
Confocal Microscopy (In Vivo Corneal Confocal Microscopy)
Key points at a glance
Section titled “Key points at a glance”1. What is confocal microscopy?
Section titled “1. What is confocal microscopy?”Confocal microscopy is a high-resolution microscope based on the optical principle that illumination and observation light share the same focal plane. In ophthalmology, it is applied to in vivo observation of the cornea and is also used for evaluation of Fuchs endothelial corneal dystrophy 7).
The main devices currently used clinically are the following two types.
Laser scanning type (HRT III-RCM): Manufactured by Heidelberg Engineering. It uses a 670 nm diode laser as the light source, with a 63× objective lens and a field of view of 400×400 μm. It has high resolution with lateral resolution of 1–2 μm and depth resolution of approximately 4 μm 1)2). It is used by contacting the cornea via a PMMA disposable cap (TomoCap) and ophthalmic gel.
Slit scanning type (Confoscan 4): Manufactured by Nidek. It uses a halogen light source and can also be used in non-contact mode. It has an automatic scanning function and is relatively easy to operate, but depth resolution is limited to 25–27 μm. Production has now been discontinued.
IVCM has three imaging modes. Section scan acquires a still image at a single depth. Volume scan acquires 30–40 consecutive sections at 2 μm intervals. Sequence scan acquires up to 100 frames of video at the same depth, suitable for observing dynamic changes.
IVCM requires specialized equipment and an operator skilled in its use, so it is mainly introduced at university hospitals and specialized ophthalmology facilities. It is not available at all eye clinics, but it is particularly useful for diagnosing corneal infections and corneal dystrophies, and patients are referred as needed.
2. Main findings and clinical significance
Section titled “2. Main findings and clinical significance”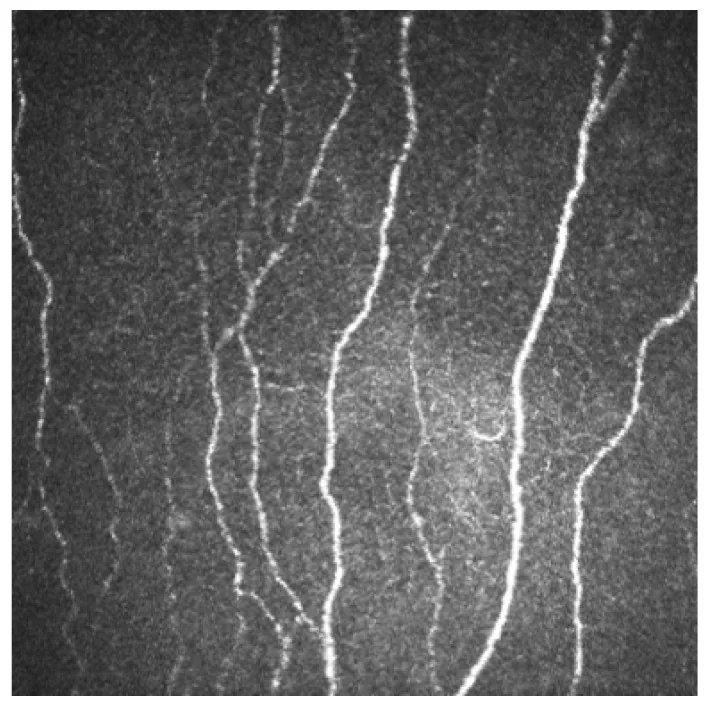
Normal corneal findings
Section titled “Normal corneal findings”IVCM allows individual observation of each layer of the cornea.
Corneal epithelium: Superficial cells are polygonal, 40–50 μm. Wing cells are 20–30 μm with bright cell borders, approximately 5,000 cells/mm². Basal cells are 8–10 μm with dark cytoplasm and bright borders (desmosomes), showing a honeycomb pattern. Basal cell density is 3,600–8,996 cells/mm².
Bowman’s layer: Observed as a structureless gray-white layer about 10 μm thick. Nerve bundles run through it.
Corneal stroma: Keratocytes appear dark when inactive and bright amoeboid when activated. Cell density is higher in the anterior stroma.
Descemet’s membrane: Recognized as a hazy layer 6–10 μm thick. Cellular structures are usually not identifiable.
Corneal endothelium: Hexagonal cells arranged in a mosaic. Cell diameter about 20 μm, density 2,550–2,720 cells/mm², decreasing by approximately 0.6% per year with age.
Subbasal nerve plexus: Observed as beaded curved structures running just beneath Bowman’s layer3). They form a whorl pattern about 1–2 mm inferonasal to the corneal center. Fiber diameter is 0.52–4.6 μm.
Dendritic cells (Langerhans cells): Distributed at 34 ± 3 cells/mm² in the central cornea and 98 ± 8 cells/mm² in the periphery. They serve as indicators of immune response.
Abnormal findings
Section titled “Abnormal findings”Corneal infections: In fungal keratitis, filamentous hyphae are clearly observed within the stroma8). Acanthamoeba is detected as cysts (spherical with double-walled structure), but differentiation from inflammatory cells requires experience8). Bacteria are difficult to visualize directly because individual organisms are too small5).
Corneal dystrophies: In Avellino corneal dystrophy (GCD2), highly reflective granular deposits are seen in the basal epithelial layer, and irregular highly reflective clusters are present in the superficial to mid stroma1). Lattice corneal dystrophy type I is characterized by reticular deposits and branching fibers1). Macular corneal dystrophy shows deposits with indistinct borders, and Schnyder crystalline corneal dystrophy shows needle-shaped crystals1).
Corneal ectasia: In keratoconus, decreased keratocyte density is observed, correlating with severity. Breaks in Bowman’s layer are also noted. In post-PRK corneal ectasia, loss of Bowman’s layer and reduction of anterior keratocytes are characteristic, showing a different IVCM pattern from keratoconus2).
3. Examination technique and method
Section titled “3. Examination technique and method”The IVCM examination procedure is as follows.
Pre-treatment: Administer topical anesthesia (e.g., oxybuprocaine). Apply ophthalmic gel to a disposable TomoCap (made of PMMA) and attach it to the tip of the objective lens.
Examination: Applanate the cornea while monitoring the contact between the lens and cornea using a CCD camera. Adjust the focus manually and observe sequentially from the superficial layer (0 μm) to the deeper layers. Fixation is very important, and patient cooperation is essential. The examination takes about 5 to 15 minutes.
Complications: Corneal epithelial abrasion and infection (higher risk if pre-existing epithelial defect) may rarely occur.
| Parameter | HRT III-RCM | Confoscan 4 |
|---|---|---|
| Light source | Diode laser | Halogen light |
| Depth resolution | 4 μm | 25–27 μm |
| Contact | Required (TomoCap) | Optional |
Because topical anesthesia is applied, there is almost no pain during the examination. You may only feel a light touch of the cap on the cornea. A temporary foreign body sensation may occur after the examination, but it usually resolves quickly.
4. Optical Principles and Device Characteristics
Section titled “4. Optical Principles and Device Characteristics”The basic principle of confocal microscopy is “confocality” 7). Both the illumination and detection systems have pinhole apertures, selectively detecting only reflected light from the focal plane of the objective lens. Scattered light from outside the focal plane is blocked by the pinhole, providing high contrast and depth resolution.
In the laser scanning type (HRT III-RCM), a 670 nm diode laser functions as a point light source, scanning point by point over the cornea to construct an image. The thickness of the focal plane (optical section thickness) is extremely thin, about 4 μm, providing clear cross-sectional images at the cellular level.
In the slit scanning type (Confoscan 4), scanning is performed with a slit-shaped light beam, so true confocality is obtained only in the direction perpendicular to the slit. Therefore, the depth resolution is 25–27 μm, and image sharpness is inferior to that of the laser scanning type.
Limitations of IVCM include: only grayscale images (no color observation), inability to resolve intracellular structures, difficulty in observing cases with severe corneal opacity, difficulty in accurately locating the observation site, and dependence on the examiner’s skill.
5. Main Clinical Applications
Section titled “5. Main Clinical Applications”The clinical applications of IVCM are diverse.
Diagnostic aid for corneal infections: Useful as an adjunct to culture tests. Fungal hyphae and Acanthamoeba cysts can be detected, but interpretation of findings requires specialized knowledge 8). In herpetic keratitis, changes in corneal nerves can be captured 8). In cytomegalovirus corneal endotheliitis, owl’s eye inclusion bodies may be observed.
Differentiation of corneal dystrophies: Each dystrophy has characteristic IVCM findings, allowing non-invasive differentiation by pattern recognition 1). It is highly useful as a screening tool prior to genetic testing or biopsy. It can also be applied to evaluate treatment outcomes (PTK, DALK, etc.) 1).
Evaluation of limbal stem cell deficiency (LSCD): Loss of the palisades of Vogt, replacement of corneal epithelium by conjunctival epithelium, and presence of goblet cells are diagnostic criteria 6). Measurement of basal epithelial cell density allows quantitative assessment of LSCD severity 6).
Evaluation of corneal edema and endothelial diseases: IVCM can observe endothelial cells even through moderate corneal edema, making it particularly useful in cases where specular microscopy is difficult to perform 7). In Fuchs endothelial corneal dystrophy (FECD), in addition to evaluating endothelial cells, decreased nerve density and changes in dendritic cell density can also be captured 7).
Evaluation of corneal nerves: Quantitative assessment of the subbasal nerve plexus is used for diagnosis and treatment monitoring of neurotrophic keratopathy 3). In the evaluation of nerve regeneration after surgical corneal neurotization, IVCM can confirm regenerative findings at 6 months to 1 year postoperatively 3).
Early detection of systemic autoimmune diseases: In Sjögren’s syndrome, decreased corneal nerve fiber density and increased activated dendritic cells in the central cornea may be detected before clinical onset 4). The presence of two or more activated dendritic cells with three or more processes in the central cornea has been reported to have 60% sensitivity and 77% specificity for systemic immune disease 4).
Postoperative evaluation: It is used to monitor flap edges, Bowman’s layer changes, and nerve regeneration after LASIK and PRK 2). It is also applied for early detection of rejection after corneal transplantation.
6. Latest Research and Future Perspectives
Section titled “6. Latest Research and Future Perspectives”Automated image analysis: Automated quantification systems for subbasal nerve fibers, such as ACCMetrics software, have been developed 2). They objectively calculate parameters such as nerve fiber density, branch density, and tortuosity, reducing inter-observer variability.
Detection of preclinical disease stages: In cases of Sjögren’s syndrome, IVCM abnormalities have been reported to be detected several years before antibody positivity or clinical symptom onset 4). This suggests that IVCM may serve as a screening tool for autoimmune diseases.
Subtype classification of dry eye: Analysis of dendritic cell morphology and distribution patterns may help differentiate immune-mediated dry eye from evaporative dry eye 4).
Monitoring of postoperative nerve regeneration: Research is progressing on longitudinal tracking of nerve regeneration after corneal neurotization and corneal cross-linking using IVCM 3). Future use as an objective endpoint for treatment efficacy is anticipated.
Integration of artificial intelligence: Development of automated classification and diagnosis systems for IVCM images using deep learning is underway, with expected applications in rapid diagnosis of infections and automated differentiation of corneal dystrophies.
7. References
Section titled “7. References”-
Ozturk HK, Ozates S, Ozkurt ZG, et al. In Vivo Confocal Microscopy in Avellino Corneal Dystrophy. Cureus. 2024;16(9):e68561.
-
Alvani A, Hashemi H, Pakravan M, et al. Corneal ectasia following PRK: a confocal microscopic case report. Arq Bras Oftalmol. 2024;87(6):e2023-0072.
-
Rathi A, Bothra N, Priyadarshini SR, et al. Neurotization of the human cornea - A comprehensive review and an interim report. Indian J Ophthalmol. 2022;70(6):1905-1917. doi:10.4103/ijo.IJO_2030_21. PMID:35647955; PMCID:PMC9359267.
-
Mercado CL, Galor A, Felix ER, et al. Confocal Microscopy Abnormalities Preceding Antibody Positivity and Manifestations of Sjogren’s Syndrome. Ocul Immunol Inflamm. 2023;31(5):1004-1009.
-
Austin A, Lietman T, Rose-Nussbaumer J. Update on the management of infectious keratitis. Ophthalmology. 2017;124(11):1678-1689. doi:10.1016/j.ophtha.2017.05.012. PMID:28942073; PMCID:PMC5710829.
-
Deng SX, Borderie V, Chan CC, et al. Global consensus on the definition, classification, diagnosis, and staging of limbal stem cell deficiency. Cornea. 2019;38(3):364-375.
-
Aggarwal S, Kheirkhah A, Cavalcanti BM, et al. In vivo confocal microscopy in Fuchs endothelial corneal dystrophy: a review. Eye Contact Lens. 2020;46(5):S46-S52.
-
日本眼感染症学会. 感染性角膜炎診療ガイドライン(第3版). 日眼会誌. 2023.
