Penetrating Keratoplasty (PKP)
Rejection rate: Approximately 4.9–28.9% 7)
Main cause of failure: Rejection (early) + endothelial failure (late) 3)
Characteristics: Highest antigenicity due to inclusion of full-thickness donor tissue

Corneal transplantation is one of the most successful organ transplants. For first-time penetrating keratoplasty (PKP) in low-risk eyes, the 5-year survival rate reaches 95%. This high success rate is supported by the immune privilege of the cornea.
Factors contributing to corneal immune privilege include:
However, in high-risk eyes with corneal neovascularization, the failure rate at 3 years can exceed 35%. Even under immune privilege, the most common cause of graft failure is irreversible immunological rejection 1).
“Graft rejection” refers to the host’s specific immune response against the donor cornea. Primary graft failure is a condition in which the graft does not become clear within 8 weeks after surgery due to defects in the donor tissue itself, surgical trauma, or improper preservation, and is not immune-mediated 3). It occurs in about 0.1% of PKP cases.
The diagnosis of rejection is made only for grafts that have maintained clarity for at least 2 weeks after surgery. More than half of the cases occur within the first year after surgery, with a peak between 6 months and 1 year. However, it can also occur more than 20 years after surgery. The rate of progression from rejection to failure is reported to be approximately 49%.
Corneal transplantation is the most frequently performed tissue transplant worldwide. According to an international survey in 2012, PKP accounted for about 70% of all corneal transplants 1). In recent years, DSAEK and DMEK for endothelial diseases have rapidly become widespread, and DALK for keratoconus and post-keratitis scars has become a standard option, significantly changing the composition of surgical procedures. However, PKP remains essential for extensive corneal opacities and deformities and carries the highest risk of rejection 1).
Penetrating Keratoplasty (PKP)
Rejection rate: Approximately 4.9–28.9% 7)
Main cause of failure: Rejection (early) + endothelial failure (late) 3)
Characteristics: Highest antigenicity due to inclusion of full-thickness donor tissue
Deep Anterior Lamellar Keratoplasty (DALK)
Rejection rate: 1–24% 4)
Advantage: Eliminates the risk of endothelial rejection
Challenge: Stromal rejection can still occur 4)
Descemet's Stripping Automated Endothelial Keratoplasty (DSAEK)
Rejection rate: Average 10% (range 0–45%)
Primary failure rate: Average 5% (range 0–29%)
Features: Some reports indicate no significant difference compared to PKP3)
Descemet Membrane Endothelial Keratoplasty (DMEK)
Rejection rate: Average 1.9% (range 0–5.9%)7)
Primary failure rate: 1.7%
Features: Lowest antigenicity and low rejection rate3)
Large cohort studies have shown that DMEK has a significantly lower risk of rejection compared to PKP and DSAEK3). A meta-analysis comparing UT-DSAEK and DMEK found no significant difference in rejection risk at 12 months postoperatively2). In a Dutch multicenter randomized controlled trial of 54 eyes, the DMEK group had a significantly higher rate of achieving 20/25 or better vision at 12 months compared to the DSAEK group (66% vs 33%, P=0.02), while there were no significant differences in endothelial cell density or refractive changes11).
More than half of cases occur within the first year after surgery, especially between 6 months and 1 year. However, rejection can also occur long after surgery, so if symptoms such as redness, blurred vision, or decreased vision appear even years later, prompt medical attention is necessary. There have been reports of rejection triggered by vaccination more than 20 years after surgery10).
The rejection rate for PKP is approximately 4.9–28.9%, whereas for DMEK it is significantly lower at an average of 1.9% (range 0–5.9%). This difference is mainly due to the amount of donor tissue transplanted. PKP transplants epithelium and stroma containing dendritic cells, resulting in higher antigenicity. In contrast, DMEK transplants only Descemet’s membrane and endothelium, which has lower antigenicity and does not require sutures, reducing the risk. However, reports indicate that about 6% of DMEK cases experience rejection after discontinuation of steroids, highlighting the importance of long-term steroid use.
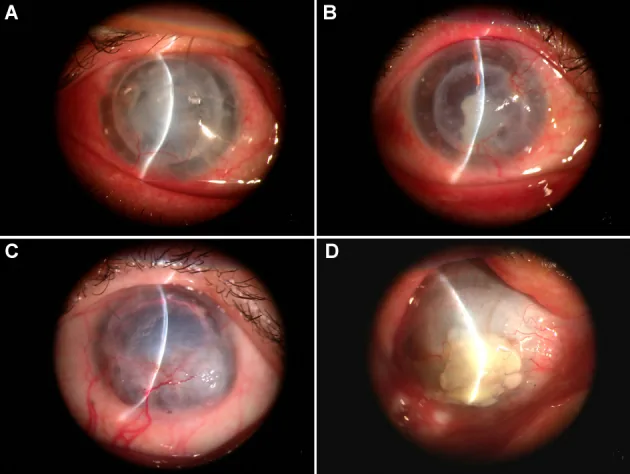
Patients present with blurred vision, conjunctival injection, eye pain, foreign body sensation, and decreased visual acuity. There are peaks of onset at 3 months and 1 year after surgery. If subjective symptoms appear, prompt medical attention is necessary.
The diagnostic criteria for rejection include any of the following: conjunctival injection, photophobia, decreased visual acuity, anterior chamber cells, keratic precipitates (KP), endothelial or epithelial rejection lines, subepithelial infiltrates, and localized graft edema1).
Graft-localized KP are characteristic, and KP on the recipient cornea are not observed. The Khodadoust line is a linear deposit on the corneal endothelium and represents the advancing front of endothelial rejection.
Rejection is classified into three types based on the layer affected: epithelial, stromal, and endothelial. Endothelial rejection has the greatest impact on graft prognosis, and delay in treatment leads to irreversible endothelial failure and vision loss.
| Rejection Type | Frequency | Main Findings |
|---|---|---|
| Epithelial | Approximately 2% | Linear ridge migrating from the limbus (epithelial rejection line) |
| Stromal | — | Stromal edema is the only finding |
| Endothelial type | Approximately 50% | KP, Khodadoust line, edema |
Epithelial rejection has a low frequency of about 2% of all rejection episodes. As a precursor lesion, round subepithelial infiltrates of 0.2–0.5 mm are observed just below Bowman’s membrane. As it progresses, an edematous raised linear lesion (epithelial rejection line) forms. It has little effect on the clear healing of the graft itself, but can trigger endothelial rejection.
Stromal rejection has stromal edema as the only finding. In DALK, stromal infiltration and interface neovascularization may be seen as stromal immune rejection 4). It is difficult to differentiate from corneal edema due to endothelial rejection in PKP eyes.
Endothelial rejection accounts for about 50% of all rejection episodes and is clinically the most important. Localized corneal endothelial deposits within the graft are a key finding, and when a Khodadoust line forms, it is accompanied by stromal edema at the same site. Mixed type (epithelial + endothelial, etc.) is seen in about 30%.
Khodadoust line is a linear corneal endothelial deposit characteristic of endothelial rejection. It represents the advancing front of rejection that progressively moves across the graft endothelium; in the area where the line has passed, endothelial cells are damaged, causing stromal edema. If Khodadoust line is observed, prompt and intensive steroid treatment is necessary.
| Finding | Rejection | HSV/VZV endotheliitis | CMV endotheliitis |
|---|---|---|---|
| Distribution of KP | Localized within the graft | Also attached outside the graft | Also attached outside the graft |
| Color of KP | White to gray-white | Brown | Brown to white |
| Characteristic findings | Khodadoust line | Arlt’s triangle | Coin lesion |
Rejection is characterized by KP localized to the graft, whereas viral endotheliitis is differentiated by the presence of KP also outside the graft. Since corneal endothelial deposits may sometimes be donor-derived at the time of corneal transplantation, recording the distribution of KP during daily examinations is useful for differentiation.
Cases in which PKP is prone to rejection are called high-risk eyes, and the following factors are listed.
In DMEK, rejection may be triggered during steroid tapering. In one case, rejection occurred 15 months after DMEK when switching from betamethasone to fluorometholone7). Peripheral anterior synechiae (PAS) have also been reported as a risk factor for DMEK rejection7).
While the benefit of HLA matching is clear in organ transplantation, results are inconsistent in corneal transplantation1). The Corneal Transplant Follow-up Study II (CTFS II) conducted in the UK was a large prospective clinical trial examining the impact of HLA class II (HLA-DR) matching in high-risk PKP1). From 1998 to 2011, 1133 transplants were collected and stratified into 0, 1, or 2 HLA-DR mismatches under the condition of ≤2 HLA class I mismatches1). DNA-based methods (PCR-SSP/PCR-SSO) were used for donor and recipient tissue typing to avoid errors from serological techniques1).
In CTFS II, no clear relationship was found between the number of HLA-DR mismatches and the incidence of rejection1). As shown in rodent models, corneal graft rejection involves multiple distinct immune pathways, and this redundancy of immune responses is thought to partly explain the inconsistent results of HLA matching studies1).
Multiple cases of corneal transplant rejection after COVID-19 vaccination have been reported. It can occur with mRNA vaccines (BNT162b2), viral vector vaccines (ChAdOx1), and inactivated vaccines (Sinopharm).
Two cases of acute PKP rejection were reported 2 weeks after the first dose of BNT162b2 vaccine6). Neither had a history of previous rejection, and both responded well to topical and systemic steroids6).
A case of endothelial rejection after femtosecond laser corneal transplantation occurred 2 weeks after ChAdOx1 vaccination8). Khodadoust line and anterior chamber inflammation were observed, and recovery occurred after 5 weeks of steroid treatment8).
Two cases of rejection after Sinopharm inactivated vaccine have also been reported9). A literature review has accumulated at least 20 cases of vaccine-related rejection, with the majority recovering with steroid treatment9).
There is a case of rejection occurring 10 days after BNT162b2 vaccination in a PKP that had been stable for over 20 years post-surgery10).
Although causality is not established, a hypothesis has been proposed that vaccination may trigger rejection through induction of MHC class II antigen-presenting cells9).
Corneal transplant rejection after COVID-19 vaccination has been reported with mRNA, vector, and inactivated vaccines. It often occurs 1 to 3 weeks after vaccination, and most cases respond to steroid treatment. Meta-analyses have ruled out vaccine-induced rejection in solid organ transplants, but case accumulation is ongoing for corneal transplants. Since causality is not established, it is recommended that corneal transplant patients increase steroid use before vaccination and seek early medical attention after vaccination.
Check for graft-localized KP, Khodadoust line, corneal edema, and anterior chamber cells. Epithelial rejection lines start near congested limbal vessels and move across the graft border.
The main conditions requiring differentiation from endothelial rejection are as follows.
In rejection, KP is confined to the graft, and intraocular pressure elevation is rare. In HSV/VZV endotheliitis, KP also attaches outside the graft and is accompanied by acute intraocular pressure elevation. In CMV endotheliitis, coin lesion-like KP and chronic persistent intraocular pressure elevation are characteristic. If confirmation is difficult, anterior chamber PCR, serum antibody tests, and response to steroids are comprehensively evaluated.
The basic treatment for rejection is anti-inflammatory therapy with steroids.
Mild (epithelial/stromal type)
Severe (endothelial type, Khodadoust line positive)
With early treatment, more than 50% of acute rejection episodes recover; however, delayed treatment can lead to irreversible endothelial cell loss and graft failure. Patient education is important: patients should be aware of postoperative symptoms (redness, blurred vision, eye pain, photophobia) and seek early medical attention if any changes occur.
For prevention of rejection after corneal transplantation, a two-step protocol based on risk stratification is used.
Postoperative Management for Normal-Risk Eyes
Steroid eye drops: Rinderon eye drops 0.01% (betamethasone) 5 times/day → switch to Flumetholon eye drops 0.1% (fluorometholone) 2-3 times/day (taper over 6 months)
Systemic administration: Prednisone 20 mg for a few days, or no administration
CsA: Combined eye drops, continued for several months
Postoperative management of high-risk eyes
Steroid eye drops: Betamethasone 0.1% × 4 times/day → continued for 1 year or more
Systemic steroids: Rinderon injection 0.4% 2 mg once daily IV drip for 3 days from surgery day, then Rinderon tablets 0.5 mg 2 tablets divided in 1, tapered over 2 weeks
CsA: Neoral capsules 25 mg 3 mg/kg/day, trough level 70-100 ng/mL
Cyclosporine A (CsA) is used in high-risk cases such as corneal stromal vascular invasion involving 2 or more quadrants, regrafts, and history of rejection. During systemic administration, maintain trough level at 70-100 ng/mL and monitor systemic side effects including renal function. Continue for about 6 months postoperatively.
In an 18-year-old PKP case with bilateral simultaneous rejection, after remission with IV methylprednisolone pulse, due to steroid response, switched to CsA 1% eye drops and maintained successfully5). CsA 1% eye drops allow early tapering of potent steroids in steroid responders and are useful for long-term graft maintenance5).
Tacrolimus is used as a switch drug for cases that develop rejection while on oral cyclosporine. Target trough level is 8-10 ng/mL until 2 months postoperatively, then 5-6 ng/mL thereafter (Prograf 0.05-0.1 mg/kg/day). There are reports that 0.03% tacrolimus eye drops are effective in preventing rejection in high-risk corneal transplantation.
Loose or broken sutures can trigger both rejection and late infection, so remove them promptly upon discovery. After suture removal, temporarily intensify steroid and antibiotic eye drop treatment. Even after remission, long-term continuation of steroid eye drops is expected to suppress recurrence3).
When starting with betamethasone 0.1%, first continue a maintenance phase of 4 times/day for several months to 1 year, then switch to a low-concentration steroid such as fluorometholone 0.1% and maintain long-term at 1-2 times/day. If intraocular pressure rises, consider switching to loteprednol or adding glaucoma eye drops.
If intraocular pressure rises due to steroid response, consider switching from prednisolone acetate to loteprednol, which has less effect on intraocular pressure, or combining with CsA 1% eye drops. CsA 1% eye drops allow early tapering of steroids and are useful for balancing intraocular pressure management and rejection suppression. Concomitant use of glaucoma eye drops is also performed as needed.
The cornea maintains physiological immune tolerance through anterior chamber-associated immune deviation (ACAID). In ACAID, antigen-presenting cells become tolerogenic in a TGF-β-dominant environment, suppressing delayed-type hypersensitivity and complement-fixing antibody production against donor antigens. However, when high-risk factors such as neovascularization, inflammation, or loose sutures are present, this immune privilege is easily disrupted.
The central mechanism of rejection is delayed-type hypersensitivity, and the main effector cells are CD4+ Th1 cells. Activated Th1 cells produce IFN-γ, inducing MHC class II antigen-presenting cells throughout the transplanted cornea, leading to cell-mediated immune response 8). The involvement of antibody-mediated mechanisms has also gained attention in recent years, suggesting that anti-HLA antibodies may cause chronic endothelial cell damage via complement activation 1).
Differences in rejection rates among surgical techniques are mainly due to the amount and antigenicity of donor tissue transplanted 3).
Peripheral anterior synechiae (PAS) have been noted as a risk factor for rejection after DMEK. In a mouse corneal transplant model, the group with PAS showed significantly increased rejection 7). Direct contact between the iris and donor endothelium due to PAS is thought to induce cytotoxic T lymphocyte activity and promote rejection 7).
Corneal transplant rejection is primarily mediated by cellular immunity, but rodent studies have identified multiple distinct immune pathways leading to rejection 1). This redundancy in immune response is considered one reason for inconsistent results in HLA matching studies 1). Recently, the role of anti-HLA antibodies and antibody-mediated rejection has also gained attention, potentially elucidating mechanisms of late endothelial failure 1).
COVID-19 vaccination elicits a systemic immune response, inducing SARS-CoV-2 neutralizing antibodies as well as antigen-specific CD8+ and Th1-type CD4+ T cell responses 6). This immune activation may trigger rejection of the transplanted cornea through cross-reactivity or non-specific immune activation 6). For inactivated vaccines (Sinopharm), the immunogenicity of the adjuvant aluminum hydroxide has been suggested as a possible main cause of rejection 9). However, at the meta-analysis level, no increase in rejection after COVID-19 vaccination in solid organ transplantation has been confirmed, and a causal relationship in corneal transplantation has not been established at this time.
Case reports of corneal transplant rejection associated with COVID-19 vaccines are accumulating worldwide 9). A literature review has collected over 20 cases, revealing that the majority are regrafts, onset occurs 1–2 weeks after vaccination, and most recover with steroid treatment 9). Prophylactic steroid administration before vaccination has been proposed, but no randomized controlled trials exist, and decisions must be made on a case-by-case basis 8)9).
Regarding the effectiveness of HLA matching, the CTFS II trial prospectively investigated this, but at present, no clear clinical benefit of HLA-DR matching in corneal transplantation has been demonstrated 1). However, the role of anti-HLA antibodies and antibody-mediated rejection is becoming clearer, which may contribute to elucidating the mechanism of late endothelial failure 1).
In comparisons between UT-DSAEK and DMEK, both the Sela 2023 meta-analysis 2) and the Dunker 2020 multicenter RCT 11) showed no significant difference in 12-month rejection rates, while the DMEK group achieved better corrected visual acuity. However, graft failure tended to be slightly higher in the DMEK group 2), and the choice of surgical technique should be based on a comprehensive assessment of the patient’s individual ocular pathology, history, and institutional experience.
The following directions are attracting attention for the future.