CNA1 (autosomal dominant type)
Severity: Mild
Causative gene: Unidentified (candidate gene in the 12q region)
Corneal refractive power: approximately 38 D (3–7 D lower than normal)
Central cornea: clear
Hyperopia: mild to moderate (mean +6.33 D)

Cornea plana is a congenital disorder in which the peripheral cornea becomes scleralized, resulting in flattening of the cornea. The border between the cornea and sclera becomes indistinct, and the clear corneal diameter is reduced. It is often bilateral and affects both males and females.
It is thought to arise from abnormalities in the limbal primordium during development. It is classified into two types: autosomal dominant CNA1 and autosomal recessive CNA2.
It is an extremely rare disease. CNA2 shows clustering due to founder effects in northern Finland and Saudi Arabia. Finnish cases account for approximately 80% of all CNA2 cases worldwide. Families with CNA1 have been reported in Denmark, Germany, the United States, the Netherlands, and Cuba.
CNA1 (autosomal dominant type)
Severity: Mild
Causative gene: Unidentified (candidate gene in the 12q region)
Corneal refractive power: approximately 38 D (3–7 D lower than normal)
Central cornea: clear
Hyperopia: mild to moderate (mean +6.33 D)
CNA2 (autosomal recessive type)
Severity: severe
Causative gene: homozygous mutation in KERA gene (12q)
Corneal refractive power: approximately 30 D or less1)
Central cornea: often with opacity
Hyperopia: high (greater than +10 D)
CNA1 is an autosomal dominant mild type. The central cornea is clear, corneal refractive power is about 38 D (close to normal), and hyperopia is mild. In contrast, CNA2 is an autosomal recessive severe type caused by KERA gene mutation. It involves central corneal opacity, corneal refractive power reduced to about 30 D, and high hyperopia exceeding +10 D. CNA2 often has other ocular complications such as angle abnormalities and iris abnormalities, and the prognosis is worse than CNA1.
Patients often present in childhood due to poor vision from high hyperopia. CNA2 may be accompanied by amblyopia. Some patients complain of decreased vision due to corneal opacity.
The cornea becomes flattened, and the border between the cornea and sclera is indistinct. The peripheral cornea shows opacification, and in CNA2, central opacification may also be observed. Arcus lipoides appears at a relatively young age. A shallow anterior chamber is noted, with reported anterior chamber depths ranging from 0.8 to 2.1 mm.
| Finding | CNA1 | CNA2 |
|---|---|---|
| Corneal refractive power | Approximately 38 D | Approximately 30 D or less |
| Central cornea | Clear | Opacification present |
| Hyperopia degree | Around +6.33 D | Over +10 D |
In CNA2, a round opaque thickening of 4–6 mm in diameter may be observed in the central cornea. This thickening adheres to Descemet’s membrane, and the cornea is thinnest around it. Iris abnormalities (iris atrophy, iris nodules, pupillary abnormalities) may also be present.
In some cases, K values were extremely flat at 21.8/27.0 D and 19.4/28.7 D, anterior chamber depths were 1.15 mm and 0.97 mm, and central corneal thicknesses were thin at 410 μm and 315 μm1).
CNA2 is caused by a homozygous mutation in the KERA gene on chromosome 12. The KERA gene encodes keratocan, a cornea-specific proteoglycan involved in regulating collagen fibril spacing and diameter.
Both missense and nonsense mutations have been reported. In particular, seven different KERA mutations have been identified in the Saudi Arabian population. To date, 15 KERA mutations have been reported, of which seven are missense mutations1).
The causative gene for CNA1 has not been identified. Analysis of two candidate genes on chromosome 12 is ongoing, but known genes such as DCN, DSPG3, LUM, PITX2, and FOXC1 have been excluded as causes.
Confirm scleralization of the peripheral cornea and corneal flattening. When the observation system of the slit lamp is swung widely to the side, the corneal flattening becomes obvious. In CNA2, central corneal opacity is also assessed.
Measurement of corneal refractive power is useful for differentiating CNA1 and CNA2. In CNA2, the cornea flattens to about 30 D, and in CNA1 to about 38 D. Older keratometers cannot measure below 30 D, so a technique of attaching a weak plus lens to extend the measurement range is used.
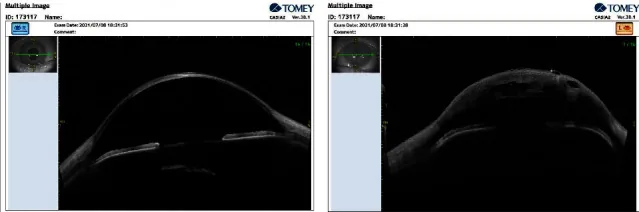
It is useful for visualizing the relationship between the cornea, angle, and iris. It is also used to evaluate the depth and extent of central corneal opacity.
| Disease | Key Points for Differentiation |
|---|---|
| Sclerocornea | Corneal opacity and flattening only |
| EDS Type VI | Associated with systemic symptoms |
| Rieger Anomaly | Anterior segment dysgenesis |
In addition, differentiation from posterior polymorphous corneal dystrophy (PPCD), mucopolysaccharidosis, and microcornea is necessary. Isolated corneal flattening without systemic malformations is likely to be cornea plana.
The diagnosis of cornea plana is based on slit-lamp examination showing corneal flattening and peripheral sclerocornea, and keratometry showing reduced corneal refractive power. In CNA2, corneal refractive power decreases to about 30 D. Anterior segment OCT is useful for detailed evaluation of the cornea and angle. Genetic testing of the KERA gene is used for definitive diagnosis. Differential diagnoses include sclerocornea, Ehlers-Danlos syndrome type VI, and Rieger anomaly, which must be excluded.
When opacities are limited to the periphery, the basic approach is hyperopic refractive correction. For spectacle prescription, it is recommended to prescribe lenses that are 3 to 4 diopters weaker than the objective measurement, based on subjective refraction testing.
If opacities extend to the central cornea, corneal transplantation should be considered. However, outcomes of full-thickness corneal transplantation are poor, and attention must be paid to glaucoma associated with angle abnormalities.
In flat cornea, cataract surgery is challenging due to shallow anterior chamber and abnormal K values. For intraocular lens power calculation, standard optical biometers may not be usable, and the SRK regression formula is sometimes employed1). In one case, a 34 D toric intraocular lens was used, achieving a postoperative corrected visual acuity of 6/121). During surgery, adequate use of viscoelastic substances and maintenance of the anterior chamber with high irrigation pressure are important1).
Due to shallow anterior chamber, there is a high risk of angle-closure glaucoma. Cases of open-angle glaucoma have also been reported. Regular intraocular pressure measurement and gonioscopy monitoring are essential.
It is possible but technically difficult. Due to shallow anterior chamber and abnormally flat cornea, intraocular lens power calculation may not be feasible with standard biometers. Alternative calculation methods such as the SRK regression formula are used. During surgery, it is important to maintain the anterior chamber with sufficient viscoelastic substances and protect the corneal endothelium. Toric intraocular lenses can also correct high astigmatism. Recent reports have shown good refractive outcomes.
Keratocan, encoded by the KERA gene, belongs to the Small Leucine-Rich Proteoglycan (SLRP) family. Keratocan has 10 leucine-rich repeat (LRR) motifs and controls the spacing (42–44 nm) and diameter of collagen fibrils in the corneal stroma1). This uniform fibril arrangement is essential for maintaining corneal transparency, refractive power, and strength.
KERA mutations destabilize the structure of keratocan and impair its binding to collagen fibrils. A novel mutation (c.659T>C, p.Leu220Ser) was shown by protein modeling to disrupt the hydrophobic core within the LRR motif and compromise the stability of the asparagine ladder1).
PACD is an autosomal dominant disorder caused by heterozygous deletions of four SLRP genes (KERA, LUM, DCN, EPYC) located at 12q21.332). Similar to cornea plana, it presents with decreased K values (less than 41 D) and corneal thinning2). Both conditions share corneal structural changes due to KERA gene abnormalities, but PACD involves deletions of multiple genes, whereas cornea plana CNA2 results from point mutations in the KERA gene alone.
Advances in genetic testing have led to the identification of novel KERA mutations. To date, 15 KERA mutations have been reported, contributing to improved accuracy of genetic counseling1). Pathogenicity prediction of variants of uncertain significance (VUS) using protein modeling is gaining attention as a new approach in genetic diagnosis1).
In cataract surgery, the use of toric intraocular lenses and refined biometry has enabled favorable refractive outcomes1). In the future, participation in clinical trials of cell therapy and gene therapy may become possible1).