Congenital Cataract
Key Points at a Glance
Section titled “Key Points at a Glance”1. What is Congenital Cataract?
Section titled “1. What is Congenital Cataract?”Congenital cataract, in a narrow sense, refers to lens opacity present at birth. In contrast, opacities that develop after birth without being present at birth are called developmental cataracts. Cataracts in infancy without causes such as trauma are often collectively referred to as congenital cataracts.
In terms of having a congenital predisposition, developmental cataracts that appear early after birth and progress belong to the same category. Clinically, they need to be managed similarly as “early-onset cataracts.” “Childhood cataract” is a comprehensive term that includes all cataracts during childhood, including traumatic cases, and encompasses congenital (narrow sense), developmental, and acquired cataracts.
Epidemiology
Section titled “Epidemiology”It is a rare disease occurring in 1 in 1,000 to 10,000 births. Visual prognosis and treatment strategies differ greatly between bilateral and unilateral cases, with unilateral cases having a worse visual prognosis (due to easier establishment of form deprivation amblyopia).
Persistent fetal vasculature (PFV) is an important cause of congenital cataract, often associated with unilateral involvement and microphthalmia. PFV is found in about 20% of infantile cataracts 1).
In a narrow sense, congenital cataract is lens opacity already present at birth, while developmental cataract appears after birth but shares a congenital predisposition. Clinically, both are managed similarly as “early-onset cataracts,” requiring early diagnosis and surgery to prevent form deprivation amblyopia.
2. Main Symptoms and Clinical Findings
Section titled “2. Main Symptoms and Clinical Findings”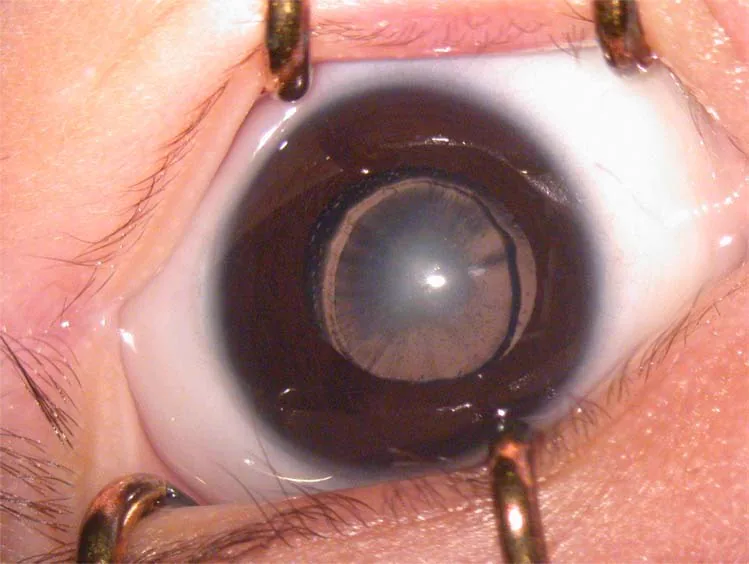
Subjective Symptoms and Clues for Detection
Section titled “Subjective Symptoms and Clues for Detection”Since infants cannot complain of symptoms, observation by parents and detection during infant health checkups are important.
- Leukocoria (white pupil): When opacity is severe, parents may notice that the eye appears white. This is one of the most common clues for detection.
- Strabismus: In unilateral cases, amblyopia may cause misalignment of the eyes.
- Nystagmus: A sign of advanced amblyopia in bilateral cases. When present, urgent surgery is required.
- Poor visual acuity / poor fixation: Suggests the development of form deprivation amblyopia.
Clinical Findings
Section titled “Clinical Findings”Indicators suggesting form deprivation amblyopia are shown below.
| Indicator | Description |
|---|---|
| Decreased visual acuity | Visual acuity reduction disproportionate to the degree of cataract |
| Poor fixation | Delayed development of fixation and pursuit responses |
| Strabismus | Ocular misalignment due to unilateral amblyopia |
| Nystagmus | Sign of progressive bilateral amblyopia |
| Axial length difference between eyes | Asymmetry of 0.5 mm or more |
As a criterion for determining whether opacity affects visual function, in unilateral cases, an opacity of 3 mm or more is considered to affect visual function. Generally, posterior capsule opacities affect visual function more than anterior capsule opacities, central opacities more than peripheral ones, and dense opacities more than faint ones.
It is often discovered when a parent notices a white pupil or during fundus examination at infant health checkups. Some cases are diagnosed after an ophthalmology visit prompted by strabismus or poor fixation. In unilateral cases, the healthy eye is predominantly used, making visual impairment less noticeable, so active screening during checkups is important.
3. Causes and Risk Factors
Section titled “3. Causes and Risk Factors”The etiology of congenital cataract is diverse, with idiopathic (unknown cause) accounting for 30–50%.
| Etiology | Frequency / Representative diseases |
|---|---|
| Idiopathic (unknown cause) | 30–50%. Most common |
| Hereditary | Autosomal dominant is most common. Also autosomal recessive and X-linked recessive |
| Intrauterine infection | Rubella (congenital rubella syndrome), herpes, CMV, toxoplasmosis, etc. |
| Metabolic disorders | Galactosemia, hypocalcemia, homocystinuria, Lowe syndrome, Alport syndrome, etc. |
| Chromosomal abnormalities | Down syndrome, etc. |
| Associated ocular diseases | Microphthalmia, congenital aniridia, PFV (persistent fetal vasculature), etc. |
| Systemic diseases/syndromes | Hallermann-Streiff syndrome, Pierre Robin syndrome, etc. |
PFV (persistent fetal vasculature) is an important cause of congenital cataract, resulting from failure of regression of the primary vitreous vessels during the fetal period. It is often unilateral and associated with microphthalmia, and is found in about 20% of infantile cataracts1). When it extends posteriorly (posterior type PFV), the visual prognosis is poor.
Hereditary congenital cataracts are most often autosomal dominant. Genetic counseling and family screening are recommended if there is a family history. It should be noted that 30–50% of congenital cataracts are idiopathic, so they can occur even without a family history.
4. Diagnosis and Examination Methods
Section titled “4. Diagnosis and Examination Methods”Morphological Classification
Section titled “Morphological Classification”Since treatment indications differ depending on the location and morphology of lens opacification, morphological classification is fundamental to diagnosis.
| Classification Name | English Name | Characteristics |
|---|---|---|
| Nuclear Cataract | nuclear cataract | Opacity inside the Y-suture. Congenital. Poor visual prognosis. |
| Lamellar Cataract | lamellar (zonular) cataract | Layered opacity peripheral to the Y-suture. Common in developmental cataracts. |
| Posterior Lenticonus | posterior lenticonus | Protrusion of the posterior capsule with opacity. Unilateral. May progress to total cataract. |
| Total cataract | total cataract | Complete opacity of the lens |
| Capsular cataract | capsular cataract | Opacity of the anterior capsule, posterior capsule, or subcapsular region |
| Sutural cataract | sutural cataract | Opacity at the Y-shaped suture. Little effect on visual function. |
| Punctate cataract | punctate cataract | Punctate opacities. Rarely cause visual impairment. |
| Cerulean cataract | cerulean cataract | Blue punctate opacities in the cortex. No visual impairment. |
Required Tests
Section titled “Required Tests”- Slit-lamp examination: Basic test to evaluate the morphology, location, severity, and extent of opacity.
- Fundus examination: Determines cataract opacity based on fundus visibility. If visibility is poor, add B-mode ultrasound.
- Axial length measurement: Essential for IOL power calculation, lens size prediction, and early detection of postoperative glaucoma (abnormal axial elongation).
- Refraction test: Use a handheld autorefractometer or retinoscope.
- Visual acuity assessment: Use PL method (preferential looking: evaluation of preferential visual response using grating acuity) and VEP (visual evoked potential). VEP values below 40% of normal indicate reduced visual function.
- Systemic examination: Systemic disease screening by a pediatrician is mandatory.
The indication for surgery is generally determined based on visual acuity (grating acuity) and the developmental state of visual responses such as fixation and pursuit.
Timing of Surgical Indication
Section titled “Timing of Surgical Indication”The following are guidelines for when active surgical indication is considered.
| Eye Type | Indication Criteria | Actual Surgical Timing Guideline |
|---|---|---|
| Unilateral (total cataract) | Treat as an emergency. | Preferably within a few days after birth. |
| Unilateral (severe opacity) | Severe opacity before 6 weeks of age | Around 1 month of age |
| Bilateral (severe opacity) | Severe opacity before 12 weeks (3 months) of age | Around 2 months of age |
| Forms suitable for observation | Punctate, sutural, anterior capsular cataract; symmetric lamellar or pulverulent nuclear cataract | Regular follow-up |
Unilateral total cataract is an emergency condition, and surgery within the first few days of life is desirable unless there are severe complications.
Unilateral total cataract is an emergency; surgery within the first few days of life is desirable to prevent form deprivation amblyopia. For bilateral cases, early surgery is performed around 2-3 months of age. Surgical indication is determined comprehensively based on visual acuity and development of visual responses such as fixation and pursuit. For forms that do not affect visual function, such as punctate cataract or sutural cataract, observation is prioritized.
5. Standard Treatment
Section titled “5. Standard Treatment”Surgical Methods
Section titled “Surgical Methods”The basic procedure for children under 6 years old is lens (phaco) aspiration + posterior capsulotomy + anterior vitrectomy (± IOL implantation). In children, to prevent posterior capsule opacification (visual axis opacification), posterior capsulotomy and anterior vitrectomy are standard, unlike in adults. After anterior capsulotomy with CCC, the lens cortex and nucleus are aspirated, then a posterior CCC is performed and anterior vitrectomy is carried out with a vitreous cutter.
The three main surgical procedures are described below.
| Procedure | Main Indications | Key Points |
|---|---|---|
| Lens aspiration + posterior capsulotomy + anterior vitrectomy | Basic procedure for children under 6 years old | Posterior capsulotomy and anterior vitrectomy are performed together to prevent posterior capsule opacification. |
| Lens aspiration + posterior capsulotomy + anterior vitrectomy + IOL implantation | When IOL implantation is performed | The ideal capsulotomy opening is about 5 mm. |
| Lens aspiration + IOL implantation (same as in adults) | Age 6 years and older | Posterior capsulotomy and anterior vitrectomy may be omitted. |
Considerations for IOL Indications
Section titled “Considerations for IOL Indications”Regarding the appropriateness of IOL implantation in infants, the Infant Aphakia Treatment Study (IATS) provides criteria. IOL implantation within the first 6 months of life showed no difference in visual acuity at 4.5 years compared to contact lens (CL) management, and the rate of reoperation for visual axis opacification was significantly higher in the IOL group. Therefore, IOL implantation within the first 6 months is limited to select cases. For children aged 2 years and older, primary IOL implantation is generally chosen.
Target IOL Power
Section titled “Target IOL Power”In children, the eyeball grows and becomes more myopic with age, so overcorrection anticipating future ocular growth is the basic approach.
| Age at Implantation | Postoperative Target Refraction | Purpose |
|---|---|---|
| 1 year old | Overcorrection of about +5 D | Focus on near vision to promote visual development |
| 2 years old | Overcorrection of about +4 D | Same as above |
| After growth (around age 20) | Mild myopia (about -1 to 0 D) | Aim for long-term refractive stability |
Acrylic material is desirable for both the haptic and optic parts of the IOL. An optic diameter of 6 mm or less is recommended.
Accuracy of IOL Power Calculation
Section titled “Accuracy of IOL Power Calculation”In primary IOL implantation in children, the predictive accuracy of current calculation formulas is significantly lower than in adults. In a study of 108 eyes of 83 patients, the prediction accuracy within ±1.00 D of eight formulas (Holladay 1, SRK/T, Hoffer Q, Haigis, Barrett Universal II, Kane, EVO 2.0, Ladas Super Formula) was only 35.2–43.5% (compared to 93.5–100% in adults) 2). The SRK/T and Kane formulas showed relatively better performance, and young age and short axial length were major predictors of refractive surprise (prediction error ≥ ±2.00 D) 2). There was no significant difference in prediction accuracy by surgical technique or IOL type 2).
Postoperative Refractive Correction
Section titled “Postoperative Refractive Correction”- Unilateral postoperative: CL correction is the standard. Soft CLs are often limited to a power of about +25 D; beyond that, hard CLs are used.
- Bilateral postoperative: Spectacle management is possible. In infancy, special frames are used.
- Even in eyes with IOL implantation, spectacles are needed if residual hyperopia remains.
- After school age, bifocal spectacles may be considered.
Amblyopia Treatment
Section titled “Amblyopia Treatment”In unilateral cases and bilateral cases with interocular difference, occlusion of the better eye is necessary. According to the IATS occlusion protocol, up to 8 months of age, occlusion time equals the age in months (e.g., 4-month-old → 4 hours); after 8 months, occlusion is aimed at half of waking hours.
Surgical Outcomes of Congenital Cataract with PFV
Section titled “Surgical Outcomes of Congenital Cataract with PFV”In a multicenter registry study by the Pediatric Eye Disease Investigator Group (PEDIG), 5-year outcomes of 64 eyes with PFV (48 aphakic, 16 pseudophakic) were reported 1). The median age at surgery for aphakic eyes was 2 months, and 96% underwent anterior vitrectomy. Only 10% (4/42 eyes, 95% CI 3–23%) achieved age-appropriate visual acuity at 5 years. Visual acuity better than 20/200 was achieved in 59% (17/29 eyes) of aphakic PFV eyes, compared to only 23% (3/13 eyes) of pseudophakic PFV eyes 1). Posterior PFV (with vitreous, retinal, or optic nerve abnormalities) tended to have worse visual prognosis than anterior PFV (median visual acuity 20/800 vs. 20/100) 1).
Based on the results of the IATS, IOL implantation within 6 months of age shows no difference in visual acuity compared to contact lens management but has a higher rate of visual axis opacification requiring reoperation. Therefore, IOL implantation during this period is limited to select cases. For children aged 2 years and older, primary IOL implantation is generally chosen. The IOL power is selected with the goal of overcorrection in anticipation of myopic shift with growth.
6. Pathophysiology and Detailed Mechanisms
Section titled “6. Pathophysiology and Detailed Mechanisms”Mechanism of Form Deprivation Amblyopia
Section titled “Mechanism of Form Deprivation Amblyopia”Lens opacities cause form deprivation amblyopia by blocking light along the visual axis. When appropriate visual stimulation is blocked during the critical period of visual development, the development of the visual cortex is impaired.
The critical period extends up to 6 weeks after birth for monocular cases and up to 10–12 weeks for binocular cases. If a cataract affecting visual function is present before the critical period, amblyopia develops particularly rapidly in monocular cases. Visual function acquisition begins during the critical period and is completed around 10 years of age.
Form deprivation amblyopia is more severe and more resistant to treatment than ordinary refractive amblyopia. This is the rationale for the necessity of early surgery.
Mechanism of After-Cataract (Posterior Capsule Opacification)
Section titled “Mechanism of After-Cataract (Posterior Capsule Opacification)”In children, lens epithelial cells are highly active, and posterior capsule opacification (after-cataract) occurs more frequently and earlier after surgery compared to adults. Residual lens epithelial cells proliferate postoperatively, migrate onto the posterior capsule, and block the visual axis. Therefore, in children under 6 years of age, the standard surgical technique includes intraoperative posterior capsulotomy and anterior vitrectomy to prevent the development of after-cataract.
Differences in Opacity Morphology and Impact on Visual Function
Section titled “Differences in Opacity Morphology and Impact on Visual Function”The impact on visual function and the indication for surgery differ depending on the location and morphology of the lens opacity. Nuclear cataracts produce dense opacities directly in the visual axis, leading to poor visual prognosis and requiring early surgery. Sutural and punctate cataracts are less likely to affect visual development and can often be observed. Posterior lenticonus is often unilateral, with protrusion of the posterior capsule and progressive opacification that may eventually lead to total cataract.
Lens Development and Causes of Opacification
Section titled “Lens Development and Causes of Opacification”The occurrence of congenital cataracts is based on abnormalities in the embryonic development of the lens. The lens forms from the lens vesicle derived from the ectoderm; the nucleus is composed of primary lens fibers (the basis of nuclear cataracts) and the cortex of secondary lens fibers (the basis of lamellar and sutural cataracts). Intrauterine infections such as rubella virus directly impair fetal lens fiber formation. In metabolic disorders (e.g., galactosemia), accumulated galactitol increases osmotic pressure within the lens, causing lens overhydration and opacification. In hereditary cataracts, functional abnormalities of crystallin proteins or membrane proteins (e.g., connexins) impair the maintenance of lens transparency.
7. Postoperative Complications and Long-Term Prognosis
Section titled “7. Postoperative Complications and Long-Term Prognosis”After-Cataract (Visual Axis Opacification)
Section titled “After-Cataract (Visual Axis Opacification)”In cases operated on during infancy, contraction of the anterior capsulotomy margin and Elschnig pearl-type opacities occur in nearly 100% of cases. Treatment is as follows.
- Age 1 year or older: Nd:YAG laser posterior capsulotomy is possible
- If laser is difficult in children under 6 years: perform surgical posterior capsulectomy and anterior vitrectomy
The 5-year cumulative incidence of visual axis opacification in pseudophakic eyes with PFV was 45% (95% CI 13–66%) 1). The incidence of visual axis opacification when anterior vitrectomy was performed at the primary surgery was 18% (95% CI 8–28%), which was significantly lower than 60% (95% CI 0–84%) when not performed 1).
Secondary Glaucoma
Section titled “Secondary Glaucoma”This is the most important late complication after congenital cataract surgery.
- Incidence: occurs in 18–26% of postoperative cases
- Risk factors: younger age at surgery, microphthalmos
- Onset: can occur more than 10 years after surgery. Long-term follow-up is essential.
The 5-year cumulative incidence of glaucoma-related adverse events in aphakic eyes with PFV was 24% (95% CI 9–37%), and in pseudophakic eyes it was 7% (95% CI 0–20%) 1). There was no significant difference in glaucoma risk between PFV and non-PFV eyes (age-adjusted HR=1.20, 95% CI 0.54–2.64, P=.66), and PFV itself was not an independent risk factor for glaucoma 1).
5-Year Cumulative Incidence of Complications in Congenital Cataract with PFV
Section titled “5-Year Cumulative Incidence of Complications in Congenital Cataract with PFV”| Complication | PFV Aphakic Eye | PFV Pseudophakic Eye |
|---|---|---|
| Glaucoma-related adverse events | 24% (95% CI 9–37%) | 7% (95% CI 0–20%) |
| Visual axis opacification (with anterior vitrectomy) | 18% (95% CI 8–28%) | — |
| Visual axis opacification (without anterior vitrectomy) | 60% (95% CI 0–84%) | 45% (95% CI 13–66%) |
| Retinal detachment | 4% (95% CI 0–10%) | 7% (95% CI 0–19%) |
Other complications
Section titled “Other complications”- Anterior capsule contraction: The anterior capsulotomy edge contracts, reducing the capsulotomy opening. Progression can cause visual impairment.
- Retinal detachment: Risk is particularly high in cases with PFV.
Factors affecting visual prognosis
Section titled “Factors affecting visual prognosis”- Degree of form deprivation amblyopia (presence of early surgery is most important)
- Presence of ocular comorbidities (microcornea and microphthalmos indicate poor prognosis)
- Appropriate refractive correction and continuation of amblyopia treatment after surgery
- Understanding and cooperation of parents/guardians
Secondary glaucoma is the most important complication. It occurs in 18–26% of cases after surgery and can develop even 10 years or more postoperatively, so regular intraocular pressure checks are necessary even in adulthood. Posterior capsule opacification (visual axis opacification) occurs in nearly all cases of infant surgery, but YAG laser treatment is possible for children aged 1 year or older, and invasive procedures may be required for those under 6 years of age.
8. Latest Research and Future Prospects
Section titled “8. Latest Research and Future Prospects”Improvement of Accuracy in Pediatric IOL Power Calculation Formulas
Section titled “Improvement of Accuracy in Pediatric IOL Power Calculation Formulas”The predictive accuracy of current pediatric IOL power calculation formulas is insufficient compared to adults, and even new-generation formulas (e.g., Kane, Barrett Universal II) have not shown clear superiority over conventional formulas (e.g., SRK/T) 2). The prediction accuracy within ±1.00D is only 35.2–43.5% for all eight current formulas, a significant gap from the 93.5–100% in adults 2).
There is a need to develop dedicated algorithms that account for the short axial length, steep corneal curvature, shallow anterior chamber depth, and ocular growth specific to children 2). There is no significant difference in prediction accuracy by surgical technique or IOL type, and at present, active postoperative refractive correction and follow-up are important complementary measures.
9. References
Section titled “9. References”- Haider KM, Repka MX, Sutherland DR, et al. Outcomes and Complications 5 Years After Surgery for Pediatric Cataract Associated With Persistent Fetal Vasculature. Am J Ophthalmol. 2024;261:156-165.
- Jin J, Shen Y, Qu Y, et al. Accuracy of new-generation and traditional intraocular lens power calculation formulas in pediatric primary implantation. J Cataract Refract Surg. 2024.
- Kaur S, Yangzes S, Ram J. Unilateral Congenital Cataract. J Pediatr Ophthalmol Strabismus. 2019;56(1):60-61. PMID: 30673102.
