Simple episcleritis
Frequency: More common
Onset: Acute
Course: Peaks in about 12 hours and resolves in 2–3 days
Findings: Sectoral (about 67%) or diffuse (about 33%) hyperemia

Episcleritis is a benign, self-limited inflammatory condition of the episcleral tissue. It involves inflammation of the superficial vascular plexus, such as the Tenon’s capsule vessels, and is milder and less vision-threatening than scleritis, which affects deeper vessels. Most cases are idiopathic and recurrent, with a tendency for bilateral involvement. The incidence is reported as 41.0 per 100,000 person-years, with a prevalence of 52.6 per 100,000.
While it is a relatively common cause of red eye, it can be confused with conjunctivitis or scleritis, leading to misdiagnosis at initial presentation. In episcleritis, the scleral stroma itself is not involved, and progression to severe structural complications such as globe perforation is rare. However, in recurrent cases or those associated with systemic autoimmune diseases such as rheumatoid arthritis or granulomatosis with polyangiitis, treatment of the underlying disease and long-term follow-up are necessary. Understanding episcleritis not merely as an isolated ocular condition but as an ocular manifestation of systemic disease is crucial for recurrence management and prognosis improvement.
The Watson classification is widely used for the clinical classification of inflammatory diseases of the sclera and episclera. It broadly divides them into three groups by site: episcleritis, anterior scleritis, and posterior scleritis. Anterior scleritis is further subdivided by morphology into diffuse, nodular, and necrotizing (inflammatory/non-inflammatory). An important difference from anterior scleritis is that episcleritis has no necrotizing type and is morphologically classified into two types: simple (diffuse) and nodular. Since this classification reflects the depth of inflammation (superficial or deep) and the severity of progression and prognosis, determining the disease type at diagnosis forms the basis for treatment strategy and prognosis explanation. Episcleritis is positioned as the mildest and most prognostically favorable group in this classification.
Simple episcleritis
Frequency: More common
Onset: Acute
Course: Peaks in about 12 hours and resolves in 2–3 days
Findings: Sectoral (about 67%) or diffuse (about 33%) hyperemia
Nodular episcleritis
Frequency: Slightly less common
Onset: Gradual
Course: Symptoms tend to last longer than simple type
Findings: Localized episcleral nodule near the limbus (mobile)
The sclera consists of three layers: the episclera, scleral stroma, and lamina fusca. The episclera is a connective tissue containing blood vessels overlying the scleral stroma, understood as a fibroelastic structure located between the scleral stroma and Tenon’s capsule. It consists of an outer parietal layer (superficial episcleral capillary network) and a deep visceral layer (highly anastomotic vascular network), both derived from the anterior ciliary arteries. Most nerve fibers are branches of the trigeminal nerve. The episclera forms the episcleral vascular plexus between the rectus muscle insertions and the limbus, normally hidden by the conjunctiva, but when inflamed, it dilates and produces bright hyperemia. The episclera gradually becomes thinner toward the posterior part of the eye, where Tenon’s capsule becomes predominant.
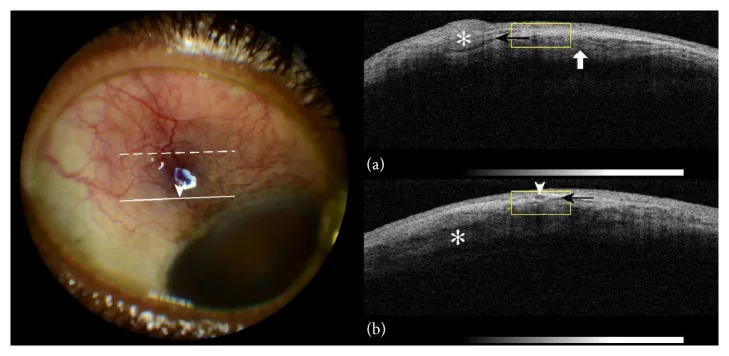
There is no tenderness and no discharge. If severe pain or obvious discharge is present, reconsider scleritis, infectious conjunctivitis, or anterior uveitis. Symptoms usually resolve or completely disappear within a few days, leaving no impact on visual function. Recurrences often occur in the same location or the contralateral eye, and patients often notice it as “the usual red eye.” Severe pain that disturbs nighttime sleep, as in scleritis, or strong tenderness when touching the upper eyelid is usually not observed in episcleritis.
Observation of the location and color of injection is central to differential diagnosis. The injection in episcleritis is bright red to pink, in contrast to the dark red (violaceous) deep injection seen in scleritis.
| Finding | Episcleritis | Scleritis |
|---|---|---|
| Color of injection | Bright red to pink | Dark red (violaceous) |
| Pain | Mild to none | Strong, radiating |
| Nodule mobility | Present | Absent |
Vision is generally normal. Complications such as conjunctival chemosis, elevated intraocular pressure, anterior uveitis, and keratitis are rare; if present, consider scleritis or other diseases. The absence of inflammatory findings in the palpebral conjunctiva is useful for distinguishing from conjunctivitis. In scleritis, inflammation may spread to surrounding tissues, causing peripheral corneal infiltration, ulceration, or anterior uveitis, whereas episcleritis is self-limiting and almost never involves adjacent tissues. Under slit-lamp examination, identify the level of the episcleral vascular plexus; if a red elevated lesion is present but the scleral vessels are not visible, consider the possibility of a neoplastic lesion.
Episcleritis is characterized by the absence of discharge and hyperemia localized near the corneal limbus. Conjunctivitis is usually painless, accompanied by discharge, and hyperemia is most prominent in the fornix, diminishing toward the limbus. Under slit-lamp examination, episcleral vessels are immobile, while conjunctival vessels are mobile. See the section on “Diagnosis and Testing Methods” for details.
Most cases are idiopathic (cause unknown), with systemic disease reported in approximately 26–36% of all cases. Even in idiopathic cases, an immunological mechanism is suggested, based on a nonspecific inflammatory reaction centered on lymphocytes in the superficial episcleral vascular plexus. The recurrent course and tendency for bilateral involvement suggest an underlying systemic immune dysregulation.
Collagen diseases/autoimmune diseases (most commonly rheumatoid arthritis) 1):
Vasculitis:
Infections: Bacteria, mycobacteria, syphilis, Lyme disease, herpes virus, herpes zoster, etc. can be causes. Episcleritis associated with ophthalmic herpes zoster is thought to be an immune response to the pathogen rather than the infection itself. There is also a report of a case of subconjunctival parasitosis due to Dirofilaria repens being misdiagnosed as episcleritis 7).
Others: Gout, atopy, foreign body, chemical trauma, medications (topiramate, pamidronate), and reports as an initial symptom of COVID-19.
Yes, about 30% of patients have an associated systemic disease. The most common is rheumatoid arthritis, but it can also be the initial symptom of diseases where early diagnosis and treatment affect prognosis, such as granulomatosis with polyangiitis (GPA) or Behçet’s disease. If recurrence is frequent or systemic symptoms are present, systemic evaluation including rheumatoid factor, antinuclear antibody, ANCA, and urinalysis is recommended.
Episcleritis is primarily a clinical diagnosis based on medical history and slit-lamp examination. The basic approach involves careful observation with a slit lamp of the level of scleral vessels (superficial or deep), the color of congestion, presence of nodules, and thinning or necrosis.
2.5% phenylephrine eye drops constrict conjunctival vessels and are useful for differentiating conjunctivitis from episcleritis. 10% phenylephrine eye drops constrict the superficial episcleral vascular network but not the deep network, allowing differentiation between episcleritis and scleritis.
The response test with 1:1000 epinephrine eye drops is a simple method to assess deep vessel involvement. If congestion subsides after instillation, it suggests episcleritis; if not, it suggests scleritis. A comprehensive evaluation is made by combining three factors: number and mobility of nodules, presence of pain/tenderness, and epinephrine response.
Response tests with epinephrine and phenylephrine are particularly useful as adjunctive diagnostics when the layered structure of congestion cannot be directly observed with a slit lamp or in cases with small nodules. Observation 10–15 minutes after instillation determines the presence of superficial vessel constriction; if deep vessel congestion remains, priority is given to managing scleritis.
Tenonitis is also considered a type of episcleritis, and clinical differentiation between the two is difficult. Judgment is made by combining nodule mobility, presence of pain/tenderness, epinephrine eye drop response, and fluorescein staining findings.
For isolated, mild episcleritis, extensive systemic workup is unnecessary. In cases of recurrence or when accompanied by systemic symptoms, consider the following tests.
In cases where episcleritis appears as the initial manifestation of granulomatosis with polyangiitis, renal dysfunction may coexist 3). When both ocular inflammation and renal function abnormalities are present, promptly investigate for systemic vasculitis including granulomatosis with polyangiitis. For refractory or recurrent episcleritis, it is desirable to evaluate disease activity and initiate treatment for the underlying disease in collaboration with rheumatology and internal medicine.
In addition to slit-lamp microscopy, evaluation of episcleral thickness and vascular patterns using anterior segment optical coherence tomography (AS-OCT), and assessment of scleral thickness using ultrasound (B-mode) may be used as ancillary diagnostic tools. To rule out necrotizing scleritis or evaluate posterior scleritis, ultrasound can check for the T-sign (fluid around the optic nerve sheath). In typical episcleritis, these imaging studies often lack specific findings, and diagnosis is made by combining direct slit-lamp examination with history and systemic evaluation.
Most cases of episcleritis resolve spontaneously within days to weeks without treatment. The first step in management is to explain the benign nature and natural course of the disease, the need for systemic evaluation, and to provide reassurance. Cold compresses and cooled artificial tears are effective in reducing subjective symptoms such as irritation and warmth. In mild cases, active pharmacological intervention is not required; short-term follow-up over several days to confirm spontaneous improvement can avoid rebound and side effects associated with treatment.
Low-concentration topical steroids are the first-line treatment. Antibiotic eye drops are often used concurrently to help differentiate from scleritis.
If response to topical treatment is poor, consider switching to evaluation and treatment for scleritis. While topical steroids rapidly suppress symptoms, long-term or repeated use may increase the risk of recurrence and induce rebound hyperemia.
Treatment should generally be tapered and discontinued after symptom resolution; avoid prolonged continuous administration. Long-term use of topical steroids carries risks of steroid-induced intraocular pressure elevation and posterior subcapsular cataract, so improvement should be confirmed within 1–2 weeks before tapering. In recurrent cases, assess disease activity for each episode individually and prioritize optimization of treatment for the underlying systemic disease.
In episcleritis associated with collagen diseases such as rheumatoid arthritis, treatment of the underlying disease directly affects prognosis 1). If local treatment is ineffective, add oral prednisolone (tapering from 20–30 mg/day). Cases requiring systemic steroids are very rare unless there is clear evidence of systemic inflammatory disease.
In episcleritis associated with granulomatosis with polyangiitis, remission induction therapy with cyclophosphamide or rituximab is effective 3)4). Rituximab has been reported to have a higher remission rate at 6 months compared to cyclophosphamide (64% vs. 53%) 3).
Steroid eye drops quickly suppress symptoms of episcleritis, but discontinuation may cause rebound redness and potentially lead to more severe recurrence. Therefore, the use of steroids is debated; for mild cases, some recommend observation without treatment or prioritizing NSAIDs. In recurrent cases, oral COX-2 inhibitors or evaluation for systemic disease are recommended.
The pathogenesis of episcleritis is not yet fully understood. In affected areas, vasodilation and congestion occur in the superficial episcleral vascular network, with infiltration of inflammatory cells, mainly lymphocytes, into the episclera and Tenon’s capsule. The essential difference from scleritis is that the scleral stroma itself is not involved. The inflammatory cell infiltrate consists mainly of T cells and a few plasma cells; neutrophilic purulent inflammation or granuloma formation is usually absent.
Histopathologically, it is a non-granulomatous inflammation, primarily characterized by vasodilation and lymphocytic infiltration. In nodular episcleritis, fibrinoid necrosis is observed in the center of the lesion, surrounded by a palisade of epithelioid cells. These findings resemble the granulomatous inflammation seen in scleritis, and some consider episcleritis and scleritis as a spectrum based on the depth of inflammation. The small-scale fibrinoid necrosis observed in episcleritis can be understood as a mild form of the more extensive necrotizing changes in scleritis.
Inflammation increases the production of reactive oxygen species (ROS) and enhances oxidative stress2). The total vitamin C content in the human retina is about 20 times higher than in plasma, and ocular tissues rely heavily on the antioxidant system. In autoimmune episcleritis, it has been suggested that dysfunction of this antioxidant system may lead to chronic inflammation and tissue damage in the episclera2). ROS damage vascular endothelium and induce the release of inflammatory cytokines, causing persistent vasodilation and increased permeability. Chronic exposure of the ocular surface and episclera to oxidative stress is considered a contributing factor to recurrent episcleritis, and the therapeutic potential of antioxidant intervention is being investigated.
Clinically, episcleritis rarely transitions directly to scleritis. However, since most cases of scleritis also show inflammation in the episclera (episcleritic changes), the two are understood as a continuum based on the depth of the vascular layer affected, rather than completely independent diseases. Episcleritis primarily involves the superficial episcleral vascular network (parietal layer), while scleritis involves the deep vascular network and scleral stroma.
At the rectus muscle insertion, the sclera is thinnest (about 0.3 mm), making it more vulnerable to inflammation and trauma. The episcleral vascular plexus receives abundant blood supply via the anterior ciliary arteries, so congestion becomes apparent rapidly during inflammation. In contrast, the sclera itself is a poorly vascularized tissue, making deep inflammation like scleritis rare. The anatomical feature that vessels derived from the anterior ciliary arteries reversibly congest in episcleritis provides the mechanistic basis for the rapid resolution of congestion with the epinephrine test; this response is absent in deep scleral vasculitis, serving as a pathophysiological basis for differentiation.
There is a case report of a 60-year-old man with idiopathic recurrent episcleritis who had no recurrence for 7 months after starting oral vitamin C 500 mg/day 2). Vitamin C is a potent antioxidant, and it has been suggested that it may suppress inflammation in ocular tissues by reducing oxidative stress. Ocular tissues are known to be highly dependent on the antioxidant system, with retinal vitamin C concentrations reaching about 20 times those in plasma, and supplementation with vitamin C and other antioxidant nutrients may be a candidate strategy for recurrence prevention 2). However, controlled case-control studies and clinical trials are needed to establish efficacy 2). At present, it remains at the stage of being considered adjunctively in cases with severe recurrent symptoms or with underlying dry eye or chronic ocular surface inflammation.
Granulomatosis with polyangiitis is a fatal disease with a 1-year mortality rate of up to 80% if untreated, but immunosuppressive therapy can reduce mortality to 10% 3). Since episcleritis can be the initial symptom of GPA, ophthalmologists should recognize this association and actively perform systemic evaluation in recurrent episcleritis 3)4). In particular, the coexistence of ocular inflammation and renal dysfunction is a finding strongly suggestive of granulomatosis with polyangiitis 3).
The efficacy of biologic agents such as TNFα inhibitors and rituximab has been reported for episcleritis and scleritis associated with rheumatoid arthritis 1). Infliximab and adalimumab have a track record in rheumatoid arthritis and uveitis, and their application to refractory scleritis and episcleritis is being considered. On the other hand, etanercept is known to cause paradoxical reactions that induce or exacerbate ocular inflammation, so caution is required in drug selection 1). Rituximab is a monoclonal antibody targeting B cells, and its efficacy for vasculitis-related ocular inflammation has been suggested. The use of these biologic agents is determined in close collaboration with rheumatology and collagen disease departments.
Cases have been reported where patients diagnosed with episcleritis actually had intraocular metastatic tumors 6) or subconjunctival parasitic infections 7). In refractory or recurrent episcleritis, it is important to rule out malignant diseases and infections. Imaging studies and detailed evaluation of slit-lamp findings of the mass, including its vascularity, provide diagnostic clues. Assessment should integrate the mobility of the mass, visibility of scleral vessels, adhesion to surrounding tissues, and treatment response. Persistent elevated lesions that do not respond to standard steroid eye drops warrant consideration of biopsy and advanced imaging.
Long-term observational studies on the natural course of episcleritis and the time to manifestation of systemic diseases are limited. In particular, data on incidence rates and profiles of associated diseases in the Japanese population are insufficient. Previous reports from Western countries indicate an annual incidence of approximately 40–60 cases per 100,000 people, but the figures vary due to differences in ethnicity, living environment, and uveitis registry operations. Future establishment of clinical registries and multicenter collaborative studies are expected to identify risk factors for recurrence and the timeline for the emergence of systemic diseases.
- Promelle V, Goeb V, Gueudry J. Rheumatoid Arthritis Associated Episcleritis and Scleritis: An Update on Treatment Perspectives. J Clin Med. 2021;10(10):2118.
- Goyal L, Ajmera K, Pandit R. Reoccurring Episcleritis and the Role of Antioxidants. Cureus. 2022;14(4):e24111.
- Foster LD, Nyugen M, Margolin E. Conjunctivitis, episcleritis and anterior uveitis as the first presenting features of granulomatosis with polyangiitis. BMJ Case Rep. 2021;14:e243558.
- Ciotoracu AC, Dimăncescu MG, Mitulescu TC, et al. A clinical case of recurrent episcleritis as the initial manifestation of granulomatosis with polyangiitis. Rom J Ophthalmol. 2021;65(4):386-390.
- Jari M, Nasiri S, Ghandehari M. Episcleritis and posterior uveitis misdiagnosed as orbital cellulitis in a child patient with Behçet’s disease. SAGE Open Med Case Rep. 2023;11:1-4.
- Chong YJ, Azzopardi M, Ng B, et al. Ocular Metastasis as First Presentation of Large-Cell Neuroendocrine Carcinoma. Case Rep Ophthalmol. 2023;14:684-691.
- Redón-Soriano M, Blasco A, Gomila B, et al. Subconjunctival human dirofilariasis by Dirofilaria repens in the Mediterranean Basin. Am J Ophthalmol Case Rep. 2022;26:101570.