Pinguecula
Location: Remains on the bulbar conjunctiva and does not invade the cornea.
Shape: Yellow-white elevation. Triangular to elliptical.
Treatment: Usually observation. Eye drops for inflammation.

A pinguecula is a yellowish-white to yellowish-brown small elevation that develops on the bulbar conjunctiva in the interpalpebral region, with the corneal limbus as its base. It is a fibrofatty degenerative tissue that does not invade the cornea. The name derives from the Latin word pinguis (fat). It is often structureless, but occasionally a lobulated internal structure may be observed. It frequently forms a triangle with the corneal side as the base, and may also appear oval or irregularly shaped.
Prevalence increases with age, and after the age of 50, it is observed to some degree in most people. It is the most common conjunctival degeneration and one of the most noticeable age-related changes. It has long been known to occur more frequently in low-latitude regions, suggesting a causal relationship with ultraviolet exposure. It commonly occurs on the nasal side but can also appear on the temporal side or bilaterally. It is usually binocular.
Pinguecula increases sharply with age. In a Spanish population-based epidemiological study (O Salnés study), the prevalence of pinguecula in individuals aged 40 and older was reported to be approximately 47.9%, with 47.2% on the nasal side and 6.0% on the temporal side1). The same study found that pinguecula was significantly more frequent than pterygium, and age and outdoor activity time were identified as independent risk factors1). A community-based survey in South India reported even higher prevalence rates, particularly among outdoor workers and the elderly, with outdoor work hours and age identified as risk factors2). While population-based studies specific to Japan are limited, it is almost universally observed as an age-related change after the age of 50.
Pinguecula rarely causes symptoms despite its high prevalence and is often discovered incidentally during routine examinations. However, epidemiologically, the presence of pinguecula itself is recognized as a risk factor for conjunctivochalasis and dry eye disease, and is also listed as a representative example of anatomical irregularity of the ocular surface in the TFOS DEWS III report3).
ICD-10 code: H11.1.
Pinguecula is a non-malignant age-related change and does not become malignant. It may slowly enlarge but does not cause visual impairment. However, differentiation from other conjunctival lesions such as conjunctival intraepithelial neoplasia (CIN) or conjunctival nevus may be necessary. For details, see the Diagnosis and Examination Methods section.
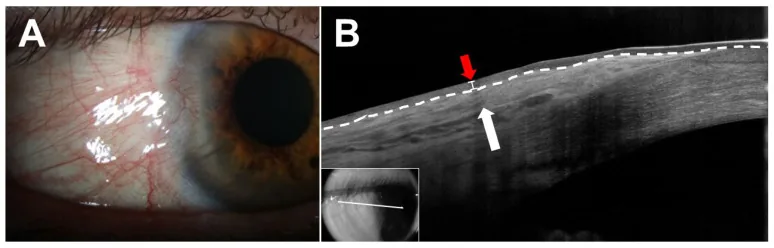
Most pingueculae are asymptomatic. Patients often have no complaints other than being concerned about their appearance, and are discovered incidentally during health checkups or examinations for other diseases.
When symptoms occur, the following findings are representative.
Ultraviolet (UV) exposure is deeply involved in the development of pinguecula, and a pathogenesis similar to that of pterygium formation is suspected3,4). Proteins such as collagen and elastin in the subconjunctival tissue are thought to undergo post-translational modifications such as glycation or racemization, becoming resistant to degradation and forming abnormal aggregates.
At the molecular level, elevated nuclear p53 protein expression due to UV-induced DNA damage response has been reported in both pinguecula and pterygium4). In pterygium, increased expression of p53 and MDM2 (mouse double minute 2) has been confirmed, and a similar mechanism is thought to be involved in pinguecula, which lies on the same UV-induced degenerative spectrum.
The reason why both pterygium and pinguecula occur more frequently on the nasal side is thought to be that light passing through the cornea medially focuses on the area of the nasal limbus, while the shadow of the nose reduces light intensity on the temporal side. This optical focusing mechanism is widely cited as a pathological model for pterygium4).
Pinguecula
Location: Remains on the bulbar conjunctiva and does not invade the cornea.
Shape: Yellow-white elevation. Triangular to elliptical.
Treatment: Usually observation. Eye drops for inflammation.
Pterygium
Location: Triangular invasion from the conjunctiva onto the cornea.
Shape: White membranous tissue rich in blood vessels. Destroys Bowman’s membrane.
Treatment: Surgery when visual function is impaired (e.g., conjunctival flap transplantation).
Some believe that pinguecula can be a precursor to pterygium. Inflammatory pinguecula with nasal corneal epithelial defects is considered to carry a high risk of progression to pterygium. However, not all pingueculae progress to pterygium, and progression is slow.
Pinguecula can be easily diagnosed with slit-lamp microscopy. Special tests are usually unnecessary.
Biopsy for histopathological confirmation is usually unnecessary, but may be indicated in atypical cases to differentiate from conjunctival intraepithelial neoplasia (CIN).
| Condition | Key points for differential diagnosis |
|---|---|
| Pterygium | Triangular invasion onto the cornea. Destroys Bowman’s membrane. |
| Pseudopterygium | Scar tissue from conjunctival adhesion to the cornea after trauma or inflammation |
| Conjunctival intraepithelial neoplasia (CIN/OSSN) | Hyperemia, pigmentation, papillary growth. Atypical cases require biopsy. |
| Corneal limbal dermoid | Congenital, yellowish-white elevation, straddling the corneal limbus |
| Conjunctival nevus | Pigmented lesion, present since childhood |
The essence of pinguecula is elastic fiber degeneration beneath the conjunctival epithelium.
Asymptomatic pinguecula does not require treatment; observation alone is sufficient. It is important to explain to the patient that there is no risk of malignant transformation and that it is an age-related change that may enlarge slowly.
When inflammation occurs in the pinguecula (pingueculitis), topical eye drop treatment is administered. Representative Japanese ophthalmology treatment guidelines recommend the following combination regimen.
Additionally, nonsteroidal anti-inflammatory drug (NSAID) eye drops (such as indomethacin ophthalmic solution) are used to reduce inflammatory symptoms. The fact that “steroid eye drops are effective in treating pingueculitis” is also emphasized in Japanese slit-lamp microscopy clinical textbooks.
For dry eye-like symptoms caused by abnormal tear distribution, artificial tears or sodium hyaluronate ophthalmic solution (0.1% or 0.3%) are prescribed to stabilize the tear film.
Steroid eye drop use should generally be limited to the short term. Long-term use carries risks of increased intraocular pressure and cataracts, so the dosage should be reduced and discontinued promptly after symptom improvement.
Surgical excision is considered when medical treatment is ineffective, when it interferes with contact lens wear, or when it is cosmetically prominent. At the pinguecula stage, active excision is rarely performed, and conservative treatment is prioritized.
Pinguecula is listed in the TFOS DEWS III report as a representative example of anatomical abnormalities of the ocular surface, and it is clearly indicated that it induces and exacerbates dry eye through shortened tear film breakup time (TBUT) and tear distribution abnormalities3). Clinically, the following stepwise treatment is recommended.
Pinguecula and conjunctivochalasis share risk factors, and the presence of pinguecula has also been reported as an independent risk factor for conjunctivochalasis3). In cases where poor tear dynamics lead to refractory symptoms, evaluate both conditions together.
Surgical removal is possible, but there is a risk of recurrence, and redness may not completely disappear. Removal for cosmetic reasons alone should be carefully considered; conservative treatment (artificial tears, low-concentration steroid eye drops) is generally tried first. Surgery may be indicated when pingueculitis recurs repeatedly or when it interferes with contact lens wear.
Pinguecula is essentially caused by degeneration of collagen fibers beneath the conjunctival epithelium and accumulation of abnormal proteins. Ultraviolet exposure disrupts the collagen fibers under the conjunctival epithelium, leading to elastoid degeneration. When proteins such as collagen and elastin undergo post-translational modifications such as glycation or racemization, they become resistant to protease degradation. These degradation-resistant proteins accumulate as abnormal aggregates, forming yellowish-white elevations.
Increased nuclear p53 protein expression is observed in the epithelium of pinguecula and pterygium. p53 is a tumor suppressor that normally induces apoptosis or cell cycle arrest in response to cellular stress. In pterygium studies, both p53 and its antagonist MDM2 are strongly expressed, with p53 trapped in the cytoplasm and unable to exert transcriptional activity4). Furthermore, it has been shown that p53 reactivation by the MDM2 antagonist Nutlin can selectively induce apoptosis in pterygium cells4). Pinguecula is thought to lie on the same UV-induced degenerative spectrum as pterygium, and these molecular mechanisms may also be applied to understanding the pathogenesis of pinguecula.
The elevation of pinguecula alters the alignment between the eyelid and the ocular surface, affecting tear film distribution and function3). When the elevation is pronounced, discontinuity of the tear meniscus occurs, forming a dry delle (dell) in the adjacent peripheral cornea. The TFOS DEWS III report treats pinguecula as a representative example of “anatomical irregularity of the ocular surface,” and its contribution to dry eye pathology is clearly stated3). It has also been suggested that when ocular surface irregularity improves after surgical excision, the signs and symptoms of dry eye are reduced3).