Phenotype 1
Distribution: Type concentrated in the macular area.
Complication risk: Lowest among the three types, with a 5-year estimated incidence of GA of 12.9%.

Basal laminar drusen (BLD) is a disease concept first described by Gass in 1977 as numerous small yellow drusen scattered in the fundus. The “stars-in-the-sky” pattern on fluorescein angiography (FA) is characteristic. 1)
Initially named “basal laminar drusen” as nodular thickening of the RPE basement membrane, subsequent research revealed that deposits occur between the RPE basement membrane and the inner collagenous layer of Bruch’s membrane, i.e., the same location as typical soft drusen. Therefore, the term “cuticular drusen” is now recommended. 1)
Although it belongs to the age-related macular degeneration (AMD) spectrum, it has the following features distinct from typical AMD:
Although it is part of the age-related macular degeneration spectrum, it has unique features such as relatively younger age of onset, female predominance, characteristic “stars-in-the-sky” and “saw-tooth” imaging findings, and strong association with CFH gene mutations. It is also considered a precursor lesion of age-related macular degeneration and may progress to macular complications during long-term follow-up. 1)
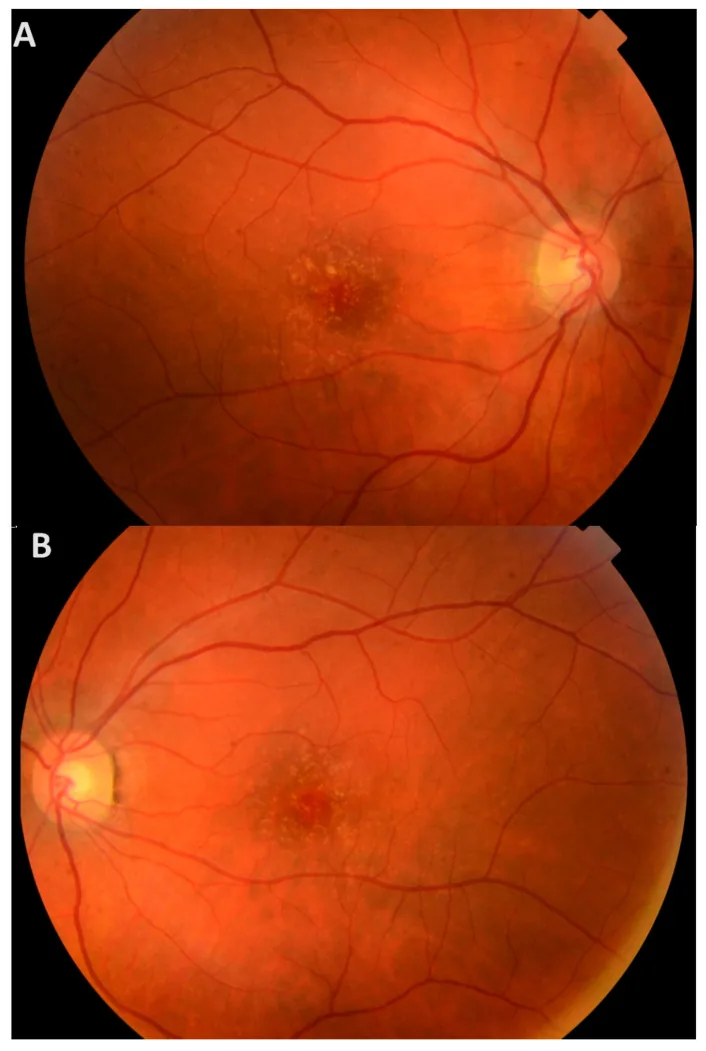
In the early stages, subjective symptoms are scarce, and most patients remain asymptomatic until later in life. Even if drusen are present in the macula, there are no symptoms unless the outer retinal layers are damaged.
If drusen persist for a long time, RPE dysfunction leads to outer retinal damage. When the fovea is involved, the following symptoms appear.
On dilated fundus examination, numerous small yellow drusen are observed throughout the retina. They are recognized as multiple hard, elevated yellowish-white drusen beneath the RPE.
The clinical phenotype is classified into three types. 1)
Phenotype 1
Distribution: Type concentrated in the macular area.
Complication risk: Lowest among the three types, with a 5-year estimated incidence of GA of 12.9%.
Phenotype 2
Distribution: Type scattered in the posterior pole, extending to the periphery and nasal side.
Complication risk: 5-year estimated incidence of GA is 38.7%. MNV risk is also high.
Phenotype 3
Distribution: Type with mixed large drusen (>200 μm).
Complication risk: Highest, with a 5-year estimated incidence of GA of 43.6%.
Main clinical findings are shown below.
On OCT, it is classified into three types.1)
The etiology of basal laminar drusen is not fully understood. Currently identified risk factors are listed below.
Genetic Factors
Sex Differences and Family History
Association with Systemic Diseases
This is because abnormalities in the alternative complement pathway due to CFH gene mutations cause similar deposits in both the eye (between the RPE and Bruch’s membrane) and the kidney (glomerular basement membrane). Especially in young patients (ages 5–30), screening for MPGN and atypical hemolytic uremic syndrome (aHUS) is recommended. 1)
Diagnosis involves combining multiple imaging modalities. Confirmation with at least three of the following is recommended: color fundus photography (CFP), SD-OCT, FA, and fundus autofluorescence (FAF). 1)
Typical findings for each examination are shown below.
| Examination Method | Typical Findings | Notes |
|---|---|---|
| Color Fundus Photography (CFP) | Numerous small yellow drusen | Scattered from the posterior pole to the periphery |
| FA | ”Starry-sky” pattern | Multiple hyperfluorescent spots in the arteriovenous phase 1) |
| FAF | Central hypoautofluorescence with hyperautofluorescent rim | Accurate identification rate: 62% in present cases, 100% in absent cases 1) |
| SD-OCT | Triangular elevation of RPE-BL, “saw-tooth” | Classified into 3 types 1) |
ICG angiography (ICGA): Shows early hyperfluorescence, but drusen cannot be detected in about 50% of cases. 1)
Key points for differential diagnosis
No established treatment exists to eliminate drusen themselves. Isolated basal laminar drusen do not require treatment; regular follow-up is the standard.
The following complication rates have been reported. 1)
Treatment strategies according to complications are shown below.
When CNV is present
Intravitreal anti-VEGF injection: First-line treatment. Use ranibizumab, aflibercept, brolucizumab, etc.
Photodynamic therapy (PDT): Considered when anti-VEGF is ineffective or contraindicated.
When GA is present
No effective treatment: No established method to halt progression.
Continued observation: Regular examinations to detect CNV early.
Vitelliform lesions
Observation: May regress spontaneously.
Cases with CNV: Consider anti-VEGF therapy.
About supplements
The efficacy of AREDS (Age-Related Eye Disease Study) supplements for patients with basal laminar drusen has not been established. Common AREDS formulations include Ocuvite PreserVision 2 (3 tablets, 3 times daily) and Sante Lutax 20+ Vitamin & Mineral (3 tablets, 3 times daily).
Self-monitoring
Daily monitoring with an Amsler chart can detect early changes in metamorphopsia and central scotoma.
Currently, there is no treatment to eliminate drusen themselves. If CNV is present, intravitreal anti-VEGF injection or photodynamic therapy (PDT) may be indicated. There is no established treatment for GA, and continuous observation is important. See “Standard treatment” section for details.
Basal laminar drusen are polymorphous material that accumulates between the RPE basement membrane and the inner collagenous layer of Bruch’s membrane. Their components include membranous debris, unesterified cholesterol, and complement.
Formation Mechanism
RPE is responsible for lipoprotein excretion, but its metabolic turnover deteriorates with age. Degenerated extracellular matrix accumulates, forming the basis of drusen. Overactivation of the complement cascade due to CFH gene mutations (especially Tyr402His) accelerates this process. 1)
Drusen Life Cycle
Cuticular drusen are dynamic and undergo cycles of growth, fusion, and regression. 1)
Course of Vitelliform Lesions
Vitelliform lesions progress in the following order. 1)
Carriers of rare CFH gene variants (e.g., p.Arg1210Cys) tend to develop the disease at a younger age. Chronic overactivation of the alternative complement pathway due to CFH mutations is thought to cause drusen formation earlier than in typical AMD. 1)
Therapies Targeting the Complement Pathway
Since abnormalities in the alternative complement pathway due to CFH gene mutations form the pathological basis, compounds targeting the complement pathway are being actively researched. Because systemic administration carries a high risk of side effects, local (intraocular) administration is attracting attention as a promising approach.
Prognostic Prediction Using Multimodal Imaging
Advances in multimodal imaging are enabling early identification of patients at high risk for complications. 1) Phenotype classification (1–3) is useful for prognosis prediction, and Phenotypes 2 and 3, as high-risk groups for GA and MNV, require particularly careful follow-up. 1)
Potential for Personalized Medicine
Advances in genetic analysis are expected to enable personalized medicine based on variants in AMD-related genes, including the CFH gene. In particular, carriers of rare variants (e.g., p.Arg1210Cys) may become targets for early screening and intervention.