Gelatinous
Translucent elevated mass: The most common form. Conjunctival vessels show a hairpin-like course.
Feeder vessels: Abnormally dilated and tortuous feeding arteries suggest malignant growth 1).

The term was proposed by Lee et al. in 1995 and encompasses non-complex squamous epithelial tumors arising from the cornea, conjunctiva, and limbus 1). It is the most common non-pigmented tumor of the ocular surface; in a single-center study, OSSN accounted for 23% (179 cases) of 771 non-melanocytic conjunctival tumors 1).
OSSN includes the following lesions:
The incidence varies greatly by geographic region. In high-latitude areas, it is 0.02 per 100,000, while in low-latitude regions near the equator, it reaches 3.5 per 100,0001). The global age-standardized incidence rate is 0.26 per 100,000 per year and is increasing1). Africa has the highest rate (3.4 per 100,000 per year), with high frequencies of HIV and HPV infection contributing1).
In the Western Hemisphere, it commonly occurs in white men over 60 years old, but in parts of Africa and Asia, it also affects younger individuals with HIV infection and follows a more aggressive course1).
The AJCC 8th edition classifies OSSN based on primary tumor (T), regional lymph nodes (N), and distant metastasis (M)1).
| Classification | Definition |
|---|---|
| Tis | Carcinoma in situ (confined to epithelium) |
| T1 | Confined to conjunctiva, no invasion of adjacent structures |
| T2 | Invasion of adjacent ocular structures (cornea, fornix, caruncle, sclera, globe) |
| T3 | Invasion of orbit, paranasal sinuses, or eyelid |
| T4 | Invasion of central nervous system or distant sites |
Untreated SCC can lead to orbital invasion (approximately 10%), regional lymph node metastasis, and rarely distant metastasis, with reported mortality rates of 8–24% 1).
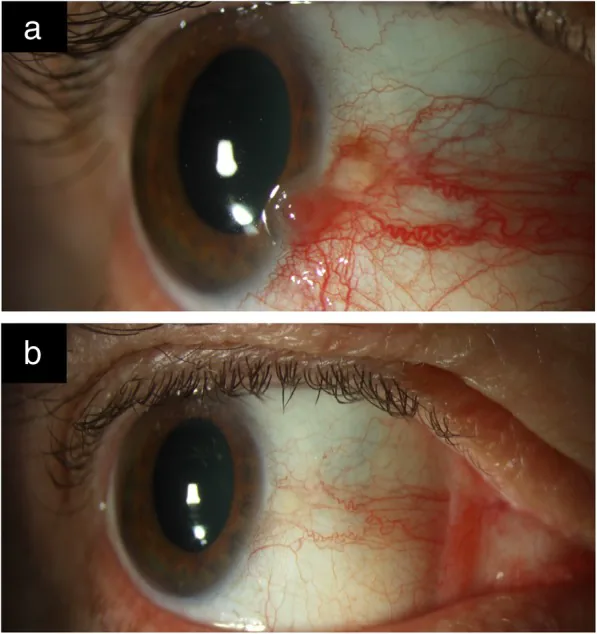
OSSN lesions are often asymptomatic and discovered incidentally. When symptoms are present, they mainly include redness, foreign body sensation, and ocular irritation 2). Pain may also occur. Visual loss is seen when the tumor involves the cornea or in cases of intraocular invasion.
A unilateral vascularized mass is most common, typically occurring near the limbus in the interpalpebral fissure (nasal or temporal side) 1). It frequently involves the cornea and bulbar conjunctiva, while palpebral conjunctival involvement is rare 1). In Caucasians, the lesion appears yellow-pink; in pigmented races, it may be pigmented 1)3).
Gelatinous
Translucent elevated mass: The most common form. Conjunctival vessels show a hairpin-like course.
Feeder vessels: Abnormally dilated and tortuous feeding arteries suggest malignant growth 1).
Leukoplakic
White thickened surface: Keratinization of the tumor surface due to hyperkeratosis.
Dry appearance: It presents an opaque white color, in contrast to gelatinous.
Papillomatous
Papillomatous growth: Known as an HPV-associated morphology 4).
Nodular surface: May have a wart-like appearance.
Noduloulcerative
Rare but invasive: A morphology strongly suggestive of invasive neoplasm 1).
Ulceration: Worse prognosis than other forms.
Corneal involvement presents a translucent, grayish ground-glass appearance with fimbriated or pseudopodial extension.
Intraocular invasion is rare but may present as a white mass in the anterior chamber, anterior chamber inflammation, or secondary glaucoma 2).
Yes. Especially in HIV-positive individuals of color, CIN has been reported to mimic pigmented lesions 3). Even in the absence of typical OSSN findings, conjunctival pigmented lesions in HIV-positive patients should include OSSN in the differential diagnosis.
The etiology of OSSN is multifactorial. Major risk factors are listed below 1).
Because immunosuppression due to HIV infection impairs tumor immune surveillance. In HIV-positive individuals, OSSN risk increases 10–13 times 3), and it can occur at a young age. In Africa, co-infection with HIV and HPV is considered a contributing factor to the increase in the disease 1).
The definitive diagnosis of OSSN is histological examination by excisional or incisional biopsy (gold standard) 1). For small tumors less than 4 hours, excisional biopsy is indicated; for large tumors 4 hours or more, incisional biopsy is appropriate.
Special stains such as rose bengal, lissamine green, methylene blue, and toluidine blue are used to aid diagnosis 1). Rose bengal stains apoptotic and metabolically inactive epithelial cells a vivid pink, clarifying the tumor margins 1).
The main diagnostic modalities are summarized below.
| Test | Features |
|---|---|
| Biopsy (histopathology) | Gold standard. Can assess depth of invasion. |
| HR-OCT | Noninvasive. Characterized by epithelial thickening and hyperreflectivity. |
| IVCM | Cellular-level assessment. Technically demanding. |
| Impression cytology | Noninvasive but only evaluates surface tissue. |
OSSN must be differentiated particularly from pterygium and pinguecula 1). They may coexist, making diagnosis challenging. Other differentials include corneal pannus, actinic keratosis, pyogenic granuloma, conjunctival nevus, and malignant melanoma.
When clinical findings alone make differentiation difficult, epithelial thickening and hyperreflectivity on HR-OCT, as well as special staining with rose bengal or methylene blue, can provide clues for differential diagnosis 1). Biopsy is required for definitive diagnosis.
Treatment of OSSN is individualized based on tumor size, depth of invasion, and patient factors 1). Surgical excision and topical chemotherapy have been reported to have equivalent efficacy 1).
No-touch technique plus cryotherapy is the gold standard 1).
If the excision margins are positive, postoperative adjuvant chemotherapy is recommended 1)2). Without cryotherapy or postoperative chemotherapy, the risk of intraocular invasion increases 2).
Pike et al. (2023) reported a case of OSSN in which, without cryotherapy or chemotherapy after excision, intraocular seeding (anterior chamber mass formation) occurred through a full-thickness limbal defect, leading to enucleation 2). This highlights the importance of the no-touch technique, cryotherapy, and postoperative chemotherapy.
Topical chemotherapy is used both as monotherapy and as postoperative adjuvant therapy 1).
Mitomycin C
Mitomycin C: 0.02–0.04% eye drops. It kills tumor cells by crosslinking DNA.
Efficacy rate: 80–100%. Time to resolution tends to be shorter than with IFN1).
Side effects: Ocular pain, limbal stem cell deficiency, risk of punctal stenosis.
5-FU
5-Fluorouracil: A pyrimidine analog that inhibits DNA and RNA synthesis in the S phase1).
Efficacy rate: High. Recurrence rate up to 20%. Less expensive than mitomycin C and IFN.
Administration: Used in cycles of 1% eye drops for 1 week followed by 3 weeks off.
IFNα-2b
Interferon α-2b: Has antiproliferative, antiviral, and immunomodulatory effects1).
Efficacy rate: 80–100%. Administered via subconjunctival injection (1 million IU/mL) or eye drops.
Features: Few side effects but expensive and requires refrigeration.
Other drug therapies include anti-VEGF agents (bevacizumab, ranibizumab), which show promising results for conjunctival lesions but unclear effects on corneal lesions; large-scale trials are needed1). Cidofovir may be used in HPV-related patients1).
The treatment approach based on the AJCC 8th edition is shown below1).
The following factors are associated with recurrence1).
Chemotherapy alone (MMC, 5-FU, IFNα-2b) has been reported to be as effective as surgery1). The choice depends on tumor size, depth of invasion, patient’s general condition, and adherence. For lesions larger than 4 hours, multiple lesions, or recurrent cases, topical chemotherapy may be advantageous.
OSSN is thought to arise from limbal stem cells 1). Most primary conjunctival malignancies occur near the limbus within the interpalpebral fissure, where stem cell division is active 1).
Histologically, in invasive SCC, malignant squamous epithelial cells penetrate the basement membrane and proliferate in the stroma 1). CIN I to III are classified by the extent of dysplasia within the epithelium. Full-thickness epithelial dysplasia (CIN III) is synonymous with carcinoma in situ 1).
Intraocular invasion is rare but occurs via the following routes 2):
In the case by Pike et al. (2023), a full-thickness limbal defect formed after excisional biopsy, through which tumor cells seeded into the anterior chamber and spread to the posterior corneal surface, angle, iris, ciliary body, and lens 2). Pathologically, the findings resembled a malignant form of epithelial downgrowth.
In mucoepidermoid carcinoma, mucin staining reveals dysplastic squamous epithelial cells and malignant goblet cells. When the mucin component is predominant, clinical aggressiveness is considered relatively low.
Radiation therapy is being considered as a treatment option for OSSN and SCC 1).
This is a treatment combining verteporfin and laser 1). In a pilot study, tumor disappearance was achieved in 100% of conjunctival SCC cases, with no recurrence during the observation period 1). However, high cost, limited facilities, and the need for specialized training are barriers to widespread adoption.
Zein et al. (2024) administered four intramuscular injections of the 9-valent HPV vaccine (Gardasil-9) at 6-week intervals to a patient with recurrent papillary OSSN (HPV-positive, p16-positive) after two resections 4). Dramatic tumor shrinkage was observed after the third injection, and HR-OCT confirmed improvement in epithelial thickening and hyperreflectivity. However, a cure was not achieved, and additional topical 5-FU and mitomycin C were administered.
This is the world’s first report of using the HPV vaccine for OSSN treatment 4), and research is ongoing as a future treatment option. The therapeutic mechanism is presumed to involve suppression of HPV-specific E6/E7 oncoproteins 4).