Hyperopic Anisometropia
Anisometropia (Difference in Refractive Power Between Eyes)
1. What is anisometropia?
Section titled “1. What is anisometropia?”Anisometropia is a condition in which the degree of refractive error differs between the two eyes. A small difference is considered physiological anisometropia, and a difference of approximately 1.5–2.0 D or more is generally defined as anisometropia. For astigmatic anisometropia, comparison is made using the spherical equivalent.
Classification
Section titled “Classification”Anisometropia is classified as follows based on its nature and location.
| Classification | Description |
|---|---|
| Compound anisometropia | One eye is myopic and the other is emmetropic or hyperopic. |
| Isometropic anisometropia | Both eyes have hyperopia or myopia but of different degrees (hyperopic type is common in children, myopic type in adults) |
| Axial anisometropia | No difference in refractive power of the optical system, but difference in axial length |
| Refractive anisometropia | Caused by difference in refractive power, such as unilateral aphakia |
Epidemiology
Section titled “Epidemiology”Anisometropic amblyopia is the most common cause of amblyopia. It is reported to account for about one-third to 50% of all amblyopia. It is often detected during health checkups at age 3 or before school entry. Hyperopic anisometropia is predominant in children, while myopic anisometropia becomes more common in adults.
The prevalence of amblyopia is reported to be 2–4% 1). About one-third of children with anisometropia of 2D or more have amblyopia, and even 1–2D of spherical equivalent anisometropia increases the odds of amblyopia by 4.5 times 1).
Population studies on the prevalence of anisometropia estimate that about 2–5% of the adult population have anisometropia of 1D or more. In pediatric populations, screening studies using photoscreening devices (e.g., Spot Vision Screener) have improved detection rates of anisometropia with amblyopia risk factors, and early detection before age 3 is becoming standardized 9).
The incidence of anisometropic amblyopia varies by type of refractive error. Hyperopic anisometropia is considered a risk for amblyopia from about 1D, while myopic anisometropia generally becomes a risk only at 3D or more. The Amblyopia PPP (2022 revision) identifies hyperopic anisometropia of +4.5D or more (with strabismus) or +6D or more (without strabismus) as particularly high risk 1).
Anisometropia alone is often asymptomatic, but it carries several risks. First, in children, amblyopia develops in the eye with greater refractive error during the sensitive period. Second, aniseikonia (difference in image size between the two eyes) can cause eye strain. Third, binocular vision and stereopsis are impaired. Because anisometropia is not easily noticeable externally, refractive screening during health checkups is important.
2. Main Symptoms and Clinical Findings
Section titled “2. Main Symptoms and Clinical Findings”Subjective Symptoms
Section titled “Subjective Symptoms”Anisometropia alone is usually asymptomatic or causes only mild eye strain. When amblyopia is present, decreased visual acuity in the eye with the stronger refractive error becomes apparent.
Aniseikonia
Section titled “Aniseikonia”Aniseikonia is a condition in which the images seen by the two eyes differ in size. Aniseikonia exceeding 5% can cause eye strain. Large aniseikonia of 7% or more makes binocular vision and fusion impossible. The greater the anisometropia, the higher the risk of aniseikonia.
Clinical Findings
Section titled “Clinical Findings”The diagnostic criteria for amblyopia are shown below1).
| Age | Unilateral Amblyopia | Bilateral Amblyopia |
|---|---|---|
| 3–4 years | Interocular difference ≥2 lines | Both eyes ≤20/50 |
| ≥5 years | Interocular difference ≥2 lines | Both eyes ≤20/40 |
3. Causes and Risk Factors
Section titled “3. Causes and Risk Factors”Degree of Anisometropia That Causes Amblyopia
Section titled “Degree of Anisometropia That Causes Amblyopia”Generally, anisometropia of 2D or more can cause amblyopia. Hyperopia of 3D or more and astigmatism of 1.5D or more are likely to lead to amblyopia. The risk varies depending on the type of refractive error.
Myopic Anisometropia
Astigmatic Anisometropia
Amblyopia risk: Can develop with a difference of 1.5D or more between eyes.
Characteristics: The axis direction affects visual development. Evaluation is performed by comparing the spherical equivalent.
Mechanism of Onset
Section titled “Mechanism of Onset”In anisometropia, the eye closer to emmetropia becomes the dominant eye. The eye with stronger refractive error is suppressed in the visual cortex, impairing the development of neurons corresponding to that eye. In hyperopic anisometropia, the eye with stronger hyperopia cannot obtain a clear image at distance or near, so the risk of amblyopia formation is particularly high1).
4. Diagnosis and Examination Methods
Section titled “4. Diagnosis and Examination Methods”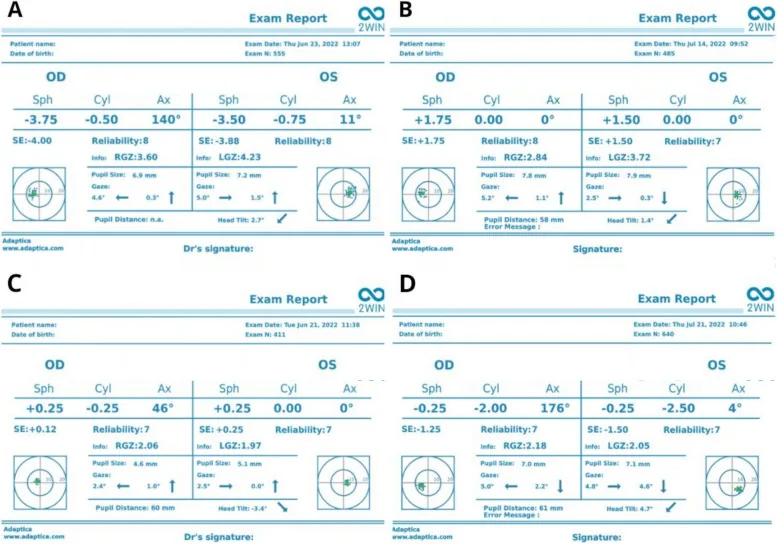
The diagnosis of anisometropic amblyopia is a diagnosis of exclusion after ruling out organic diseases and strabismus. It is diagnosed when the following conditions are met.
Refraction Examination
Section titled “Refraction Examination”Cycloplegic refraction is essential. Children have strong accommodative ability, and subjective tests or standard autorefraction cannot accurately measure the degree of anisometropia. Objective refraction is performed after instillation of cycloplegic agents (atropine or cyclopentolate). Note that the degree of anisometropia varies greatly depending on the measurement method (autorefractor, lens exchange method, presence or absence of cycloplegia).
Visual Acuity Examination
Section titled “Visual Acuity Examination”Age-appropriate visual acuity testing methods are used. Optotype visual acuity is the most common for detecting amblyopia. In amblyopic eyes, line visual acuity tends to be more reduced than single optotype acuity (crowding phenomenon).
Binocular Vision Function Examination
Section titled “Binocular Vision Function Examination”Stereopsis tests (Titmus stereotest, TNO test, etc.), fusion range measurement, and Worth 4-dot test are performed. In anisometropic amblyopia, interocular suppression from the fellow eye is involved, so evaluation of binocular vision function is useful for determining treatment strategy 1). Contrast sensitivity is reduced in the medium to high spatial frequency range, and this reduction extends to both central and peripheral visual fields, which differs from strabismic amblyopia.
Anterior Segment and Fundus Examination
Section titled “Anterior Segment and Fundus Examination”This is essential to rule out organic diseases as a cause of amblyopia. Slit-lamp microscopy and dilated fundus examination are performed.
Evaluation of Anisometropia and Aniseikonia
Section titled “Evaluation of Anisometropia and Aniseikonia”When anisometropia exceeds 1.5 D, aniseikonia (difference in image size between the two eyes) is likely to occur. The degree of aniseikonia with corrective glasses varies greatly depending on the correction method (glasses vs. contact lenses), so measuring aniseikonia (e.g., New Aniseikonia Tests) is beneficial when deciding treatment strategy. Judgment based on Knapp’s law, which states that axial anisometropia is better corrected with glasses and refractive anisometropia with contact lenses, is important 1).
Accuracy of Photoscreening
Section titled “Accuracy of Photoscreening”According to comparative studies of autorefractometers and various photoscreeners, the intraclass correlation coefficients (ICC) between the 2WIN photoscreener and calibrated autorefractor under non-cycloplegic conditions showed high agreement of 0.88–0.97 for spherical power, cylindrical power, and spherical equivalent 9). The difference between cycloplegic and non-cycloplegic measurements (i.e., latent hyperopia) tends to be small in myopic eyes and large in hyperopic eyes, and the importance of cycloplegic examination in screening for hyperopic anisometropia has been reaffirmed 9).
Children have very strong accommodative ability, and subjective tests or autorefraction without cycloplegia cannot provide accurate refractive values. Especially in hyperopic eyes, accommodation tends to underestimate the refractive error, and the degree of anisometropia may differ significantly. By measuring under complete cycloplegia with atropine (1%) or cyclopentolate (1%), the true refractive values can be obtained. Cycloplegic refraction is essential for diagnosing anisometropia and determining treatment strategies.
5. Standard Treatment Methods
Section titled “5. Standard Treatment Methods”The goal of treating anisometropic amblyopia is to make the visual acuity of both eyes as equal as possible 1). The choice of treatment is based on the patient’s age, visual acuity, and compliance.
5A. Treatment of Anisometropic Amblyopia
Section titled “5A. Treatment of Anisometropic Amblyopia”Full-time Wear of Fully Corrective Glasses
Section titled “Full-time Wear of Fully Corrective Glasses”The first step in treatment is full-time wear of fully corrective glasses based on cycloplegic refraction. Simply wearing glasses full-time can improve visual acuity in the amblyopic eye to some extent. When the anisometropic difference is about 2D, treatment with glasses alone is often sufficient.
In a PEDIG prospective study, 27% of children aged 3–6 years with anisometropic amblyopia were cured with glasses correction alone. An average improvement of 0.29 logMAR was achieved, and 77% showed improvement of 0.2 logMAR or more 3). After prescribing glasses, it is currently standard practice to observe with refractive correction alone until visual acuity stabilizes 5). Occlusion is started after confirming the time until treatment response plateaus (up to 30 weeks).
The success rate of amblyopia treatment depends on age. Electronic occlusion dose-monitoring studies showed that the occlusion time required for one line of improvement was 170 hours in 4-year-olds, 236 hours in 6-year-olds, and 490 hours in 8-year-olds 3). Early intervention at a younger age is key to better outcomes, and early screening before age 3 is strongly recommended 1).
When the anisometropic difference is 3D or more, glasses alone often have limited effect on improving visual acuity in the amblyopic eye. In such cases, refraction should be re-evaluated, and if the glasses are appropriate, occlusion of the healthy eye should be initiated.
Occlusion Therapy (Patching)
Section titled “Occlusion Therapy (Patching)”If visual acuity does not improve sufficiently with glasses alone, occlusion of the healthy eye (patching) is added. An adhesive patch is applied directly to the healthy eye to force use of the amblyopic eye.
During patching, it is important to actively use the amblyopic eye, focusing on near vision tasks such as reading and coloring. For moderate amblyopia, patching for 2 hours per day is as effective as 6 hours 6). For severe amblyopia, patching for 6 hours per day is equivalent to nearly full-time patching 7). Starting full correction glasses and occlusion of the sound eye simultaneously can cause significant stress for the patient, so patching is instructed after the patient becomes accustomed to wearing glasses.
Atropine Penalization
Section titled “Atropine Penalization”This method involves administering 1% atropine eye drops to the sound eye, causing cycloplegia and blurring near vision in the sound eye, thereby encouraging use of the amblyopic eye 7). For moderate amblyopia, it is nearly as effective as patching 7).
Bangerter Filter
Section titled “Bangerter Filter”This method involves attaching a Bangerter (translucent) filter to the spectacle lens of the sound eye. In a PEDIG study, the difference in visual acuity improvement between this method and patching after 24 weeks of treatment was within 0.5 lines 1). It is useful as an alternative when compliance with patching is poor.
5B. Selection of Correction Method (Axial vs. Refractive Anisometropia)
Section titled “5B. Selection of Correction Method (Axial vs. Refractive Anisometropia)”The choice of correction method depends on the cause of anisometropia.
Axial anisometropia (due to difference in axial length)
According to Knapp’s law, the vertex distance of glasses (usually 12 mm) is close to the anterior focal point of the eye, so spectacle correction nearly eliminates the difference in retinal image magnification. Spectacle correction is advantageous for axial anisometropia.
Refractive anisometropia (due to difference in corneal or lens refractive power, e.g., aphakia, post-cataract surgery)
Contact lenses correct at a position close to the corneal surface, deviating from the conditions of Knapp’s law, and often produce less aniseikonia than spectacles. For high refractive anisometropia such as unilateral aphakia, contact lenses are the first choice.
5C. Anisometropia in Adults
Section titled “5C. Anisometropia in Adults”In adults, amblyopia is less of an issue, but aniseikonia can cause asthenopia and binocular vision problems. When anisometropia exceeds 1.5 D, aniseikonia tends to become apparent; switching from spectacle correction to contact lenses or prescribing monovision glasses should be considered. Refractive surgery (e.g., LASIK) in adults may be effective in reducing anisometropia and aniseikonia 10).
According to the Refractive Surgery Guidelines (8th edition), LASIK, SMILE, and ICL for adults with myopic anisometropia are evaluated similarly to other refractive errors. However, surgery on only one eye carries a risk of postoperative aniseikonia, so preoperative evaluation of aniseikonia and patient explanation are essential 10).
5D. Bangerter Filters and Dichoptic Therapy
Section titled “5D. Bangerter Filters and Dichoptic Therapy”Bangerter (translucent) filters are attached to the spectacle lens of the sound eye to encourage use of the amblyopic eye. In the PEDIG study, the visual acuity improvement after 24 weeks was within 0.5 lines compared to patching, making it a useful alternative when patching compliance is poor 1).
Dichoptic digital therapy uses VR headsets or tablets to present images of different contrast to the amblyopic and sound eyes, aiming to relieve interocular suppression 3). A study using Luminopia headsets (72 hours of use in children) showed a visual acuity improvement of 0.15 logMAR 3). However, at present, evidence surpassing conventional patching therapy has not been established 1).
Practical Protocol for Occlusion Therapy
The daily occlusion time is determined by the severity of amblyopia 1).
| Severity of Amblyopia | Recommended Occlusion Time | Supporting Evidence |
|---|---|---|
| Moderate amblyopia (20/40 to 20/80) | 2 hours per day | PEDIG ATS 2B: 2 hours = effect of 6 hours (I+, strong recommendation) |
| Severe amblyopia (20/100 or worse) | 6 hours per day | PEDIG ATS 2A: 6 hours ≈ full-day occlusion effect (I+, strong recommendation) |
| Poor responders to treatment | Increase dosage (2→6 hours) | PEDIG ATS 15: Additional improvement with increased dosage (I+) |
The standard patching method involves directly applying an opaque adhesive patch to the sound eye. Fabric covers on glasses are not recommended because they are translucent and less effective. The effect is enhanced by having the child actively use the amblyopic eye during occlusion (reading, coloring, near work) 1).
The sensitive period for vision is generally considered to be until around age 8, and starting treatment within this period is expected to lead to good visual recovery. However, treatment should not be abandoned due to age alone, as some patients show a response even after the sensitive period. If detection is delayed, anisometropia is large, or treatment compliance is poor, good visual development may not be achieved. Early detection and early treatment are the most important principles.
6. Pathophysiology and Detailed Mechanism of Onset
Section titled “6. Pathophysiology and Detailed Mechanism of Onset”Binocular Accommodation and Anisometropia
Section titled “Binocular Accommodation and Anisometropia”Binocular accommodation occurs equally (Hering’s law). The starting point of accommodation is the refractive value of both eyes under cycloplegia. In anisometropia, one eye is always unable to achieve optimal focus. In hyperopic anisometropia, the more hyperopic eye cannot focus at distance or near, and during the growth period, it consistently fails to obtain a clear image.
Hering’s law of equal innervation means that both eyes show an equal accommodative response to a shared accommodative stimulus. In anisometropic eyes, when accommodation is fixed at a distance where the dominant eye (closer to emmetropia) can focus comfortably, the amblyopic eye continuously forms a retinal image that is out of optimal focus. This persistent degradation of the retinal image impairs visual development and forms the pathological basis of amblyopia 3).
Difference Between Axial Anisometropia and Refractive Anisometropia
Section titled “Difference Between Axial Anisometropia and Refractive Anisometropia”Axial anisometropia is caused by a difference in axial length, while refractive anisometropia is caused by a difference in refractive power of the cornea or lens. This distinction directly affects the choice of correction method.
According to Knapp’s law, in axial anisometropia, if spectacles are worn such that the second principal point of the lens coincides with the anterior focal point of the eye, the retinal image size becomes equal to that of a normal eye. On the other hand, in refractive anisometropia, spectacle correction tends to leave aniseikonia, so contact lenses are more suitable.
In clinical practice, the differentiation of anisometropia is performed using the following methods.
- Biometry (axial length measurement): Measure axial length in both eyes using IOL Master, Lenstar, etc., and check the difference (usually a 1 mm difference corresponds to approximately 3 D of refractive difference).
- Corneal refractive power measurement: Evaluate the difference in corneal curvature between the two eyes using keratometry and corneal topography.
- Decomposition of refractive power difference: Decompose the total anisometropia into corneal, lenticular, and axial length components to identify the main factor.
- Comparative measurement of aniseikonia with glasses and contact lenses: Measure aniseikonia under different correction conditions using tests such as the New Aniseikonia Test.
Differentiating axial from refractive anisometropia is also important in amblyopia treatment. In axial anisometropia, Knapp’s law applies with spectacle correction, minimizing aniseikonia. This makes it easier to maintain full correction for amblyopia treatment, improving compliance.
Neuropathological changes in amblyopia
Section titled “Neuropathological changes in amblyopia”If the retinal image in one eye is blurred during the sensitive period of visual development, the development of visual cortex neurons corresponding to that eye is impaired.
Interocular suppression and binocular vision impairment
Section titled “Interocular suppression and binocular vision impairment”The pathophysiology of anisometropic amblyopia involves not only the direct effect of retinal image blur but also interocular suppression from the fellow eye 1). Contrast sensitivity decreases in the medium to high spatial frequency range, and this decrease extends to both central and peripheral visual fields. This differs from strabismic amblyopia, which shows deficits only in the central visual field.
Anisometropic amblyopia accounts for 46–79% of all amblyopia cases where refractive error is a contributing factor, and 19–50% of these are mixed type with concomitant strabismus 1). The degree of interocular suppression can be quantified by the size and depth of the suppression scotoma, which is useful for monitoring the effects of amblyopia treatment (suppression release and contrast sensitivity recovery).
Neuromorphological basis
Section titled “Neuromorphological basis”Continuous exposure to a blurred retinal image during the visual sensitive period (critical period) leads to shrinkage of the amblyopic eye columns in the primary visual cortex (V1) and relative expansion of the cortical areas dominated by the fellow eye. This change has been established in animal experiments and confirmed in humans using fMRI 3). Treatment (occlusion, atropine) can reverse the plastic changes in ocular dominance columns and restore cortical representation of the amblyopic eye. This neuroplasticity persists to some extent even in adults, providing a biological basis for therapeutic interventions in adult amblyopia 3).
Mukit et al. (2023) reported a case of a 6-year-old girl with high anisometropic amblyopia (-17.50 D) due to unilateral megalophthalmos associated with neurofibromatosis type 1 (NF1) 2). The axial lengths were 22 mm and 27 mm in the right and left eyes, respectively, showing a marked difference. Because early ophthalmology referral was not made, the amblyopic eye had deteriorated to light perception only, and stereopsis was completely lost at the time of discovery.
7. Latest research and future perspectives (research-stage reports)
Section titled “7. Latest research and future perspectives (research-stage reports)”Dichoptic Digital Therapy
Section titled “Dichoptic Digital Therapy”This treatment presents images of different contrast or content to each eye to encourage use of the amblyopic eye 3). Games and video viewing using VR headsets or tablets are being attempted.
Halicka et al. (2021) reported that a 22-year-old adult with anisometropic amblyopia underwent 44 hours of dichoptic training in a VR environment, and the corrected visual acuity of the amblyopic eye improved from 0.05 to 0.5 4). Furthermore, stereopsis was gradually acquired through training, and fMRI showed changes in visual cortex activity patterns. Visual acuity of 0.4 was maintained one year after training.
Xiao (Luminopia) et al. reported that 72 hours of headset use in children resulted in a visual acuity improvement of 0.15 logMAR 3). In adults with anisometropic amblyopia, the dichoptic treatment group also showed a visual acuity improvement of 0.15 logMAR (one line improvement per 27 hours) 3).
It is not yet clear whether dichoptic therapy is superior to conventional occlusion therapy 3). Halicka et al. reported that a 22-year-old adult with anisometropic amblyopia underwent 44 hours of dichoptic training in a VR environment, and the corrected visual acuity of the amblyopic eye improved from 0.05 to 0.5, with gradual acquisition of stereopsis 4).
Treatment Potential for Adult Amblyopia
Section titled “Treatment Potential for Adult Amblyopia”Conventionally, amblyopia in adults beyond the critical period of visual development was considered difficult to treat. However, animal experiments and human studies have shown that some degree of plasticity in the visual pathway remains even after the critical period 4).
Perceptual learning, anti-suppression training, and dichoptic training in VR environments have been attempted for adult amblyopia, with reported improvements in visual acuity and stereopsis 3)4). Further research is needed on the stability of long-term effects and comparisons with existing treatments.
Halicka et al. (2021) reported that a 22-year-old adult with anisometropic amblyopia underwent 44 hours of dichoptic training in a VR environment, and the corrected visual acuity of the amblyopic eye improved from 0.05 to 0.5 4). Furthermore, stereopsis was gradually acquired through training, and fMRI showed changes in visual cortex activity patterns. Visual acuity of 0.4 was maintained one year after training 4).
Pharmacological Adjunctive Therapy
Section titled “Pharmacological Adjunctive Therapy”Attempts have been reported to enhance the treatment effect of amblyopia by combining levodopa (a dopamine precursor) with patching. A multicenter randomized controlled trial by PEDIG is being conducted.
Treatment After the Critical Period (Ages 7–17)
Section titled “Treatment After the Critical Period (Ages 7–17)”In the PEDIG study (ATS 3), 25–23% of previously untreated children aged 7–17 years who received only spectacle correction for 24 weeks achieved a visual acuity improvement of 0.2 logMAR or more 8). When additional occlusion of 2–6 hours was added, 53% of the 7–12-year-old group improved by 0.2 logMAR or more. This result refutes the old dogma that treatment is limited to the sensitive period (up to 8 years of age) and indicates that treatment should not be abandoned based on age alone 1).
Even for amblyopia detected after school age (8–12 years), active treatment intervention is now the standard of care, supported by data from the PEDIG study 1).
Compliance Management and Family Support
Section titled “Compliance Management and Family Support”Compliance with amblyopia treatment (actual occlusion time / prescribed occlusion time) is reported to average less than 50%, and decreases with higher prescribed doses. Especially for occlusion of 6 hours or more, the impact on a child’s daily life is significant, and family cooperation and support systems are key to treatment success 3).
Practical strategies to improve compliance include the following:
- Motivation: Visualization of vision improvement goals (vision record cards/graphs)
- Activity planning: Specification of activities that actively use the amblyopic eye during occlusion (reading, games, crafts)
- Gradual introduction: Start with 1 hour and increase time as the child adapts
- Electronic monitoring: Occlusion dose monitors are used in research, but calendar records at home are useful as an alternative
- Regular visits: Vision checks and prescription adjustments every 2–3 months. After vision stabilizes, gradually taper off 1)
Amblyopia treatment often requires a long period (months to years), and continuous education and support for patients and parents are important to prepare for treatment interruption and relapse after discontinuation. The recurrence rate of amblyopia is reported to be about 24% within one year after treatment discontinuation 3), and vision checks every 3–6 months for at least one year after treatment completion are recommended 1). Since response to retreatment is often good in cases of recurrence, early detection and early re-intervention lead to improved prognosis.
8. References
Section titled “8. References”-
American Academy of Ophthalmology. Amblyopia Preferred Practice Pattern 2022 Update. Ophthalmology. 2023;130(3):P136-P178.
-
Mukit FA, Cape HT, Huq SS, et al. An isolated case of unilateral macro-ophthalmia with resultant anisometropic amblyopia in neurofibromatosis 1. Cureus. 2023;15(9):e44679.
-
Meier K, Tarczy-Hornoch K. Recent treatment advances in amblyopia. Annu Rev Vis Sci. 2022.
-
Halicka J, Bittsansky M, Sivak S, et al. Virtual reality visual training in an adult patient with anisometropic amblyopia: visual and functional magnetic resonance outcomes. Vision. 2021;5(2):22.
-
Pediatric Eye Disease Investigator Group. Treatment of anisometropic amblyopia in children with refractive correction. Ophthalmology. 2006;113:895-903.
-
Pediatric Eye Disease Investigator Group. A randomized trial of prescribed patching regimens for treatment of severe amblyopia in children. Ophthalmology. 2003;110:2075-2087.
-
Pediatric Eye Disease Investigator Group. A randomized trial of atropine vs. patching for treatment of moderate amblyopia in children. Arch Ophthalmol. 2002;120:268-278.
-
Pediatric Eye Disease Investigator Group. Randomized trial of treatment of amblyopia in children aged 7 to 17 years. Arch Ophthalmol. 2005;123:437-447.
-
Kanclerz P, Przewłócka K, Arnold RW. Agreement in non-cycloplegic and cycloplegic refraction between a photoscreener and a calibrated autorefractor. BMC Ophthalmol. 2024;24:130. doi:10.1186/s12886-024-03394-0
-
日本眼科学会屈折矯正委員会. 屈折矯正手術のガイドライン(第8版). 日眼会誌. 2024;128(2):135-139.
