Endogenous
Route of infection: Hematogenous dissemination from fungemia
Main causative organism: Candida albicans (most common)
Frequency: More than half of all endogenous endophthalmitis cases
Bilaterality: Seen in approximately 30% of cases

Fungal endophthalmitis is a severe eye disease in which fungi infect and proliferate in the intraocular fluids (vitreous humor and aqueous humor). It is classified into two types based on the route of infection: endogenous (hematogenous dissemination) and exogenous (spread from surgery, trauma, or keratitis).
More than half of endogenous endophthalmitis cases are fungal, and 30% are bilateral. Exogenous cases are subdivided into three categories: postoperative, post-traumatic, and keratitis-related 7). Compared to bacterial endophthalmitis, progression is slower, with symptoms worsening over days to weeks.
Endogenous
Route of infection: Hematogenous dissemination from fungemia
Main causative organism: Candida albicans (most common)
Frequency: More than half of all endogenous endophthalmitis cases
Bilaterality: Seen in approximately 30% of cases
Exogenous
Route of infection: Spread from surgery, trauma, or keratitis
Main causative organism: Aspergillus species (most common)
Frequency: Mostly unilateral
Classification: Three categories: postoperative, post-traumatic, and keratitis-related
The causative organisms are typically Candida and Aspergillus species, but reports of rare species are increasing. Trichosporon species (naturally resistant to echinocandins) 6), C. rugosa (less than 0.1% of non-albicans Candida) 3), Arthrographis kalrae 5), and Neoscytalidium dimidiatum 7) have been reported.
After the COVID-19 pandemic, cases of fungal endophthalmitis triggered by immunosuppression due to steroid use have been reported in succession 1, 9).
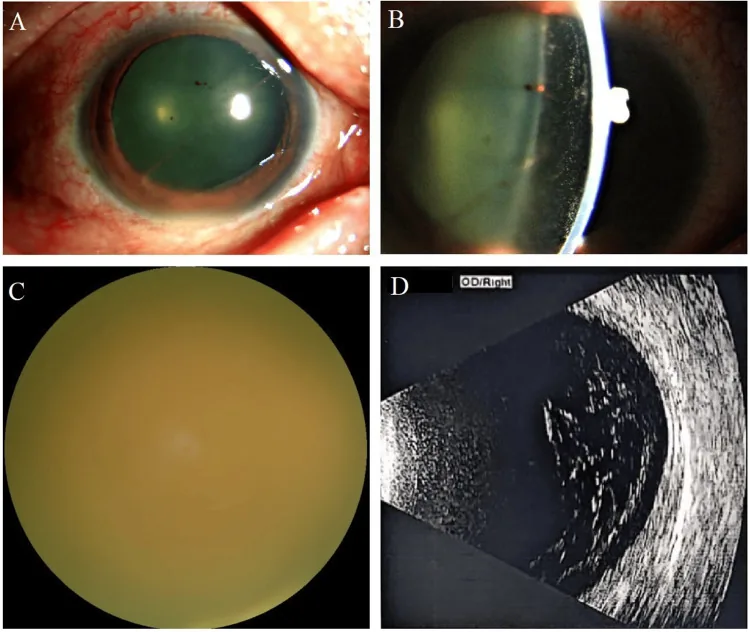
Floaters, blurred vision, and decreased visual acuity are common early symptoms. In advanced cases, eye pain and redness may occur.
The rate of disease progression varies depending on the causative organism.
Yellow-white fluffy chorioretinal infiltrates with indistinct borders, predominantly in the posterior pole. May be accompanied by intraretinal hemorrhages.
The appearance of vitreous opacities is similar to non-infectious uveitis such as sarcoidosis, and it has been reported that up to 50% are misdiagnosed in the early stage2). Additionally, fungal infection may be overlooked as postoperative recurrent vitritis4). For details, see the “Diagnosis and Examination Methods” section.
Risk factors differ between endogenous and exogenous types. The main risk factors are shown below.
| Classification | Main Risk Factors |
|---|---|
| Endogenous | Central venous nutrition, immunosuppressants, steroids |
| Endogenous | Organ transplantation, neutropenia, malignancy |
| Endogenous | Diabetes, prolonged ICU stay |
| Exogenous | Intraoperative complications, advanced age, steroid eye drops |
| Exogenous | Corneal ulcer, improper contact lens use |
COVID-19-related cases have been reported where prolonged hospitalization and systemic steroid administration led to immunosuppression and triggered fungal endophthalmitis 1, 9). Other reported causes include breast cancer chemotherapy (docetaxel-induced leukopenia) 3), immunosuppressive therapy (methotrexate + steroids) 2), keratitis spreading from poor hygiene during contact lens use 5), fungal biofilm colonization on IOL 6), infection by phytopathogenic fungi due to plant trauma 7), and poor diabetes control 4, 8).
Yes. Systemic steroids used for COVID-19 treatment and prolonged ICU stay can cause immunosuppression and trigger endogenous fungal endophthalmitis 1, 9). If vision changes occur, prompt ophthalmologic evaluation is necessary.
Intraocular fluid culture is the gold standard. Vitreous samples have a higher diagnostic yield than aqueous humor. Sabouraud agar is used, and cultures are incubated at both 37°C and room temperature for at least 2 weeks.
Blood cultures are positive in only one-third to one-half of endogenous cases, so a negative result does not rule out endophthalmitis.
Enrichment culture: Inoculating vitreous fluid into blood culture bottles has been reported to yield positivity in an average of 1.23 days 6).
Once the causative organism is identified, antifungal susceptibility testing should always be performed. Aspergillus terreus shows high resistance (98%) to amphotericin B (MIC 2 μg/mL) 2). In a case where fluconazole susceptibility (MIC 2.0 μg/mL) of Trichosporon inkin was confirmed, fluconazole monotherapy was successful 6).
Yes. Even in endogenous endophthalmitis, blood cultures are positive in only one-third to one-half of cases. β-D-glucan can also be negative 3). Direct culture and PCR of intraocular fluid are essential for definitive diagnosis.
The characteristics of major antifungal agents are shown below.
| Drug | Route | Characteristics |
|---|---|---|
| Fluconazole | IV/Oral | First-line for Candida |
| Voriconazole | Intravenous / Oral | Effective against filamentous fungi and resistant Candida |
| Amphotericin B | Intravenous / Intravitreal | Broad spectrum, limited intraocular penetration |
Treatment duration is usually 4–6 weeks. Successful control of Trichosporon inkin with 16 weeks of fluconazole has been reported 6).
It is performed for physical removal (debulking) of the infection source, enhancement of drug diffusion, and specimen collection. It is indicated in the following cases.
In IOL-related cases, removal of the IOL and the lens capsule including biofilm is important 5, 6).
Candida
Chorioretinitis only: Oral azoles (fluconazole, voriconazole)
Vitreous involvement: Add vitrectomy + intravitreal antifungal injection
Treatment duration: Generally 4–6 weeks
Aspergillus
First-line: Voriconazole (A. terreus has 98% amphotericin B resistance rate)
Local therapy: Vitrectomy + intravitreal voriconazole injection
Susceptibility testing: Mandatory (to confirm resistance)
Rare fungal species
Trichosporon: Echinocandins are ineffective. Use after confirming fluconazole susceptibility.
Others: After identification by MALDI-TOF MS, select based on drug susceptibility testing.
In principle, they should be avoided. Steroids promote fungal growth and risk rapid worsening of the condition. There are reports of rapid exacerbation after steroid (intravitreal dexamethasone) administration in A. terreus endophthalmitis2). It is important to rule out fungal infection before using steroid eye drops or intravitreal injections.
Fungemia → colonization of choroid and retina → formation of infectious foci → vitreous seeding → endophthalmitis → panuveitis → panophthalmitis, a stepwise progression.
Koide et al. (2023) reported a case of C. rugosa endophthalmitis and suggested an invasion route into the vitreous cavity via the Virchow-Robin space 3). C. rugosa is a rare species, accounting for less than 0.1% of non-albicans Candida, but can cause invasive infection in immunocompromised states.
Kaderli Tamer et al. (2022) reported that in a case of endophthalmitis that developed during COVID-19 treatment, steroids enhanced the disruption of the blood-retinal barrier and promoted fungal intraocular invasion 9).
Trichosporon species produce a capsular polysaccharide (glucuronoxylomannan; GXM) antigen, which inhibits phagocytosis by phagocytic cells, thereby establishing invasive infection 6). This mechanism, together with natural resistance to echinocandins, is the main cause of treatment-refractory disease.
As a novel treatment for refractory fungal endophthalmitis, a technique using 0.025% povidone-iodine as an irrigating solution during vitrectomy has been reported.
Huang et al. (2024) performed 0.025% povidone-iodine perfusion vitrectomy in a case of severe endophthalmitis caused by Arthrographis kalrae 5). The infection, which was difficult to control with conventional antifungal drugs, was controlled by this surgery. Povidone-iodine has no resistance mechanisms and is expected as a countermeasure against multidrug-resistant fungi.
Breakpoints (clinical thresholds) for antifungal drugs against Trichosporon species have not yet been established 6). Currently, individual treatment based on drug susceptibility test results at each facility is the norm, and the development of evidence-based treatment guidelines is a future challenge.