Independent risk factors
Dupilumab dose: Higher doses increase risk
AD severity: Higher incidence in moderate-to-severe AD
Pre-treatment history of conjunctivitis: Pre-existing ocular surface disease strongly predicts DIOSD onset

Dupilumab (brand name: Dupixent®) is a human monoclonal IgG4 antibody that binds to the IL-4 receptor alpha subunit (IL-4Rα) and inhibits signaling of IL-4 and IL-13. It is approved for atopic dermatitis (AD), bronchial asthma, and chronic rhinosinusitis with nasal polyps.
In phase 3 clinical trials, the incidence of conjunctivitis in the dupilumab plus topical corticosteroid group was 14–19%, significantly higher than 8% in the placebo group1). Post-marketing reports indicate that the frequency of DIOSD (dupilumab-induced ocular surface disease) may reach up to 50%1). 80% of ocular complications develop within 4 months of starting treatment, but late-onset cases after 52 weeks have also been reported1).
DIOSD is specific to dupilumab treatment for AD and has not been reported with its use for asthma or sinusitis. This selectivity suggests that the ocular surface of AD patients is already compromised1).
Currently, DIOSD is considered specific to dupilumab treatment for atopic dermatitis (AD)1). No increased frequency of conjunctivitis has been reported with its use for asthma or chronic rhinosinusitis with nasal polyps. It is speculated that the ocular surface of AD patients, already damaged by allergic inflammation, is more vulnerable to changes in cytokine balance induced by dupilumab.
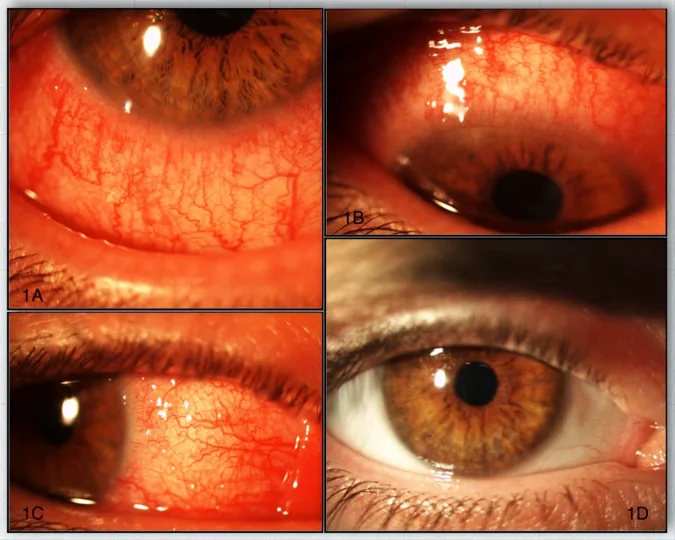
Symptoms include hyperemia, itching, irritation, foreign body sensation, photophobia, tearing, discharge, and decreased vision. Burning sensation and persistent “sandiness” are also characteristic 1). Intermittent monocular diplopia has also been reported.
With dupilumab monotherapy, onset occurs on average about 2 weeks after treatment. With concomitant topical steroid use, onset is slightly delayed to 4–8 weeks.
DIOSD progresses through the following three stages. It is usually bilateral but asymmetric.
| Stage | Findings |
|---|---|
| Early | MGD, conjunctival hyperemia, papillary conjunctivitis, punctate superficial keratopathy |
| Intermediate | Symblepharon, madarosis, fornix shortening, corneal limbitis |
| Advanced | Ankyloblepharon, fornix obliteration, ocular surface keratinization |
The clinical presentation of DIOSD is classified into the following five types1).
1. Dupilumab-associated conjunctivitis (DAC): Most frequent (36–50%), with nonspecific, papillary, follicular, and keratoconjunctivitis subtypes1). May present with limbal nodules (Trantas-like spots).
2. Corneal disorders: Manifest as superficial punctate keratitis, corneal erosion, or corneal ulcer1). Often accompanied by shortened tear break-up time.
3. Fibrotic complications: Cicatricial conjunctivitis, cicatricial ectropion, and punctal stenosis have been reported1).
4. Dry eye: Caused by goblet cell loss and lacrimal gland dysfunction1). Leads to tear film instability and mucosal inflammation.
5. Blepharitis and blepharoconjunctivitis: Present with eyelid swelling, redness, and periorbital dermatitis1). Cicatricial blepharoconjunctivitis is a severe complication.
Independent risk factors
Dupilumab dose: Higher doses increase risk
AD severity: Higher incidence in moderate-to-severe AD
Pre-treatment history of conjunctivitis: Pre-existing ocular surface disease strongly predicts DIOSD onset
Pathophysiology
IL-13 inhibition: IL-13 is the most dominant cytokine on the ocular surface and is essential for goblet cell differentiation and mucin secretion1). Dupilumab-induced IL-13 inhibition leads to goblet cell hypoplasia and reduced mucin secretion.
Th1/Th2 imbalance: IL-4/IL-13 inhibition induces a Th1-dominant immune response (excess IFN-γ), further reducing goblet cell density1).
Biopsy findings: Conjunctival biopsy shows a marked decrease in goblet cell density (median 3.3 cells/mm, control 32.3 cells/mm) and intraepithelial infiltration of CD3+/CD4+ T cells and eosinophils1)
The diagnosis of DIOSD is a clinical diagnosis based on medical history and clinical findings. Laboratory tests and biopsies are not usually indicated.
Required history: The patient is currently using dupilumab for moderate to severe AD. The onset of symptoms occurs after starting dupilumab (usually 2 to 8 weeks).
The following tests evaluate the stage and prognosis.
Corneoconjunctival staining: Assess conjunctival integrity with fluorescein and lissamine green.
Schirmer test: Tear volume less than 10 mm suggests lacrimal gland dysfunction in advanced cases.
Tear break-up time (TBUT): Shortening reflects mucin deficiency.
Corneal sensitivity test: Assess with a cotton swab or Cochet-Bonnet esthesiometer.
The most important distinction is from atopic keratoconjunctivitis (AKC) that existed before starting dupilumab. In AKC, goblet cell density is increased, whereas in DIOSD it is decreased1). Allergic conjunctivitis, viral conjunctivitis, ocular pemphigoid, and ocular GVHD are also considered in the differential diagnosis.
The goal of DIOSD treatment is to control inflammation and restore goblet cell function. Discontinuation of dupilumab is usually unnecessary; it can be continued while receiving ophthalmic treatment 1).
First-Line
Low-potency steroid eye drops: Fluorometholone 0.1% resulted in complete resolution in 2 of 5 cases and marked improvement in 3 cases 1). Monitor for increased intraocular pressure.
Tacrolimus ophthalmic ointment: Tacrolimus 0.03% ophthalmic ointment led to complete resolution in 2 of 4 cases and marked improvement in 2 cases 1). Calcineurin inhibitors may increase goblet cells and prevent epithelial cell death.
Cyclosporine eye drops: Cyclosporine A 0.05% ophthalmic solution is used as maintenance therapy. Combined with prednisolone acetate 1%, it has been reported to manage cases without recurrence.
Adjunctive Therapy
Artificial tears: Used to maintain ocular surface moisture. Often insufficient alone.
Warm compresses and eyelid massage: Effective in cases with MGD 1).
Antihistamine eye drops: Poor efficacy when used alone 1).
Patch testing: Performed when contact allergy is suspected. Avoidance of emulsifiers, surfactants, and fragrances has been reported to improve DIOSD 1).
In most cases, discontinuation is unnecessary 1). Almost all cases of DIOSD improve with ophthalmic treatment (steroid eye drops or calcineurin inhibitors). Because dupilumab is highly effective for AD and asthma, it is common to continue it while receiving ophthalmic treatment, considering the risk of worsening systemic disease upon discontinuation. However, in cases of severe cicatricial complications (cicatricial conjunctivitis, corneal ulcer) or treatment resistance, discontinuation should be considered by a multidisciplinary team.
The mechanism of DIOSD is complex, and the following five hypotheses have been proposed1).
Dupilumab inhibits IL-4/IL-13, suppressing Th2 responses while Th1 cytokines (IFN-γ) become predominant1). IFN-γ reduces goblet cell density and decreases mucin secretion.
Inhibition of IL-4/IL-13 may lead to Demodex proliferation, inducing IL-17-mediated rosacea-like inflammation1).
Abnormal OX40L-mediated signaling may be activated, leading to a vernal keratoconjunctivitis-like condition1).
Peripheral eosinophil counts may increase during dupilumab treatment, and eosinophil infiltration into the ocular surface may contribute to inflammation1).
This is the most plausible hypothesis1). IL-13 is the most predominant cytokine on the ocular surface and is essential for goblet cell differentiation and mucin secretion1). IL-13 inhibition by dupilumab causes goblet cell hypoplasia, leading to a cascade of reduced mucin secretion, tear film instability, and mucosal inflammation1). This condition is called DAMD (dupilumab-associated mucin deficiency)1).
In conjunctival biopsies by Bakker et al., DIOSD patients showed marked reduction of goblet cells (3.3 cells/mm vs. 32.3 cells/mm in controls) and infiltration of eosinophils and T cells1). This contrasts with allergic conjunctivitis and AKC, where goblet cells are increased (high IL-13 expression), supporting that DIOSD is an independent disease entity1).
In allergic conjunctivitis and atopic keratoconjunctivitis (AKC), IL-13 is highly expressed, and goblet cell density is increased with enhanced mucin secretion. In contrast, in DIOSD, dupilumab inhibits IL-13, leading to decreased goblet cell density1). Biopsy findings show goblet cell density reduced to about one-tenth of controls1). Thus, although both are inflammatory ocular surface diseases, the changes in goblet cells are opposite, indicating fundamentally different pathogenic mechanisms.
DIOSD and DAMD are relatively new disease concepts, and awareness has rapidly expanded after the marketing of dupilumab 1). Confirmation of goblet cell reduction via conjunctival biopsy has greatly advanced the understanding of this condition 1).
Contact dermatitis has been suggested as a possible modifying factor for DIOSD. There are reports that avoidance of allergens identified by patch testing (emulsifiers, surfactants, fragrances) improved DIOSD in some patients 1).
Elucidation of the mechanism by which calcineurin inhibitors restore goblet cell density, prediction of DIOSD onset using biomarkers, and accumulation of long-term prognosis data are needed. It has also been pointed out that abnormalities in retinoic acid metabolism may be involved in DIOSD 1), and the role of vitamin A supplementation may be examined in the future.
- Reji MA, Haque A, Goyal S, Krishnaswamy G. Dupilumab-induced ocular surface disease: a primer. BMJ Case Rep. 2022;15:e249019.