Diabetic Eye Screening Guidelines
Key points at a glance
Section titled “Key points at a glance”1. What is Ophthalmic Screening for Diabetic Patients?
Section titled “1. What is Ophthalmic Screening for Diabetic Patients?”Ophthalmic screening for diabetic patients is a regular eye examination system to detect ocular complications such as diabetic retinopathy, diabetic macular edema, neovascular glaucoma, cataract, and extraocular muscle palsy early, and to provide appropriate treatment intervention.
Diabetic retinopathy is the second leading cause of acquired blindness in adults. Approximately 30–40% of diabetic patients worldwide have retinopathy, and about 10% have vision-threatening retinopathy 1). It has also been reported that about 20% of patients already have retinopathy at the time of diabetes diagnosis 1), highlighting the importance of early initiation of screening.
On the other hand, more than 90% of blindness can be prevented with appropriate screening and treatment intervention 2). Retinopathy is asymptomatic in its early stages, and by the time symptoms appear, the disease is often quite advanced. Therefore, regular eye examinations are the only means of early detection.
The number of diabetic patients in Japan is estimated to exceed 10 million, and establishing a system for continuous regular ophthalmology visits is a challenge. Collaboration tools between internal medicine and ophthalmology, such as the diabetes eye notebook, help share fundus findings, examination dates, and treatment details 3).
For type 2 diabetes, it is recommended to undergo an eye examination immediately at the time of diagnosis. It has been reported that about 20% of patients already have retinopathy at diagnosis 1), and retinopathy may be progressing even without symptoms. For type 1 diabetes, eye examinations should start 5 years after onset or from adolescence 4). In both types, regular fundus examinations are essential even before symptoms such as vision loss appear.
2. Screening Targets and Recommended Frequency
Section titled “2. Screening Targets and Recommended Frequency”Target Population and Timing of Initiation
Section titled “Target Population and Timing of Initiation”The timing of screening initiation varies depending on the type and condition of diabetes.
- Type 2 diabetes: Initial eye examination at the time of diagnosis4)
- Type 1 diabetes: Start approximately 5 years after onset (or at puberty), then continue annual fundus examinations4)
- Gestational diabetes mellitus (GDM): Fundus examination in the first trimester, with follow-up until 1 year postpartum5)
- Pregnant women with pre-existing diabetes: Examination before pregnancy or in the first trimester, with follow-up each trimester5)
Recommended Screening Frequency (by Disease Stage)
Section titled “Recommended Screening Frequency (by Disease Stage)”Screening frequency is determined by combining the stage of retinopathy and HbA1c control status.
| Stage of Retinopathy | Recommended Screening Frequency |
|---|---|
| No retinopathy (HbA1c < 7.0% with good control) | Every 1–2 years4) |
| Mild nonproliferative stage | Every 6–12 months4) |
| Moderate nonproliferative | Every 3–6 months4) |
| Severe nonproliferative (preproliferative) | Every 1–3 months4) |
| Proliferative diabetic retinopathy | Every 1 month (concurrent with treatment)4) |
HbA1c Control and Retinopathy Progression
Section titled “HbA1c Control and Retinopathy Progression”HbA1c control is also an important factor in determining screening frequency.
The UKPDS (United Kingdom Prospective Diabetes Study) showed that a 1% reduction in HbA1c reduces the risk of microvascular complications by 37%6). Maintaining HbA1c below 7.0% leads to a significant reduction in the risk of retinopathy progression6).
On the other hand, rapid improvement in blood glucose, such as with insulin initiation, can temporarily worsen retinopathy, a phenomenon known as “early worsening”7). This was observed in the DCCT (Diabetes Control and Complications Trial), and attention should be paid to short-term retinal deterioration after starting insulin7).
If there is no retinopathy and HbA1c is well controlled (below 7.0%), an exam every 1–2 years is acceptable. For mild nonproliferative retinopathy, every 6–12 months; moderate, every 3–6 months; severe nonproliferative, every 1–3 months; and proliferative, monthly exams and treatment are needed4). If HbA1c is high or blood glucose control is unstable, more frequent follow-up is recommended.
3. Examination Methods
Section titled “3. Examination Methods”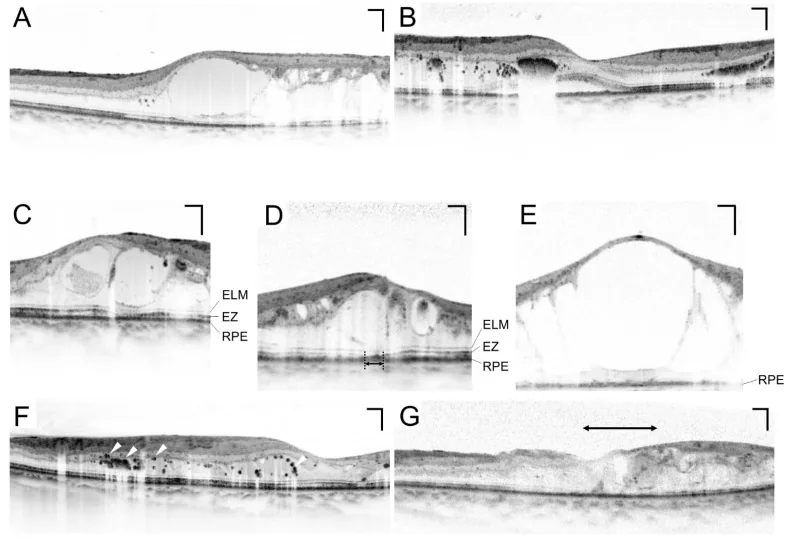
Dilated Fundus Examination
Section titled “Dilated Fundus Examination”Dilated fundus examination is the central test in diabetic eye screening. It is standard to perform fundus examination after pupil dilation with eye drops of tropicamide and phenylephrine (phenylephrine hydrochloride) 4). The 7-field stereoscopic photography of the Early Treatment Diabetic Retinopathy Study (ETDRS) is considered the reference standard 8).
Non-mydriatic fundus cameras are convenient and increasingly used in screening, with reported sensitivity of 80–90% 9). In the UK National Diabetic Retinopathy Screening Programme, a system centered on non-mydriatic digital fundus photography is in operation 9).
Optical Coherence Tomography (OCT)
Section titled “Optical Coherence Tomography (OCT)”OCT is essential for quantitative assessment of macular edema. It allows non-invasive and repeatable measurement of central retinal thickness (CRT) and is indispensable for monitoring treatment response 10). CRT > 300 μm is considered one of the thresholds for initiating treatment of diabetic macular edema (DME) 10).
Fluorescein Angiography (FA)
Section titled “Fluorescein Angiography (FA)”This test involves intravenous injection of sodium fluorescein to visualize retinal blood vessels, and is used to detect non-perfusion areas and evaluate neovascularization 8). It is used to determine the indication for photocoagulation and is useful for precise evaluation of ETDRS classification 8).
OCT Angiography (OCTA)
Section titled “OCT Angiography (OCTA)”OCTA is a non-invasive test that can three-dimensionally visualize retinal blood vessels without using contrast agents 11). It can be repeated as an alternative to fluorescein angiography and is applied to evaluate non-perfusion areas and the macular capillary network 11).
AI-Based Fundus Screening
Section titled “AI-Based Fundus Screening”Automated fundus analysis using deep learning is rapidly moving toward practical application. In a multi-ethnic cohort study by Ting et al. (2017), sensitivity of 87–97% was reported for automated detection of diabetic retinopathy, suspected glaucoma, and age-related macular degeneration 12). AI devices approved by the US FDA (e.g., IDx-DR) are already in clinical use, and their application in screening in internal medicine and primary care settings is advancing 12).
| Examination Method | Main Indication | Characteristics |
|---|---|---|
| Dilated fundus examination (7-field stereo) | Reference standard for staging | High accuracy; requires invasive dilation 8) |
| Non-mydriatic fundus camera | Primary screening | Sensitivity 80–90%, high convenience 9) |
| OCT | Quantification of macular edema and treatment monitoring | Non-invasive, CRT quantification 10) |
| Fluorescein angiography (FA) | Assessment of non-perfusion areas and neovascularization | Invasive, contrast agent use 8) |
| OCTA | Non-invasive vascular assessment | Repeatable, no contrast agent needed 11) |
| AI fundus screening | Automated primary screening | Sensitivity 87–97%, no ophthalmologist required 12) |
AI fundus screening using deep learning has been reported to have high accuracy with sensitivity of 87–97%, and is now in practical use 12). FDA-approved AI devices exist, and they are expected to be used in areas with a shortage of ophthalmologists. However, AI screening is only a primary screening; if an abnormality is detected, a detailed examination by an ophthalmologist is necessary.
4. Classification of Diabetic Retinopathy
Section titled “4. Classification of Diabetic Retinopathy”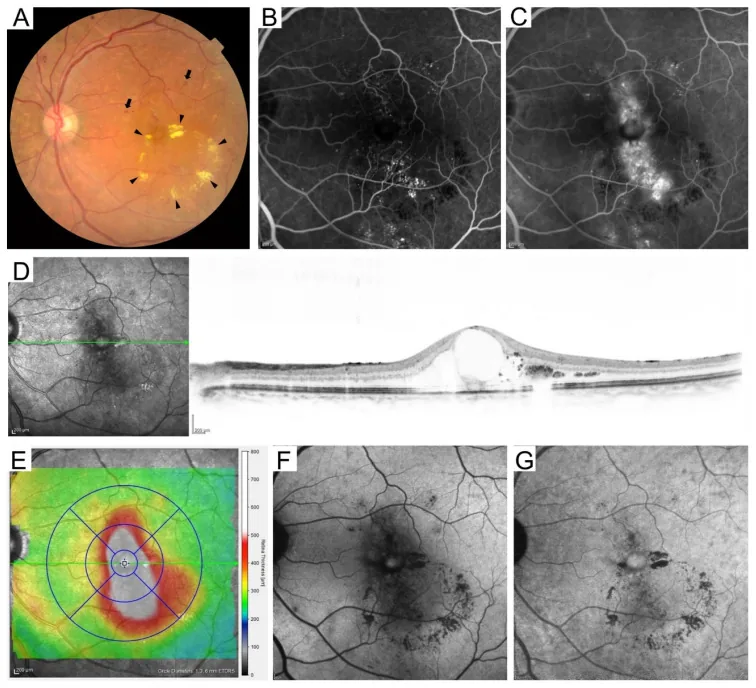
International Classification (Based on ETDRS)
Section titled “International Classification (Based on ETDRS)”The international classification of diabetic retinopathy consists of five stages based on the ETDRS criteria 8).
| Stage | Main Findings | Management |
|---|---|---|
| No retinopathy | No abnormalities | Screening every 1–2 years 4) |
| Mild nonproliferative (NPDR) | Microaneurysms only | Follow-up every 6–12 months 4) |
| Moderate NPDR | Soft exudates, retinal hemorrhages, hard exudates | Follow-up every 3–6 months4) |
| Severe NPDR (4-2-1 rule) | Retinal hemorrhages in 4 quadrants, venous beading in 2 quadrants, IRMA in 1 quadrant8) | Consider early PRP; follow-up every 1–3 months4) |
| Proliferative diabetic retinopathy (PDR) | NVD, NVE, vitreous hemorrhage, tractional retinal detachment8) | PRP, anti-VEGF, surgery. Monthly follow-up4) |
- The 4-2-1 rule in severe NPDR refers to a stage meeting any one of the following: retinal hemorrhages in 4 quadrants, venous beading in 2 quadrants, or IRMA (intraretinal microvascular abnormalities) in 1 quadrant8).
- In proliferative diabetic retinopathy (PDR), optic disc neovascularization (NVD) and retinal neovascularization (NVE) appear, progressing to vitreous hemorrhage and tractional retinal detachment8).
Diabetic Macular Edema (DME)
Section titled “Diabetic Macular Edema (DME)”Diabetic macular edema (DME) is a condition that can occur at any stage of retinopathy and is the most common cause of vision loss in diabetes10).
- CSME (clinically significant macular edema) involving the fovea is an indication for treatment10)
- On OCT, CRT > 300 μm is considered one of the criteria for initiating treatment10)
- The European Society of Retina Specialists (EURETINA) guidelines recommend anti-VEGF therapy as the first-line treatment 10)
Other Ocular Complications
Section titled “Other Ocular Complications”- Neovascular glaucoma: As a complication of PDR, new blood vessels form in the iris and angle, leading to secondary glaucoma
- Diabetic cataract: There are two forms: true diabetic cataract (rapidly progressive in young patients) and acceleration of age-related cataract
- Extraocular muscle palsy: Diplopia due to third or sixth cranial nerve palsy may occur acutely
5. Treatment Strategy (From Screening to Treatment Collaboration)
Section titled “5. Treatment Strategy (From Screening to Treatment Collaboration)”Basics of Medical Management
Section titled “Basics of Medical Management”Medical management of blood glucose, blood pressure, and lipids is the foundation for suppressing the progression of diabetic retinopathy. Target values are as follows 6).
- HbA1c: <7.0%
- Blood pressure: <130/80 mmHg
- LDL-C: <120 mg/dL
In the UKPDS, intensive glucose control reduced the risk of microvascular complications by 37% compared to conventional management, and a 1% reduction in HbA1c was associated with a significant decrease in complication risk 6).
Laser Photocoagulation (Panretinal Photocoagulation: PRP)
Section titled “Laser Photocoagulation (Panretinal Photocoagulation: PRP)”Panretinal photocoagulation (PRP) is performed for severe NPDR to proliferative diabetic retinopathy 8). By ablating ischemic retina, VEGF production is suppressed, leading to regression and prevention of new blood vessels. This treatment was established by the ETDRS and is the standard of care for preventing blindness in high-risk PDR 8).
Anti-VEGF Therapy
Section titled “Anti-VEGF Therapy”The first-line treatment for diabetic macular edema (DME) is intravitreal injection of anti-VEGF agents. The available drugs are as follows 10).
- Ranibizumab (Lucentis): 0.5 mg/0.05 mL intravitreal injection
- Aflibercept (Eylea): 2 mg/0.05 mL intravitreal injection
- Faricimab (Vabysmo): 6 mg/0.05 mL intravitreal injection (Ang-2/VEGF-A bispecific antibody)
Vitrectomy
Section titled “Vitrectomy”Vitrectomy is performed for non-absorbing vitreous hemorrhage and tractional retinal detachment 8). In recent years, small-incision vitrectomy (25-27 gauge) has become widespread, reducing surgical invasiveness.
Local Steroid Administration
Section titled “Local Steroid Administration”Intravitreal injection of triamcinolone acetonide (TA) 4 mg/0.1 mL is used as adjunctive treatment for DME 10). It is considered for cases with insufficient response to anti-VEGF therapy or in pseudophakic eyes (after cataract surgery).
Fenofibrate
Section titled “Fenofibrate”In the FIELD study (2007) and the ACCORD Eye study, the fenofibrate group showed suppression of diabetic retinopathy progression and a reduced need for photocoagulation 13). In addition to its lipid-lowering effect, activation of PPARα is thought to contribute to anti-inflammatory and anti-angiogenic effects 13).
Early detection can prevent more than 90% of blindness 2). Treatment varies depending on the stage and type of complications. For mild to moderate cases, medical management of blood glucose, blood pressure, and lipids is the mainstay. For severe nonproliferative to proliferative stages, laser photocoagulation (PRP) is performed, and for diabetic macular edema, anti-VEGF intravitreal injections (ranibizumab, aflibercept, faricimab) are first-line. Vitrectomy is indicated for non-absorbing vitreous hemorrhage or tractional retinal detachment.
6. Pathophysiology and Scientific Basis of Screening
Section titled “6. Pathophysiology and Scientific Basis of Screening”Retinal microvascular damage due to hyperglycemia
Section titled “Retinal microvascular damage due to hyperglycemia”The development of diabetic retinopathy involves multiple metabolic pathways triggered by hyperglycemia 14).
- Enhanced polyol pathway: Increased conversion of glucose to sorbitol by aldose reductase leads to elevated intracellular osmolarity and oxidative stress.
- Accumulation of AGEs (advanced glycation end products): Accumulation in the vascular basement membrane and extracellular matrix impairs vascular function.
- Activation of PKC (protein kinase C): Promotes VEGF production, contributing to increased vascular permeability and neovascularization.
- Increased oxidative stress: Overproduction of reactive oxygen species (ROS) damages endothelial cell function.
From pericyte loss to neovascularization
Section titled “From pericyte loss to neovascularization”Selective loss of pericytes (support cells of the vessel wall) in retinal capillaries is the earliest change in diabetic retinopathy 14). Pericyte loss weakens capillary walls, leading to microaneurysm formation. Increased vascular permeability causes macular edema, and capillary non-perfusion (non-perfused areas) leads to retinal ischemia 14).
Increased VEGF production from the ischemic retina is the main driver of neovascularization, forming NVD and NVE. Intravitreal VEGF also contributes to the pathology of vitreous hemorrhage and tractional retinal detachment 14).
Cost-effectiveness of screening
Section titled “Cost-effectiveness of screening”The cost-effectiveness of diabetic retinopathy screening programs is supported by multiple economic analyses. A systematic review by Jones et al. (2010) showed that early treatment through screening is significantly less costly than the management costs after blindness, demonstrating high cost-effectiveness 15).
DCCT and UKPDS scientifically established the importance of glycemic control. In DCCT, intensive insulin therapy reduced the risk of new-onset retinopathy by 76% and progression by 54% in type 1 diabetes 6). UKPDS showed that intensive glycemic control reduced the risk of microvascular complications by 37% in type 2 diabetes 6).
7. Latest Research and Future Prospects
Section titled “7. Latest Research and Future Prospects”Social Implementation of AI-Based Automated Screening
Section titled “Social Implementation of AI-Based Automated Screening”The accuracy of automated fundus analysis using deep learning has reached a level comparable to that of ophthalmology specialists, and social implementation is progressing 12). Its use in non-invasive screening in internal medicine and primary care is expected to help identify high-risk patients with low ophthalmology consultation rates.
Introduction of Ultra-Widefield Fundus Cameras
Section titled “Introduction of Ultra-Widefield Fundus Cameras”Ultra-widefield fundus cameras such as Optos cover more than 200° of the retina in a single shot, and their application to non-mydriatic screening is advancing 9). Improved accuracy in detecting peripheral retinal non-perfusion areas and neovascularization has been reported, and it is expected to achieve both higher screening accuracy and convenience.
Use of Telemedicine (Teleophthalmology)
Section titled “Use of Telemedicine (Teleophthalmology)”Remote fundus image reading using telemedicine has accumulated experience, mainly in diabetic retinopathy screening 12). It contributes to improving screening rates for diabetic patients in areas with a shortage of ophthalmologists or in remote regions, and is attracting attention as a tool to reduce disparities.
Research on New Drugs and Retinopathy Risk
Section titled “Research on New Drugs and Retinopathy Risk”With the widespread use of GLP-1 receptor agonists (e.g., semaglutide) and SGLT2 inhibitors (e.g., empagliflozin), research on the impact of these drugs on diabetic retinopathy risk is progressing 16). Some trials of semaglutide have reported an increased risk of acute worsening of retinopathy, and its association with early worsening is particularly noted 16). Continued interest exists in elucidating the mechanism of fenofibrate’s inhibitory effect on DR progression and in further large-scale trials 13).
Tear Biomarkers and Retinal Vascular Parameters
Section titled “Tear Biomarkers and Retinal Vascular Parameters”Research is ongoing on non-invasive tear sampling for quantification of VEGF and inflammatory cytokines, as well as early retinopathy risk assessment using retinal vessel diameter and fractal analysis. In the future, it may be possible to quantify retinopathy risk using only fundus camera imaging.
8. References
Section titled “8. References”- Yau JW, Rogers SL, Kawasaki R, et al. Global prevalence and major risk factors of diabetic retinopathy. Diabetes Care. 2012;35(3):556-564.
- Flaxel CJ, Adelman RA, Bailey ST, et al. Diabetic retinopathy Preferred Practice Pattern®. Ophthalmology. 2020;127(1):P66-P145.
- 日本糖尿病眼学会. 糖尿病眼手帳について. https://www.jsod.jp/techo/index.html
- Solomon SD, Chew E, Duh EJ, et al. Diabetic retinopathy: a position statement by the American Diabetes Association. Diabetes Care. 2017;40(3):412-418.
- Morrison JL, Hodgson LA, Lim LL, et al. Diabetic retinopathy in pregnancy: a review. Clin Exp Ophthalmol. 2016;44(4):321-334.
- UK Prospective Diabetes Study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet. 1998;352(9131):837-853.
- The Diabetes Control and Complications Trial Research Group. Early worsening of diabetic retinopathy in the Diabetes Control and Complications Trial. Arch Ophthalmol. 1998;116(7):874-886.
- Early Treatment Diabetic Retinopathy Study Research Group. Grading diabetic retinopathy from stereoscopic color fundus photographs — an extension of the modified Airlie House classification. ETDRS report number 10. Ophthalmology. 1991;98(5 Suppl):786-806.
- Scanlon PH. The English National Screening Programme for diabetic retinopathy 2003-2016. Acta Diabetol. 2017;54(6):515-525.
- Schmidt-Erfurth U, Garcia-Arumi J, Bandello F, et al. Guidelines for the management of diabetic macular edema by the European Society of Retina Specialists (EURETINA). Ophthalmologica. 2017;237(4):185-222.
- Spaide RF, Fujimoto JG, Waheed NK, et al. Optical coherence tomography angiography. Prog Retin Eye Res. 2018;64:1-55.
- Ting DSW, Cheung CY, Lim G, et al. Development and validation of a deep learning system for diabetic retinopathy and related eye diseases using retinal images from multiethnic populations with diabetes. JAMA. 2017;318(22):2211-2223.
- Keech AC, Mitchell P, Summanen PA, et al. Effect of fenofibrate on the need for laser treatment for diabetic retinopathy (FIELD study): a randomised controlled trial. Lancet. 2007;370(9600):1687-1697. doi:10.1016/S0140-6736(07)61607-9.
- Antonetti DA, Klein R, Gardner TW. Diabetic retinopathy. N Engl J Med. 2012;366(13):1227-1239.
- Jones S, Edwards RT. Diabetic retinopathy screening: a systematic review of the economic evidence. Diabet Med. 2010;27(3):249-256.
- Ntentakis DP, Correa VSMC, Ntentaki AM, Delavogia E, Narimatsu T, Efstathiou N, Vavvas DG. Effects of newer-generation anti-diabetics on diabetic retinopathy: a critical review. Graefes Arch Clin Exp Ophthalmol. 2024;262(3):717-752. PMID:37728754. doi:10.1007/s00417-023-06236-5.
