diameter ranges from 3 mm to as large as 10 mm.
Corneal dermoid and orbital dermoid cyst are completely different diseases and should not be confused.
Corneal dermoid is a congenital benign tumor (choristoma) with no reports of malignant transformation. It is a non-proliferative lesion that generally does not change in size after birth. However, it may cause amblyopia or cosmetic issues, so regular follow-up is important.
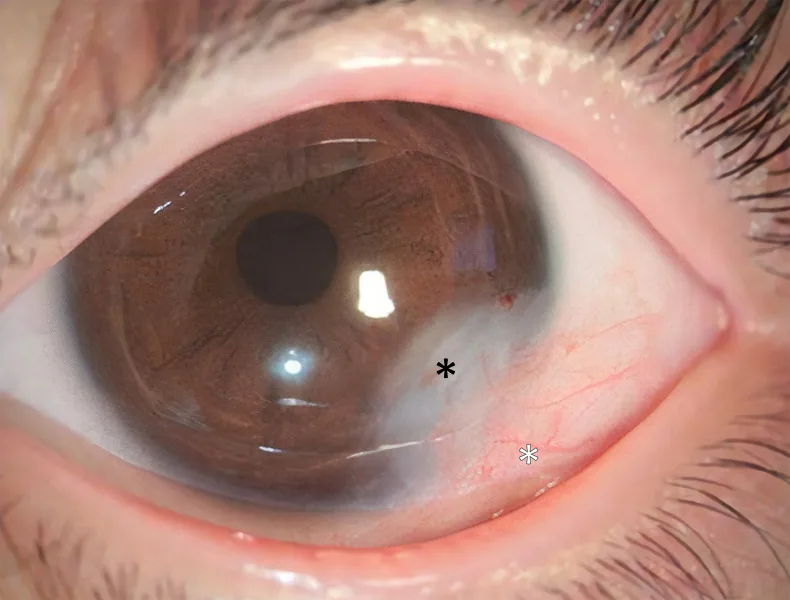
Corneal dermoid is a well-demarcated, white, hemispherical, solid tumor that commonly occurs at the inferotemporal limbus. It appears milky white to light brown and may have hairs on its surface. The dermoid surface is keratinized like skin, causing tears to be repelled.
Oblique astigmatism and anisometropic amblyopia are present in about two-thirds of cases. Bilateral involvement is seen in 10–25% of all ocular surface dermoids5).
Based on clinical and AS-OCT findings, they are classified into three grades5).
Grade I
Diameter less than 5 mm: Superficial and confined to the limbus.
Depth of invasion: Limited to about half of the corneal stroma.
Grade II
Large lesion: Covers most of the cornea and extends to just anterior to Descemet’s membrane in the deep stroma.
Characteristic: Does not involve Descemet’s membrane itself.
Grade III
Most severe: Covers the entire cornea and penetrates from the anterior surface of the eyeball to the iris pigment epithelium.
Frequency: The rarest type.
Corneal dermoid results from abnormal development of the first and second branchial arches during embryonic life. Dysplasia of the ectoderm that forms the cornea and conjunctiva causes skin tissue to become misplaced into the corneoconjunctiva, forming a dermoid. Most cases are sporadic, but it can occur in association with the following syndromes.
A syndrome in which limbal dermoid is accompanied by accessory ears, ear pits, mandibular hypoplasia, and vertebral anomalies, occurring in 1 in 3,000–5,000 births with a male-to-female ratio of 3:2 1). Most cases are sporadic. Maternal diabetes, hypertension, viral infections (influenza, rubella), and exposure to cocaine, tamoxifen, or retinoic acid have been reported as predisposing factors 1).
When limbal dermoid is found, systemic examination should be performed to check for the presence of Goldenhar syndrome.
Corneal dermoid (limbal dermoid) can be an ocular finding of Goldenhar syndrome. Goldenhar syndrome is a congenital disorder accompanied by accessory ears, ear pits, mandibular hypoplasia, and vertebral anomalies, occurring in 1 in 3,000–5,000 births 1). When limbal dermoid is found, it is important to perform systemic examination to confirm these associations.
The diagnosis of corneal dermoid is primarily based on clinical findings, and biopsy is generally not required. If a resected specimen is available, histopathological examination can confirm the diagnosis.
If Goldenhar syndrome is suspected, multidisciplinary evaluation including otolaryngology and pediatrics should be performed. Hearing tests, head CT, spinal X-rays, and cardiac/renal ultrasound are recommended 1).
The most important aspect of corneal dermoid is the prevention and treatment of amblyopia. Surgery does not improve astigmatism and therefore does not treat amblyopia. It is necessary to start amblyopia treatment with refractive correction (glasses) and occlusion of the healthy eye before surgery.
For Grade I dermoids with astigmatism less than 1D and minimal surface irregularity, conservative observation is appropriate. Regular examinations should be performed every 6 to 12 months, evaluating visual acuity, lesion size, and cycloplegic refraction.
Surgery is primarily cosmetic, and early surgery is generally not indicated. Surgery is considered in the following cases:
The surgical procedure is selected according to the grade.
| Grade | Recommended Procedure |
|---|---|
| Grade I (superficial) | Excision + superficial keratectomy |
| Grade II | Deep anterior lamellar keratoplasty (DALK) ± amniotic membrane transplantation |
| Grade III | Total anterior segment reconstruction |
Superficial keratectomy is performed after tumor excision. Simple excision alone frequently leads to recurrence or pseudopterygium. The graft does not need to be fresh cornea; frozen preserved cornea is acceptable. The trephine diameter is usually about 7.0 mm. Suturing is performed with 10-0 nylon interrupted sutures, approximately 12 stitches. Since residual tumor can cause severe postoperative inflammation and pseudopterygium, complete excision should be aimed for.
Abdulmannan et al. (2022) reported the management outcomes of 5 cases of limbal dermoid in infants and children. Astigmatism of 1D or more was observed in 4 cases, but astigmatism did not improve after surgery, and visual improvement was mainly due to spectacle use and occlusion of the healthy eye 6).
Removing the dermoid by surgery does not improve astigmatism. Surgery is for cosmetic purposes and does not treat amblyopia. For astigmatic amblyopia, refractive correction with glasses and occlusion therapy of the healthy eye are necessary. It has been reported that astigmatism does not change or tends to increase slightly after surgery 6).
Surgery for corneal dermoid is for cosmetic purposes, and in principle, there is no indication for early surgery. Dermoids covering the pupillary area require early surgery, but if they are confined to the limbus and not enlarging, surgery can be performed after infancy. Surgery at a young age requires general anesthesia.
Several hypotheses exist regarding the pathogenesis of corneal dermoid. One theory suggests that it arises from metaplastic changes in mesodermal cells between the optic cup rim and surface ectoderm during early development. Another proposes sequestration of pluripotent cells during embryonic development of surrounding ocular structures.
As a choristoma, a dermoid is composed of ectodermal tissues such as hair, skin, and rarely teeth, along with mesodermal tissues such as adipose tissue and cartilage. Histologically, it shows structures resembling epidermis and dermis containing collagenous connective tissue, hair follicles, and sebaceous glands.
The dermoid tissue itself has no proliferative activity.
Mitamura et al. (2021) reported a pterygium-like growth containing residual dermoid tissue in a 79-year-old Japanese woman who underwent limbal dermoid resection at age 12. Immunohistochemistry showed Ki67 (cell proliferation marker) positivity in pterygium epithelial cells and neovascular endothelial cells, but negativity in dermoid components (adipose tissue, connective tissue, peripheral nerves) 3). Although residual dermoid tissue after incomplete resection lacks proliferative capacity, it may act as a modifying factor for pterygium development.
This report supports the importance of complete resection and reaffirms that dermoid tissue itself is a congenital, non-proliferative lesion.
Poddi et al. (2025) reported a technique for Grade I limbal dermoid involving excision followed by mitomycin C (0.02%) application and triple-layer amniotic membrane transplantation fixed with fibrin glue. In an 18-year-old female, no recurrence was observed over 2 years of follow-up, with corneal topographic regularization, reduction in higher-order aberrations, and improvement in BCVA 2).
For a giant corneal and ocular surface dermoid (17×14 mm), Choudhary et al. (2021) reported a combined technique of pre-descemetic DALK (deep anterior lamellar keratoplasty) and SLET (simple limbal epithelial transfer) from the contralateral eye. Satisfactory cosmetic results were achieved while preventing pseudopterygium formation 4).
Lamellar keratoplasty using lenticules extracted from SMILE surgery has also been reported, attracting attention as a technique that does not require donor corneas.
Fu et al. (2024) performed whole exome sequencing (WES) on a 4-year-old boy with Goldenhar syndrome and identified five gene mutations (PRDM16, FAT4, POLR1C, PLEC, MED12) potentially associated with microtia 1). Elucidating the genetic basis of Goldenhar syndrome may contribute to future diagnosis and prevention.
- Fu Y, Yu H, Zhang J, Zhou N. Goldenhar syndrome with limbal neoformation, microtia and skeletal deformities: a case report and literature review. BMC Ophthalmol. 2024;24:81.
- Poddi M, Romano V, Borgia A, et al. Combined Multilayered Amniotic Membrane Graft and Fibrin Glue as a Surgical Management of Limbal Dermoid Cyst. J Clin Med. 2025;14:607.
- Mitamura M, Kase S, Ohguchi T, Ishida S. A case of pterygium-like proliferation containing postoperative limbal dermoid remnants: a clinicopathological study. BMC Ophthalmol. 2021;21:12.
- Juneja A, Puthalath AS, Gupta N, Verma PK. Isolated bilateral limbal dermoid, preauricular skin tags and ametropic amblyopia. BMJ Case Rep. 2021;14:e245078.
- Abdulmannan DM. Successful Management of Limbal Dermoid in Infancy and Childhood: A Case Series. Cureus. 2022;14(3):e22835.