Cribriform Type
Frequency: Most common (39.8%)
Features: Lobular structures with circular pools of mucin. “Swiss cheese” appearance. Intermediate prognosis.
Adenoid cystic carcinoma (ACC) is a rare malignant tumor arising in secretory glands, with a worldwide incidence of 3–4 cases per million population 1). It accounts for about 1% of all head and neck cancers, and when it occurs in the lacrimal gland, it is called lacrimal gland adenoid cystic carcinoma 1).
Lacrimal gland adenoid cystic carcinoma accounts for approximately 1.6% of all orbital tumors 3)5) and is the most common malignant epithelial tumor of the lacrimal gland, constituting about 13–40% of lacrimal gland carcinomas 5). Lacrimal gland tumors account for about 10% of orbital space-occupying lesions; about 20% of solid lacrimal gland tumors are epithelial-derived, of which about 45% are malignant, and about 60% of malignant epithelial lacrimal gland tumors are adenoid cystic carcinoma.
Lacrimal gland adenoid cystic carcinoma was first described by Theodor Billroth and was initially named “cylindroma” based on its histological features 5).
Epidemiological features are as follows.
It accounts for approximately 1.6% of all orbital tumors, and the global incidence of adenoid cystic carcinoma is extremely rare at 3–4 cases per million population1)5). It is the most common histological type among malignant epithelial tumors of the lacrimal gland.
According to a large review of 806 cases, the frequency of initial symptoms is as follows5).
| Symptom | Frequency |
|---|---|
| Proptosis | 27.4% |
| Pain | 21.7% |
| Eyelid swelling | 10.9% |
| Eye deviation | 10.2% |
| Restricted eye movement | 10.1% |
| Decreased vision | 9.3% |
| Double vision | 6.7% |
| Ptosis | 4.1% |
Pain is a characteristic symptom of adenoid cystic carcinoma, caused by perineural invasion (PNI). It is an important distinguishing feature from benign tumors. Since the lacrimal gland is located in the superolateral orbit, the eye often deviates inferomedially. Hypesthesia in the frontotemporal region or S-shaped ptosis may also occur. The time from symptom onset to diagnosis averages 11.1 ± 18.3 months (range 0.5–120 months) 5).
The growth rate of the tumor is faster than that of pleomorphic adenoma, and it is more likely to be accompanied by pain, which is an important point for differentiation. Proptosis due to tumor enlargement, restriction of eye movement due to invasion of extraocular muscles and nerves, and decreased visual acuity due to optic nerve compression also progress relatively rapidly.
The presence or absence of pain is the most important differentiating point. Adenoid cystic carcinoma is accompanied by pain due to perineural invasion, whereas benign tumors (such as pleomorphic adenoma) are painless and progress slowly. If bone destruction is confirmed on CT, it can be judged as malignant, but there are also cases of adenoid cystic carcinoma without bone destruction, so if there is pain, actively consider biopsy.
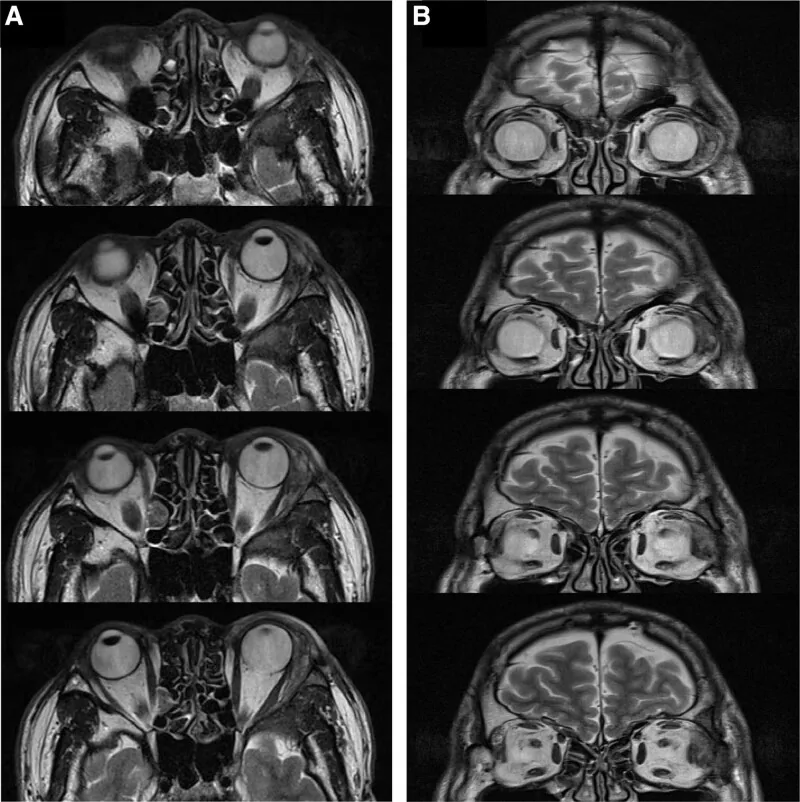
According to a report by Williams et al., histological evidence of lacrimal fossa invasion was confirmed in 82% of patients 1). If the tumor destroys the orbital bone on CT, it can be judged as malignant, but caution is needed because there are also cases of adenoid cystic carcinoma without bone destruction.
The cause of adenoid cystic carcinoma is unknown, and no specific risk factors have been established. The tumor most commonly arises from the orbital lobe of the lacrimal gland and is an unencapsulated tumor.
The following pathological and clinical features are known as poor prognostic factors.
Definitive diagnosis is based on histopathological evaluation. If malignancy is suspected, excisional biopsy is recommended to prevent intraorbital seeding of tumor cells. Fine-needle aspiration biopsy may be appropriate for unresectable tumors, but only at facilities with experienced cytopathologists.
If a lacrimal gland tumor is suspected due to symptoms such as proptosis, ophthalmic examinations including visual acuity, visual field, and eye movement are performed, along with imaging studies using plain CT and plain/contrast MRI. If systemic evaluation is needed, PET-CT or contrast CT may also be performed. Definitive diagnosis is made by histopathological examination of a biopsy or the entire resected specimen.
| T stage | Definition |
|---|---|
| T1 | Maximum diameter ≤ 2 cm |
| T2 | Maximum diameter > 2 cm to ≤ 4 cm |
| T3 | >4 cm in greatest dimension or orbital soft tissue extension |
| T4 | Invasion of paranasal sinuses, temporal fossa, pterygopalatine fossa, superior orbital fissure, cavernous sinus, or brain |
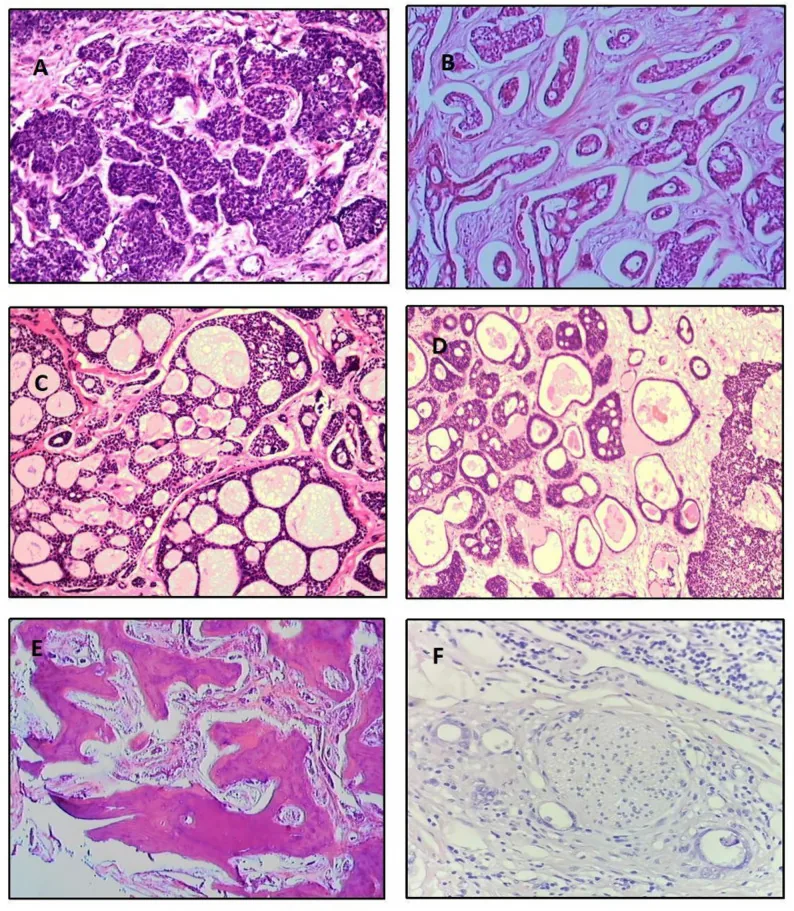
The frequency of histologic subtypes based on an analysis of 515 cases is as follows5).
Cribriform Type
Frequency: Most common (39.8%)
Features: Lobular structures with circular pools of mucin. “Swiss cheese” appearance. Intermediate prognosis.
Basaloid Type
Frequency: 31.8%
Features: Poorly differentiated. Large basophilic nuclei and scant cytoplasm. Worst prognosis.
Tubular Type
Frequency: 7.4%
Features: Epithelial tubules lined by 2–3 layers of cells. Most differentiated, best prognosis.
Other types include mixed (13.9%), undifferentiated (6.1%), and sclerosing (0.9%). A solid pattern exceeding 30% is considered poor prognosis1). Histologically, tumor cells are small with scant, basophilic cytoplasm and chromatin-rich nuclei. The border between tumor nests and stroma is distinct, clearly different from pleomorphic adenoma.
Surgery is the mainstay of treatment, and the surgical approach is determined by tumor stage and imaging findings3)5).
Kaplan-Meier analysis showed that eye-sparing surgery plus radiotherapy had better survival than orbital exenteration with or without radiotherapy (P<0.05)5).
If imaging shows a small tumor that can be completely resected, aim for complete resection. If complete resection is deemed impossible, perform an incisional biopsy to confirm pathology, then consider wide excision or radiotherapy. For adenoid cystic carcinoma of the lacrimal gland confined to the orbit, orbital exenteration may be considered, but conservative treatment may be chosen based on cosmetic concerns, patient age, and preferences. Lymph node metastasis is rare (4–9%), and lymph node dissection is usually unnecessary6).
Even after wide excision and radiation therapy, invasion into the brainstem through the lacrimal nerve may not be prevented. It is not always possible to prevent distant metastasis, and long-term follow-up is necessary.
For unresectable adenoid cystic carcinoma, heavy particle therapy is performed and is positioned as a promising treatment that can control the tumor while preserving the eyelid, eyeball, and orbit.
NIAC was first reported by Meldrum et al. in 1998, using a combination of intra-arterial cisplatin (100 mg/m²) and intravenous doxorubicin3)2).
In a study of 19 cases by Tse et al. (2013), a 10-year disease-free survival rate of 100% was reported in 8 cases where the lacrimal artery was preserved and the protocol was followed2)3).
The combination of NIAC + resection/orbital exenteration + radiation therapy shows favorable outcomes compared to other treatments, with recurrence rate 10.8%, metastasis rate 14.9%, and mortality rate 18.9%5). Major risks of NIAC include transient facial nerve palsy, vision loss, anterior ischemia, neutropenia, and thrombocytopenia3).
Kaplan-Meier analysis showed that survival rates for eye-sparing surgery plus radiotherapy were better than for orbital exenteration with or without radiotherapy (P<0.05)5). Eye-sparing surgery is recommended for T1–T2 tumors, while orbital exenteration is considered for T3–T4 tumors or cases with extraorbital extension. However, conservative treatment may be chosen due to cosmetic concerns or patient preference.
Tumor cells are small with scant, bluish cytoplasm and chromatin-rich nuclei. Histologically, the boundary between tumor nests and stroma is clear, distinctly different from pleomorphic adenoma.
In the cribriform type, true lumens (derived from ductal cells) and pseudolumens (mucin-filled spaces formed by myoepithelial cells) coexist, giving a “Swiss cheese” appearance. The sclerotic type appears as epithelial cords with dense hyalinized stroma. A solid pattern exceeding 30% is associated with poor prognosis1).
Tumor growth patterns are classified into five types: cribriform, solid, sclerotic, comedocarcinomatous, and tubular. Multiple growth patterns can be observed within the same specimen.
| Molecular abnormality | Description |
|---|---|
| MYB-NFIB fusion | t(6;9)(q23;p23) translocation. Present in over 70% of all adenoid cystic carcinomas1) |
| MYB overexpression | Promotes cell proliferation, differentiation, angiogenesis, and upregulation of growth factors1) |
| NOTCH1 activating mutation | Major driver of proliferation and invasion in metastatic adenoid cystic carcinoma1)2) |
| KRAS/NRAS/MET mutations | Reported in 46%, 8%, and 13% of cases, respectively. The EGFR-RAS-RAF cascade may be a therapeutic target1)5) |
In lacrimal gland adenoid cystic carcinoma, MYB rearrangement is detected in 58% of cases (Mayo Clinic, 12 cases/25 years), and MYB-NFIB fusion is a highly specific diagnostic marker for adenoid cystic carcinoma1). MYB-NFIB fusion is regulated by AKT-dependent IGF1R signaling, and IGF1R inhibition is considered a promising therapeutic target1)3).
Perineural invasion is observed in 45.3% of cases, and tumor cells can spread even without vascular or lymphatic invasion1). There is a strong tendency to invade nerves and lymphatics, and infiltration to the brainstem through the lacrimal nerve can occur early in the disease. Perineural invasion is associated with apoptosis resistance due to Bcl-2 upregulation.
Currently, no molecular targeted drugs are approved for lacrimal gland adenoid cystic carcinoma. The following targets are being investigated in research stages1)3).
Results from phase II clinical trials have reported dovitinib (ORR 6%, mPFS 8.2 months), lenvatinib (ORR 16%, mPFS 17.5 months), and axitinib (ORR 9%, mPFS 5.7 months)3).
Yu et al. (2022) reported an assessment combining pre- and post-treatment genomic sequencing with apoptosis markers (cCas3, PARP) analysis2). The variant allele frequency (VAF) of NOTCH1 mutation after NIAC decreased from 18.07% before treatment to 11.34% after treatment (37% reduction), suggesting it may be a predictive marker for cisplatin sensitivity.
Currently, no molecular targeted drugs are approved for lacrimal gland adenoid cystic carcinoma. MYB-NFIB fusion, Notch signaling pathway, EGFR-RAS-RAF cascade, and others are being investigated as therapeutic targets in research, and several phase II clinical trials are ongoing1)3). For those seeking standard treatment, the combination of surgery and radiation therapy is the current option.