Acute HIT
Platelet count: Decreased
Functional assay (SRA): Positive
Immunoassay (ELISA): Positive
The highest risk period. Discontinue all heparin immediately and switch to a non-heparin anticoagulant.

Heparin-Induced Thrombocytopenia (HIT) is an immune-mediated reaction to heparin administration. IgG antibodies are produced against the complex of platelet factor 4 (PF4) and heparin, leading to platelet activation. This paradoxically results in a decrease in platelet count while the blood becomes hypercoagulable.
HIT is classified into type I and type II. HIT type I is a transient thrombocytopenia that resolves spontaneously. HIT type II is antibody-mediated and a severe condition with thromboembolic complications. 1)
Ocular complications occur when HIT-related thrombosis or hemorrhage affects the ocular vasculature. Ophthalmic management is required in parallel with systemic management of HIT.
In patients receiving UFH, the incidence is up to 5%, and in those receiving LMWH, it is about one-tenth of that. After cardiac surgery, the incidence is 0.1–3%, higher than in the general population, and the mortality rate can reach up to 10%. 2)
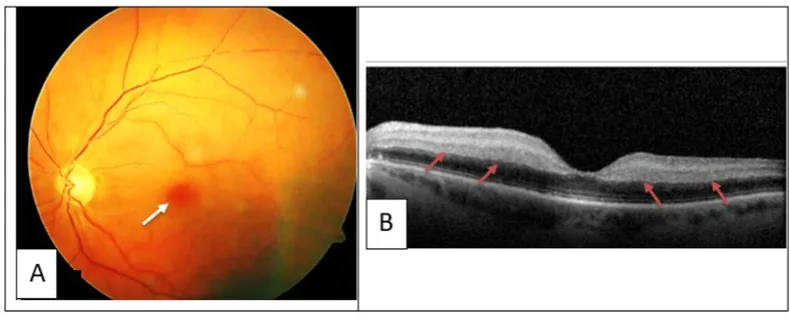
Ocular symptoms vary depending on the location of thrombosis or hemorrhage.
The cause of HIT is an immune-mediated reaction to heparin administration. The main risk factors are as follows.
HIT can also occur with LMWH, but the incidence is about 1/10 that of UFH. Even though the risk is lower, platelet monitoring cannot be omitted.
HIT is a diagnosis of exclusion. It is suspected when thrombosis and thrombocytopenia occur 5–14 days after heparin administration and other causes are ruled out.
The 4T score is a clinical probability score consisting of four criteria: Thrombocytopenia, Timing, Thrombosis, and oTher causes. Each criterion is scored 0–2 points, and the total score classifies the probability.
| Total Score | Probability Classification |
|---|---|
| 6–8 points | High probability |
| 4–5 points | Moderate |
| 0–3 points | Low probability |
The positive predictive value of the 4T score is approximately 10% for a moderate score of 4 points and approximately 80% for a high probability score of 8 points. 4)
In cardiac surgery patients, platelet count typically decreases by 30–50% after CPB, and if a biphasic pattern of thrombocytopenia is observed, it is considered a characteristic finding of HIT. 3)1)
The main diagnostic tests are the anti-PF4/heparin antibody test (anti-PF4 ELISA) and the serotonin release assay (SRA).
The ASH guidelines recommend anti-PF4 ELISA for patients with a moderate or higher 4T score, and if positive, confirm with SRA. 2)
Rare discordant cases of anti-PF4 ELISA negative and SRA positive (16 of 8,546 cases, 0.2%) have been reported; caution is needed when HIT is strongly suspected clinically. 2)
The management strategy for HIT differs depending on the phase. 3)
Acute HIT
Platelet count: Decreased
Functional assay (SRA): Positive
Immunoassay (ELISA): Positive
The highest risk period. Discontinue all heparin immediately and switch to a non-heparin anticoagulant.
Subacute HIT A
Platelet count: Normalized
Functional assay (SRA): Positive
Immunoassay (ELISA): Positive
Platelet count has recovered but functional assay is positive. Cardiac surgery should be postponed if possible.
Subacute HIT B
Platelet count: Normal
Functional assay (SRA): Negative (median 50 days to seroconversion)
Immunoassay (ELISA): Positive
Intraoperative short heparin exposure is low risk. Continue heparin avoidance postoperatively.
Remote HIT
Platelet count: Normal
Functional assay (SRA): Negative
Immunoassay (ELISA): Negative (PF4/H antibodies disappear at a median of 85 days)
Short-term heparin administration upon re-exposure is possible.
In patients with ocular symptoms secondary to HIT, a thorough history and physical examination should be performed to check for other systemic symptoms.
This score evaluates the clinical probability of HIT based on four criteria: degree of thrombocytopenia, timing of onset, presence of thrombosis, and exclusion of other causes. The total score ranges from 0 to 8, with a score of 6 or higher indicating high probability.
When HIT is diagnosed or strongly suspected, the following should be implemented immediately.
Argatroban
Classification: Direct thrombin inhibitor
Half-life: 39–51 minutes (normal liver function), up to 181 minutes with liver impairment
Metabolism: Hepatobiliary excretion. Advantageous in patients with renal impairment.
Bivalirudin
Classification: Direct thrombin inhibitor
Half-life: 25 minutes (normal to mild renal impairment)
PCI dose: 0.75 mg/kg bolus + 1.75 mg/kg/h
CPB dose: 1 mg/kg bolus + 2.5 mg/kg/h (add 50 mg to priming solution)3)
The ASH 2018 guidelines recommend bivalirudin for PCI. In 52 cases of PCI use, a procedure success rate of 98% and major bleeding of 1.9% have been reported.3)
For refractory HIT in which thrombosis progresses despite appropriate non-heparin anticoagulation, consider the following. 5)
If a patient with acute HIT/subacute HIT A requires cardiac surgery, consider the following three options. 3)
Retinal vascular occlusion and orbital hemorrhage are managed ophthalmologically in parallel with systemic management of HIT.
Switch to a direct thrombin inhibitor such as argatroban or bivalirudin. Both directly inhibit thrombin through a mechanism different from heparin and are not affected by HIT antibodies. The choice of agent depends on liver function, kidney function, and clinical situation.
The onset of HIT progresses through the following stages.
Step 1: Complex formation and antibody production
PF4 (platelet factor 4) is a positively charged chemokine derived from megakaryocytes and stored in platelet alpha granules. PF4 forms complexes with negatively charged heparan sulfate (GAG on vascular endothelial surface) or LPS (gram-negative bacterial outer membrane). These complexes act as “danger signals” and enable rapid production of IgG antibodies.
Step 2: Formation of PF4-heparin complexes
Heparin administration leads to the formation of PF4-heparin complexes. Because heparin has a molecular structure similar to LPS and heparan sulfate, pre-existing IgG binds to it.
Step 3: Platelet activation and hypercoagulability
The IgG-PF4-heparin complex binds to FcγRII receptors (FcγRIIa) on platelets, activating them. The intrinsic pathway of the coagulation cascade is initiated, leading to widespread thrombosis and thrombocytopenia due to platelet consumption. This is the essence of the “paradoxical” pathophysiology of HIT.
In addition to normal PF4, antibodies targeting IL-8, protamine, and NAP-2 are detected in less than 1% of HIT workup cases. 2) Furthermore, anti-PF4 antibodies that activate platelets even in the absence of heparin (heparin-independent HIT antibodies) may be involved in the pathophysiology of refractory HIT and autoimmune HIT. 5)
After CPB, seroconversion of PF4/H antibodies occurs in up to 50% of patients, but only 1–2% develop clinical HIT. 1) High-dose UFH administration during CPB, release of tissue factor, and inflammation are thought to promote HIT antibody production. 3)
This is a deglycosylated formulation of the mouse monoclonal IgG2b anti-PF4/H antibody KKO. By blocking FcγRIIa-mediated binding, it suppresses platelet activation and complement activation.
In mouse models, it has been reported to improve thrombocytopenia and reduce thrombus size. 5) This is a stage that requires evaluation in future clinical trials.
A strategy of re-administering heparin during CPB using the P2Y12 inhibitor cangrelor is being investigated.
In a case series of 10 patients, the dose was adjusted using VerifyNow P2Y12 PRU, and no thrombotic complications were reported. 5) A standardized protocol has not yet been established.
Although the efficacy of TPE is recognized, a unified protocol has not been established.
A systematic review (30 cases) reported an average of 4 TPE sessions and a plasma exchange volume of 1.3 PV, but the level of evidence is low, and future prospective studies are needed. 5)
Tugulan C, Chang DD, Bates MJ. Heparin-Induced Thrombocytopenia After Mitral Valve Replacement. Ochsner J. 2021;21(1):100-104.
Attah A, Peterson C, Jacobs M, et al. Anti-PF4 ELISA-Negative, SRA-Positive Heparin-Induced Thrombocytopenia. Hematol Rep. 2024;16(1):90-97.
Pishko AM, Cuker A. Heparin-induced thrombocytopenia and cardiovascular surgery. Hematology Am Soc Hematol Educ Program. 2021;2021(1):478-485.
Mele M, Iacoviello M, Casavecchia G, et al. Coronary thrombosis due to heparin-induced thrombocytopenia after percutaneous coronary intervention. Clin Case Rep. 2021;9(6):e04291.
Adeoye O, Zheng G, Onwuemene OA. Approaches to management of HIT in complex scenarios, including cardiac surgery. Hematology Am Soc Hematol Educ Program. 2024;2024(1):267-278.