Grade 0-1 (None to Mild)
Findings: 12 or fewer central guttae (Grade 0) or more than 12 non-confluent guttae (Grade 1)
Symptoms: Usually asymptomatic. Detected as dark spots on specular microscopy.

Fuchs endothelial corneal dystrophy (FECD) is a progressive disease in which corneal endothelial cells become abnormal bilaterally. In 1910, Ernst Fuchs first reported 13 cases as “dystrophia epithelialis corneae,” and it was later identified as an endothelial disease, leading to its current name 1).
Guttae (guttata) appear on the endothelial surface of the central cornea and gradually spread to the periphery. When the barrier and pump functions (Na⁺/K⁺-ATPase) of endothelial cells decline, corneal stromal edema develops, progressing to epithelial edema and bullae formation. Thickening and irregularity of Descemet membrane lead to loss of corneal transparency.
In the IC3D (International Classification of Corneal Dystrophies) 2nd edition (Weiss 2015), FECD is classified under the “Corneal Endothelial Dystrophies” category 15). It is broadly divided into the following two types based on age of onset.
| Indicator | Value | Source |
|---|---|---|
| Frequency of guttae in preoperative cataract patients | 1.2% | Domestic multicenter survey |
| Prevalence in Japan (Kumejima Study, age ≥40) | 4.1% | Higa 20117) |
| Prevalence in Japanese women (age ≥40) | 5.8% | Higa 20117) |
| Japan: Male prevalence (age 40+) | 2.4% | Higa 20117) |
| Iceland: Reykjavik Eye Study, age 55+ | Women 11%, men 7% | Zoega 200610) |
| Sex ratio (international) | 2.5:1 to 3.5:1 (female predominance) | Matthaei 20191) |
| Frequency of TCF4 repeat expansion in Japanese | 12 out of 47 cases (26%) | Nakano 20158) |
Japanese people, as a yellow race, tend to have a lower incidence of FECD compared to white and black races. However, with the aging of society in Japan, the number of cases is expected to increase further. Japanese people have a higher corneal endothelial cell density than white people, which is thought to delay the onset of the disease.
In Japanese people, who often have narrow angles, laser iridotomy (LI) frequently leads to a decrease in endothelial cells, so careful attention is needed for early detection of FECD.
In a population-based study conducted in Okinawa’s Kumejima Island (Kumejima Study), corneal guttae were detected in 4.1% of individuals aged 40 years or older. The prevalence was 5.8% in women and 2.4% in men 7). There is also domestic data showing that 1.2% of patients undergoing preoperative cataract examination had cornea guttata. Although the frequency is reported to be lower in Japanese than in Western populations, it is increasing with the aging society.
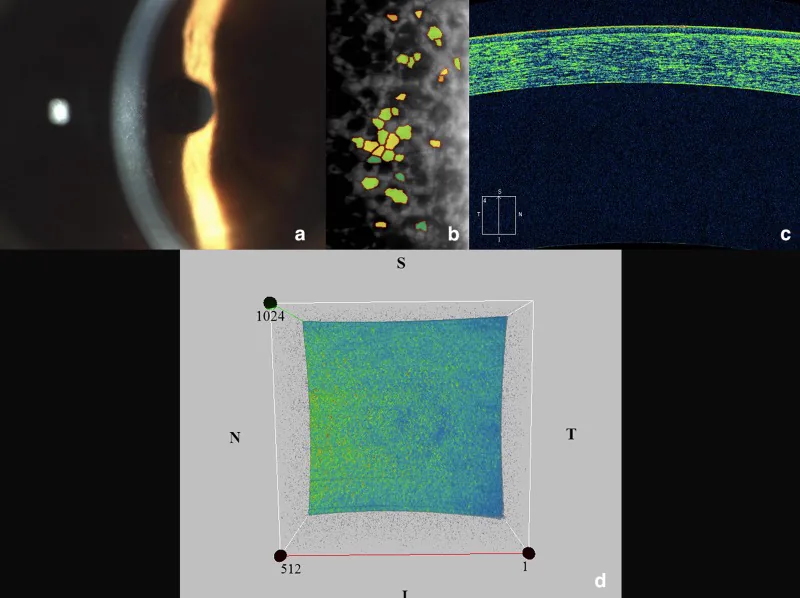
Typically, patients under 50 years of age are asymptomatic. Symptoms progress slowly, correlating with the degree of edema.
Slit-lamp microscopy is fundamental. Observation is performed using a combination of direct illumination, retroillumination, and specular reflection.
Grade 0-1 (None to Mild)
Findings: 12 or fewer central guttae (Grade 0) or more than 12 non-confluent guttae (Grade 1)
Symptoms: Usually asymptomatic. Detected as dark spots on specular microscopy.
Grade 2-3 (Moderate)
Findings: Confluent guttae in the central 1–5 mm. Mild beaten-metal appearance.
Symptoms: Morning blurring. Endothelial image becomes unclear on specular microscopy.
Grade 4 (Severe)
Findings: Extensive confluent guttae >5 mm centrally. Beaten-metal appearance with pigmentation.
Symptoms: Persistent blurring and photophobia from morning through the day.
Grade 4+ Edema (Very Severe)
Findings: Stromal edema, epithelial edema, and bullae formation. Marked corneal opacity.
Symptoms: Severe vision loss, eye pain, and tearing throughout the day. Quality of life significantly reduced.
This clinical staging is based on the modified classification by Krachmer et al. (1978)5).
Details of slit-lamp findings:
In a healthy cornea, endothelial cells constantly pump water out to the anterior chamber to maintain transparency. In FECD, because the endothelial pump function is reduced, evaporation of water also stops while sleeping (with eyes closed), so the cornea is most edematous in the morning and foggy vision is worse. When the eyes are open, water evaporates from the corneal surface, and during the day the edema improves to some extent, so vision recovers. As the disease progresses, this diurnal variation disappears and foggy vision persists all day.
FECD is mainly autosomal dominant, but there is variability in penetrance and expressivity, and some cases have no clear family history.
Major causative genes:
In Japanese patients, the frequency of TCF4 repeat expansion is lower than in Western populations, so other genetic backgrounds need to be elucidated8).
FECD is mainly inherited in an autosomal dominant pattern. Theoretically, the probability of passing it to a child is 50%. However, there is great variability in the age of onset and severity (incomplete penetrance); many people who inherit the gene live their entire lives with only very mild symptoms. In particular, the proportion of TCF4 gene abnormalities, which is the most common cause in Western countries, is low in Japanese people8), suggesting possible differences in genetic background. If you are concerned, we recommend consulting a genetic specialist.
Although there are no unified diagnostic criteria in Japan, clinical diagnosis is made by combining the following examinations.
This is the most important examination for diagnosing and monitoring FECD.
| Parameter | Normal Value | Abnormal Threshold |
|---|---|---|
| Endothelial cell density (neonatal period) | 3,500–4,000 cells/mm² | — |
| Endothelial cell density (20s) | 2,700 cells/mm² | — |
| Endothelial cell density (70 years and older) | Average 2,200 cells/mm² | — |
| Minimum for corneal transparency | — | 400–500 cells/mm² or less |
| CV (coefficient of variation) | 0.2–0.3 | ≥ 0.35 |
| Hexagonality | 60–70% | ≤ 50% |
| Disease | Key Points for Differentiation |
|---|---|
| Posterior polymorphous corneal dystrophy (PPCD) | AD inheritance, bilateral, band-like and vesicular opacities of Descemet’s membrane. Genes: PPCD1 (20p11.2-q11.2), PPCD2 (COL8A2), PPCD3 (ZEB1) |
| Congenital hereditary endothelial dystrophy (CHED) | AR inheritance (SLC4A11 mutation), onset at birth to infancy, corneal edema and opacity from birth |
| Pseudophakic bullous keratopathy (PBK) | Endothelial damage after cataract surgery. No guttae, history of surgery |
| Pseudoexfoliation syndrome keratopathy | PEX material deposition, elevated intraocular pressure, PEX material on the anterior lens capsule is key for differentiation |
| Iridocorneal endothelial (ICE) syndrome | Unilateral, with iris atrophy, anterior synechiae, and glaucoma. No guttae |
| Endothelial changes in narrow-angle eyes | May show cornea guttata-like findings. Differentiated by intraocular pressure and angle morphology |
A specular microscope (specular reflection endothelial camera) is a device that non-invasively photographs and measures the endothelial cells in the innermost layer of the cornea using special light reflection. The examination measures endothelial cell count (cell density), variation in cell size (CV value), and uniformity of shape (hexagonal cell ratio). In FECD, guttae appear as black spots (dark spots), helping to assess disease stage. The imaging takes a few minutes and is painless.
The goal of treatment is to restore corneal transparency and maintain vision. Depending on the stage of the disease, symptomatic therapy or surgical treatment is selected.
Aimed at symptom relief before surgery is indicated. It does not restore endothelial cell count or suppress disease progression.
DMEK (Descemet Membrane Endothelial Keratoplasty)
Graft: Descemet membrane + endothelium only (thickness about 15 μm)
Features: First reported by Melles in 2006 11). Rapid visual recovery, low rejection rate. Requires a skilled surgeon.
Insurance coverage in Japan: Since 2016
DSAEK (Descemet Stripping Automated Endothelial Keratoplasty)
PKP (Penetrating Keratoplasty)
Graft: Full-thickness cornea (diameter 7.0–8.5 mm)
Features: Classic option. Challenges include suturing, astigmatism management, and long-term rejection risk. In FECD, it is gradually being replaced by endothelial keratoplasty.
DSO (Descemetorhexis Without Endothelial Keratoplasty)
Procedure: Selective stripping of the central 4 mm of Descemet membrane only. No graft required.
Indications: Cases where residual peripheral endothelial cells can migrate and proliferate to the center. Approximately 75% achieve corneal clearance 14).
ROCK inhibitor eye drops: Postoperative use of ripasudil promotes clearance even in non-responsive cases 14).
| Parameter | DMEK | UT-DSAEK | Source |
|---|---|---|---|
| 12-month BCVA (logMAR difference) | −0.06 (DMEK superior) | — | Sela 2023 meta-analysis 3) |
| Achievement rate of 20/25 or better | 66% | 33% (p=0.02) | Dunker 2020 RCT4) |
| OR of rebubbling | — | 2.76 (favoring DSAEK) | Sela 20233) |
| 12-month ECD | No difference | No difference | Dunker 20204) |
| Graft thickness <70 μm | — | No difference in visual acuity with DMEK | Sela 20233) |
In a meta-analysis by Sela et al. (2023) of 8 studies (376 eyes), BCVA at 12 months was significantly better with DMEK (−0.06 logMAR)3). A multicenter RCT by Dunker et al. (2020) also showed a higher rate of achieving 20/25 or better with DMEK compared to UT-DSAEK (66% vs 33%, p=0.02)4). However, for UT-DSAEK with graft thickness less than 70 μm, the difference from DMEK was reduced3).
The Kyoto University group (Kinoshita 2018) developed a treatment method in which cultured healthy donor corneal endothelial cells are injected into the anterior chamber together with a ROCK inhibitor (Y-27632)13).
ROCK inhibitors exert their effect by promoting endothelial cell adhesion, suppressing apoptosis, and advancing the cell cycle13).
FECD is often associated with cataracts, and careful consideration is needed regarding the timing and method of surgery.
DMEK uses the thinnest graft (about 15 μm), offering faster visual recovery and fewer postoperative refractive changes. Meta-analyses also show superior BCVA at 12 months with DMEK3). On the other hand, DSAEK graft manipulation is somewhat easier, with a shorter learning curve for surgeons, and it is widely performed in Japan. Ultra-thin DSAEK (<70 μm) has been reported to achieve visual outcomes nearly equivalent to DMEK3). The choice is made based on a comprehensive assessment of the surgeon’s experience, the facility’s experience, and the patient’s corneal condition. Both procedures have been covered by insurance in Japan since 2016 (DMEK) or 2009 (DSAEK).
Normal corneal endothelial cells do not undergo cell division in the anterior chamber. When endothelial cells are lost, adjacent cells enlarge and migrate to cover the defect, causing cell density to irreversibly decrease with age. When it falls below 400–500 cells/mm², maintaining corneal transparency becomes difficult.
In FECD, degenerated endothelial cells produce and deposit abnormal collagen-like material on the posterior surface of Descemet membrane, forming guttae. Descemet membrane becomes thickened and irregular, creating a vicious cycle that further impairs endothelial function.
Aging, UV exposure, and smoking all increase oxidative stress and serve as entry points for the vicious cycle 2).
The pump function of the corneal endothelium depends on Na⁺/K⁺-ATPase. When endothelial cells are damaged, edema occurs through the following pathways.
If intraocular pressure rises (ocular hypertension) beyond the swelling pressure of the corneal stroma, epithelial edema may occur even if the endothelium is relatively healthy. Caution is required.
Since the contribution of TCF4 repeat expansion is relatively small in Japanese individuals8), elucidating the genetic and environmental backgrounds unique to Japanese people is an important future issue.
Matthaei M, Hribek A, Clahsen T, Bachmann B, Cursiefen C, Jun AS. Fuchs Endothelial Corneal Dystrophy: Clinical, Genetic, Pathophysiologic, and Therapeutic Aspects. Annu Rev Vis Sci. 2019;5:151-175.
Ong Tone S, Kocaba V, Böhm M, Wylegala A, White TL, Jurkunas UV. Fuchs endothelial corneal dystrophy: The vicious cycle of Fuchs pathogenesis. Prog Retin Eye Res. 2021;80:100863.
Sela TC, Iflah M, Muhsen K, Zahavi A. Descemet membrane endothelial keratoplasty compared with ultrathin Descemet stripping automated endothelial keratoplasty: a meta-analysis. Br J Ophthalmol. 2023.
Dunker SL, Dickman MM, Wisse RPL, et al. Descemet Membrane Endothelial Keratoplasty versus Ultrathin Descemet Stripping Automated Endothelial Keratoplasty: A Multicenter Randomized Controlled Clinical Trial. Ophthalmology. 2020;127(9):1152-1159.
Krachmer JH, Purcell JJ Jr, Young CW, Bucher KD. Corneal endothelial dystrophy. A study of 64 families. Arch Ophthalmol. 1978;96(11):2036-2039.
Gain P, Jullienne R, He Z, et al. Global Survey of Corneal Transplantation and Eye Banking. JAMA Ophthalmol. 2016;134(2):167-173.
Higa A, Sakai H, Sawaguchi S, et al. Prevalence of and risk factors for cornea guttata in a population-based study in a southwestern island of Japan: the Kumejima study. Arch Ophthalmol. 2011;129(3):332-336.
Nakano M, Okumura N, Nakagawa H, et al. Trinucleotide repeat expansion in the TCF4 gene in Fuchs’ endothelial corneal dystrophy in Japanese. Invest Ophthalmol Vis Sci. 2015;56(8):4865-4869.
Wieben ED, Aleff RA, Tosakulwong N, et al. A common trinucleotide repeat expansion within the transcription factor 4 (TCF4) gene predicts Fuchs corneal dystrophy. PLoS One. 2012;7(11):e49083.
Zoega GM, Fujisawa A, Sasaki H, et al. Prevalence and risk factors for cornea guttata in the Reykjavik Eye Study. Ophthalmology. 2006;113(4):565-569.
Melles GR, Ong TS, Ververs B, van der Wees J. Descemet membrane endothelial keratoplasty (DMEK). Cornea. 2006;25(8):987-990.
Price MO, Feng MT, Price FW Jr. Endothelial Keratoplasty Update 2020. Cornea. 2021;40(5):541-547.
Kinoshita S, Koizumi N, Ueno M, et al. Injection of Cultured Cells with a ROCK Inhibitor for Bullous Keratopathy. N Engl J Med. 2018;378(11):995-1003.
Moloney G, Petsoglou C, Ball M, et al. Descemetorhexis Without Grafting for Fuchs Endothelial Dystrophy-Supplementation With Topical Ripasudil. Cornea. 2017;36(6):642-648.
Weiss JS, Møller HU, Aldave AJ, et al. IC3D classification of corneal dystrophies—edition 2. Cornea. 2015;34(2):117-159.
Seitzman GD, Gottsch JD, Stark WJ. Cataract surgery in patients with Fuchs’ corneal dystrophy: expanding recommendations for cataract surgery without simultaneous keratoplasty. Ophthalmology. 2005;112(3):441-446.