Cytotoxic chemotherapy
5-FU: Tearing (lacrimal duct stenosis), conjunctivitis, corneal epithelial damage1)
Cisplatin: Optic neuropathy, color vision disturbance (rare)1)
Taxanes (docetaxel, paclitaxel): Tearing, cystoid macular edema4)
Ocular side effects of anticancer drugs and molecular targeted drugs are a general term for conditions in which drugs used for cancer treatment and immune disease treatment cause adverse events in ocular tissues.
The categories of drugs involved are extensive.
With advances in cancer treatment, survival has been prolonged, and the importance of managing ocular side effects during long-term therapy is increasing1). Ocular side effects range from mild discomfort to irreversible vision loss, and appropriate screening and early intervention directly affect visual prognosis.
If symptoms such as decreased vision, redness, eye pain, floaters, photopsia, or metamorphopsia (distorted vision) occur, promptly see an ophthalmologist. Even if symptoms are mild, neglecting them can lead to irreversible damage, so it is important to report to your primary oncologist and coordinate with an eye doctor.
Ocular side effects vary by drug category. The main drugs and their ocular symptoms are shown below.
Cytotoxic chemotherapy
5-FU: Tearing (lacrimal duct stenosis), conjunctivitis, corneal epithelial damage1)
Cisplatin: Optic neuropathy, color vision disturbance (rare)1)
Taxanes (docetaxel, paclitaxel): Tearing, cystoid macular edema4)
Hormone therapy and immunomodulators
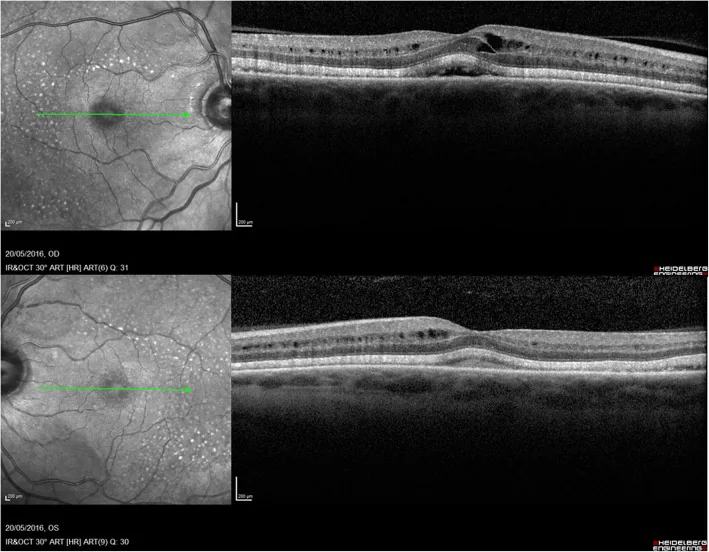
Tamoxifen: Perifoveal crystalline deposits, corneal vortex keratopathy, CME. Incidence: 3–6% overseas, 0.2% in Japan.
HCQ/chloroquine: Bull’s eye maculopathy (annular atrophy), decreased vision, night blindness, color vision abnormalities. Approximately 0.5% after 5 years of use.
Interferon: Small hemorrhages and cotton-wool spots at the posterior pole of the fundus. Occur within 2–3 months after administration. Mostly asymptomatic and resolve spontaneously.
Molecular Targeted Drugs
MEK inhibitors (e.g., trametinib): Serous retinal detachment and PED. Incidence approximately 10–25%2)
EGFR inhibitors (e.g., gefitinib): Trichomegaly, blepharitis, corneal epithelial disorders5)
BRAF inhibitors (e.g., vemurafenib): Uveitis, serous retinal detachment6)
ALK inhibitors (e.g., crizotinib): Photopsia, blurred vision. Approximately 60% develop visual disturbances7)
Immune Checkpoint Inhibitors (ICIs)
Uveitis (anterior, posterior, panuveitis): Incidence approximately 1%3)
VKH-like syndrome: Severe form requiring high-dose steroids3)
Dry eye, conjunctivitis, orbital inflammatory disease, external ophthalmoplegia8)
ADC (e.g., T-DXd): Corneal disorders (punctate superficial keratitis, corneal stromal opacity)10)
Fingolimod (Imusera/Gilenya), used to prevent relapse of multiple sclerosis, is known to cause macular edema. The incidence of macular edema is 0.2% with fingolimod 0.5 mg/day and 1.4% with 1.25 mg/day, and most cases occur within 3–4 months after starting treatment.
Risk factors for ocular adverse effects differ by drug category.
| Drug Category | Major Risk Factors |
|---|---|
| HCQ/Chloroquine | Treatment duration >5 years, CQ >3.0 mg/kg/day or HCQ >6.5 mg/kg/day, elderly, renal/hepatic impairment, concurrent retinal disease |
| Tamoxifen | High dose and long-term use (increased cumulative dose) |
| Interferon | High initial dose, elderly, diabetes, hypertension, anemia |
| MEK Inhibitors | Dose-dependent (higher frequency with higher doses) 2) |
| ICI | Ocular irAEs are more likely in patients with systemic irAEs (e.g., thyroiditis, colitis) 3) |
| ADC | Drug-specific corneal toxicity mechanism 10) |
Specific risk factors for HCQ retinopathy include the following.
Interferon retinopathy is more frequent and severe in patients with systemic conditions such as diabetes, hypertension, and anemia, and is also more likely to occur in treatment-resistant or recurrent cases.
Based on the recommendations of the American Academy of Ophthalmology (AAO)9), the following screening is performed.
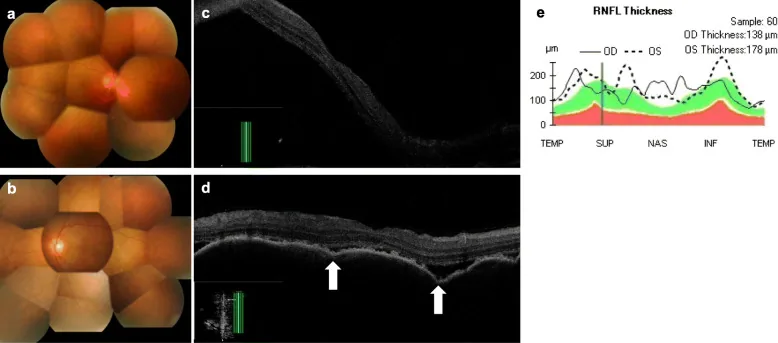
Useful tests include Humphrey 10-2 central visual field testing, SD-OCT, fundus autofluorescence, multifocal ERG, and full-field ERG. Bull’s eye maculopathy (annular atrophy) is a characteristic fundus finding in advanced HCQ retinopathy.
| Drug | Screening timing and tests |
|---|---|
| HCQ/Chloroquine | Baseline at start of treatment + annually from year 5 onward (SD-OCT, 10-2 visual field, FAF) 9) |
| Tamoxifen | At symptom onset and periodic fundus examination, SD-OCT |
| Interferon | Fundus examination 2–3 months after starting treatment. Prompt ophthalmology consultation if symptoms occur. |
| MEK inhibitors | OCT to confirm serous retinal detachment when decreased vision or metamorphopsia occurs 2) |
| ICI | Prompt ophthalmology consultation when decreased vision, redness, or eye pain occurs 3) |
| Fingolimod | Baseline fundus examination before treatment + follow-up 3–4 months after starting |
In patients using ICI who develop decreased vision, redness, or eye pain, evaluate for anterior chamber inflammatory cells, vitreous opacities, and choroidal lesions, and consider the possibility of uveitis or VKH-like syndrome as an irAE 3).
At the start of treatment, it is recommended to undergo an eye examination including visual acuity, fundus examination, and SD-OCT as a baseline. Thereafter, if there are no risk factors (treatment duration ≥5 years, high dose, advanced age, renal dysfunction, etc.), annual screening should begin after 5 years. If risk factors are present, earlier and more frequent examinations are needed. SD-OCT, Humphrey 10-2 visual field test, and fundus autofluorescence are the main tests.
Discontinuation of the drug is the only treatment. Because elimination from the body is slow, the condition may progress or worsen even after stopping the medication. Therefore, early detection is extremely important, and the decision to discontinue the drug before irreversible damage occurs determines the visual prognosis.
Most cases have a good prognosis; if asymptomatic, the basic approach is to wait until the end of IFN therapy. In severe cases or those with vision loss, consider reducing or discontinuing IFN. Early intervention is needed in patients with diabetes or hypertension.
Manage according to CTCAE grade 3).
In many cases, improvement occurs upon discontinuation of administration. If necessary, NSAIDs eye drops or steroid eye drops are used concomitantly.
Discontinuation is not always necessary. The management differs depending on the severity of ocular side effects (CTCAE grade) and the type of drug. While discontinuation is the basic approach for conditions such as HCQ retinopathy and tamoxifen retinopathy, in cases of mild serous retinal detachment due to MEK inhibitors or trichomegaly due to EGFR inhibitors, continuation of administration while performing symptomatic treatment may be chosen. It is important for the oncologist and ophthalmologist to collaborate and make decisions based on the individual situation.
The mechanisms of damage to ocular tissues differ by drug category.
CQ and HCQ have high affinity for melanin and bind to melanin granules in retinal pigment epithelium (RPE) cells, accumulating intracellularly. Accumulated CQ/HCQ is taken up into lysosomes, causing an increase in lysosomal pH and impairing lysosomal enzyme activity. When the metabolic function of RPE enzymes is impaired, the degradation and renewal of photoreceptor outer segments are disrupted, leading to photoreceptor degeneration. Due to the large amount accumulated in the body, damage may progress even after discontinuation.
Spherical lesions (degeneration of nerve fibers and axonal swelling) form in the nerve fiber layer and inner plexiform layer. Crystalline deposits are thought to be degenerative products containing calcium and complex carbohydrates deposited within nerve fibers. Cystoid macular edema (CME) occurs due to Müller cell dysfunction and fluid accumulation.
IFN induces vasospasm, leukocyte infiltration, and vascular endothelial damage, and immune complexes deposit in capillaries, causing capillary occlusion. Cotton-wool spots (nerve fiber layer infarcts) occur anterior to the occluded capillaries (on the arterial side), and hemorrhages from occluded areas are observed as small hemorrhages. Most are transient and resolve after IFN discontinuation.
MEK inhibitors block the MAPK/ERK pathway (cell proliferation and survival signals). Since RPE maintains active transport (ions and water) via ERK signaling, ERK inhibition impairs RPE ion transport and pump function. This leads to subretinal fluid accumulation, manifesting as serous retinal detachment and retinal pigment epithelial detachment (PED)2).
ICIs (PD-1/PD-L1 inhibitors, CTLA-4 inhibitors) release T-cell immune checkpoints and enhance antitumor immunity. However, this mechanism also releases autoimmune suppression, triggering autoimmune reactions in ocular tissues (uvea, sclera, orbit, lacrimal gland). In VKH-like syndrome, self-reactive T cells against melanocytes (choroid, ciliary body, iris) are activated, leading to panuveitis, serous retinal detachment, and depigmented lesions 3).
To understand the overall picture of ICI-related ocular irAE, international multicenter registry studies are underway 3). By clarifying the spectrum, frequency, and outcomes of various ocular irAEs such as uveitis, VKH-like syndrome, and extraocular myositis, risk stratification and protocol establishment are expected.
ADCs such as trastuzumab deruxtecan (T-DXd) are becoming widespread as next-generation anticancer therapies, but corneal toxicity (punctate keratitis, corneal stromal opacity) is recognized as an adverse event 10). Case accumulation through pharmacovigilance studies and establishment of corneal protection strategies are challenges.
Adaptive optics OCT and multispectral fundus imaging are being used to visualize early RPE structural changes that cannot be detected by conventional methods 11). In addition, development of automated screening using artificial intelligence (AI) analysis of OCT images is progressing, which is expected to reduce the risk of missed detection 11).
With the widespread use of ICIs and molecular targeted drugs, standardized collaboration protocols between ophthalmology and oncology are needed 8). Guidelines for ocular adverse event management algorithms based on CTCAE grading are being developed.
Omoti AE, Omoti CE. Ocular toxicity of systemic anticancer chemotherapy. Pharm Pract (Granada). 2006;4(2):55-59.
Francis JH, Habib LA, Abramson DH, et al. Clinical and morphologic characteristics of MEK inhibitor-associated retinopathy: differences from central serous chorioretinopathy. Ophthalmology. 2017;124(12):1788-1798.
Dalvin LA, Shields CL, Orloff M, et al. Checkpoint inhibitor immune therapy: systemic indications and ophthalmic side effects. Retina. 2018;38(6):1063-1078.
Joshi MM, Garretson BR. Paclitaxel maculopathy. Arch Ophthalmol. 2007;125(5):709-710.
Renouf DJ, Velazquez-Martin JP, Simpson R, et al. Ocular toxicity of targeted therapies. J Clin Oncol. 2012;30(26):3277-3286.
Choe CH, McArthur GA, Caro I, et al. Ocular toxicity in BRAF mutant cutaneous melanoma patients treated with vemurafenib. Am J Ophthalmol. 2014;158(4):831-837.
Camidge DR, Bang YJ, Kwak EL, et al. Activity and safety of crizotinib in patients with ALK-positive non-small-cell lung cancer: updated results from a phase 1 study. Lancet Oncol. 2012;13(10):1011-1019.
Sun MM, Levinson RD, Filipowicz A, et al. Uveitis in patients treated with CTLA-4 and PD-1 checkpoint blockade inhibition. Ocul Immunol Inflamm. 2020;28(7):1036-1040.
Marmor MF, Kellner U, Lai TY, et al. Recommendations on screening for chloroquine and hydroxychloroquine retinopathy (2016 revision). Ophthalmology. 2016;123(6):1386-1394.
Matsuoka H, Tanaka H, Nagai Y, et al. Corneal adverse events associated with trastuzumab deruxtecan: a pharmacovigilance study. Target Oncol. 2023;18(1):77-85.
Melles RB, Marmor MF. The risk of toxic retinopathy in patients on long-term hydroxychloroquine therapy. JAMA Ophthalmol. 2014;132(12):1453-1460.