Growth Hormone
Frequency: Required in about 70% of ONH patients.
Indication: Initiate when growth hormone deficiency is confirmed.

Optic nerve hypoplasia (ONH) is the most common congenital optic nerve abnormality, characterized by a reduced number of optic nerve axons. It can occur unilaterally or bilaterally and may be associated with midline brain structural defects.
Briere first described it histologically in 1877, and Reeves provided a clinical description in 1941. In 1956, de Morsier reported its association with absence of the septum pellucidum, leading to the term de Morsier syndrome (septo-optic dysplasia, SOD). In 1970, Hoyt et al. reported detailed clinical features, increasing awareness of this condition.
SOD is diagnosed when two or more of the following three features are present3)6):
The prevalence of SOD is estimated at approximately 1 in 10,000 births2)6). Epidemiologically, it is the third most common cause of visual impairment in children under 3 years of age. Reported rates are 10.9 per 100,000 population in England and 17.3 in Sweden.
In a study of 16 cases at Niigata University in Japan, the median age at first visit was 2.4 years, 12/16 (75%) were female, and 11/16 (69%) were bilateral 1).
A severe form is optic nerve aplasia. The optic disc and retinal vessels are completely absent, and there is no light perception.
Superior segmental optic hypoplasia (SSOH) is a special type in which only the superior optic nerve fibers are hypoplastic, and an association with maternal diabetes has been noted. The prevalence in Japan is reported to be about 0.3%. There is no sex difference.
ONH refers to a morphological abnormality of the optic nerve alone. SOD is a syndrome that meets two or more of the triad of ONH, pituitary dysfunction, and midline brain structural abnormalities; ONH is one component of SOD. It has been reported that approximately 37.5% of ONH patients meet the diagnostic criteria for SOD 1).
Visual acuity in ONH ranges from normal to no light perception. Many cases have acuity of 0.1 or less, and visual acuity depends on the density of the papillomacular bundle. Optic nerve hypoplasia differs from other congenital disc anomalies in this respect: even if the macula is formed, the degree of development of the papillomacular nerve fiber bundle varies, so visual acuity ranges from 1.0 to extremely low.
In Japanese data, poor vision was found in 11/16 cases (69%), strabismus in 8/16 cases (50%), and nystagmus in 5/16 cases (31%)1).
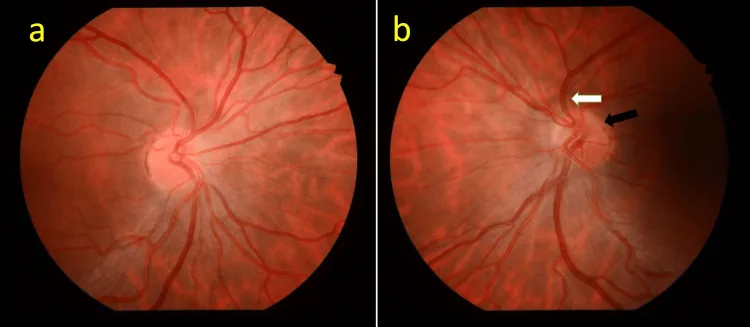
Characteristic findings are observed on ophthalmoscopy.
The frequencies of major ocular findings and systemic complications are shown below.
| Finding | Frequency |
|---|---|
| Structural CNS abnormalities | Approximately 90% |
| Neurodevelopmental disorders | Approximately 70% |
| Hypothalamic dysfunction (bilateral) | 81% |
| Hypothalamic dysfunction (unilateral) | 69% |
| Developmental delay (bilateral) | 78% |
Even in unilateral cases, hypothalamic dysfunction is found in 69%, and brain abnormalities exist in 18.2% of asymptomatic patients1). MRI and endocrine screening are essential even for unilateral cases.
The pathology of ONH is a developmental failure of retinal ganglion cells (RGC) and nerve fibers. There are two hypotheses: a developmental anomaly theory and a retrograde degeneration theory. A hypothesis that ischemia of the optic chiasm and optic nerve due to vascular disorders of the anterior cerebral artery is the cause has also been proposed.
Most cases are sporadic2)3).
The following gene mutations may be involved.
The diagnosis of ONH is based on ophthalmoscopic findings, combined with imaging and endocrine screening.
Confirmation of the double ring sign is the first step in diagnosis. A DM/DD ratio of 3 or more (3.2 or more indicates a small optic disc) is used as a guideline.
In a study of Japanese patients, brain abnormalities were found in 43.8% and SOD in 37.5%. Notably, brain abnormalities were present in 18.2% of asymptomatic patients 1). Furthermore, in 2 of 3 patients with pituitary dysfunction, MRI showed normal pituitary morphology 1).
The following tests are recommended for all ONH patients:
Differentiation from the following diseases is necessary:
Yes. In a study of Japanese patients, 2 out of 3 patients with hypopituitarism had normal MRI findings1). Endocrine screening should be performed in all patients regardless of MRI results.
There is no curative treatment for ONH itself. Management focuses on optimizing visual function and addressing systemic complications (especially endocrine abnormalities). Collaboration among a multidisciplinary team (ophthalmology, endocrinology, pediatrics, neurology, rehabilitation) is essential. Growth should be evaluated every six months, and visual function annually.
Growth Hormone
Frequency: Required in about 70% of ONH patients.
Indication: Initiate when growth hormone deficiency is confirmed.
Thyroid Hormone
Frequency: Required in about 43%.
Indication: Initiate replacement when TSH/FT4 are abnormal.
Adrenal Corticosteroids
Frequency: Required in approximately 27% of cases.
Caution: Adrenal insufficiency can be fatal during stress. Instruction on stress dosing (increasing dose during fever or surgery) is essential5).
Antidiuretic Hormone
Frequency: Diabetes insipidus occurs in about 5% of cases.
Caution: Rapid correction of sodium can cause seizures. The correction rate should be less than 0.5 mEq/L/hour4).
In adult management cases, replacement regimens such as levothyroxine 137 μg, desmopressin, and hydrocortisone 10 mg (morning)/7.5 mg (evening) have been reported2).
Optic nerve hypoplasia itself is non-progressive unless glaucoma develops. In cases without glaucoma, unnecessary eye drops or surgical treatment to lower intraocular pressure should be avoided. However, endocrine abnormalities may appear or worsen over time, making long-term follow-up essential1). Early diagnosis and initiation of hormone replacement therapy by age 3 in necessary cases can prevent sequelae, so it is important to keep this disease in mind even in unilateral cases and not miss it.
Optic nerve hypoplasia itself is non-progressive, and vision is often stable unless glaucoma develops. However, endocrine abnormalities may appear later in life, so continued periodic systemic evaluation is important.
The essence of ONH is a reduction in the retinal nerve fiber layer (RNFL) and ganglion cells, with little effect on the outer retinal layers. There are two main hypotheses for the mechanism of development.
There is also a hypothesis that ischemia in the optic chiasm and optic nerve region due to vascular disorders of the anterior cerebral artery may be involved.
Hypothalamic dysfunction is observed in 69% of unilateral ONH and 81% of bilateral ONH. The pituitary gland and optic nerve are developmentally close, and the same developmental disorder is thought to affect both.
Developmental delay is observed in 75% of all cases, with a higher rate in bilateral (78%) than unilateral (39%) cases.
SOD plus is a condition that includes cortical malformations (e.g., polymicrogyria, schizencephaly) in addition to classic SOD, and has been reported to occur more frequently than classic SOD 7). Neurodevelopmental prognosis is poorer, and the risk of epilepsy is higher.
A clinical trial (NCT06760546) of setmelanotide, a melanocortin 4 receptor (MC4R) agonist, is ongoing as a drug therapy for obesity associated with SOD 2). Hypothalamic obesity significantly reduces the QOL of SOD patients, so this is attracting attention as a new treatment option.
In SOD neonates diagnosed with normoglycemia, testosterone therapy (25 mg intramuscular injection, once monthly for 3 months) for management of micropenis and cryptorchidism, as well as trials of recombinant FSH therapy, have been reported 5). These indicate the potential for early endocrine intervention, but long-term efficacy and safety have not yet been established.