Subjective Symptoms
Vision loss: Poor vision even without DME.
Blurred vision/central scotoma: Functional decline due to macular ischemia.
Visual field defect: Localized defect corresponding to ischemic area.

Diabetic macular ischemia (DMI) is a condition in diabetic patients where retinal capillaries in the macula become occluded, atrophied, and lost, with narrowing and occlusion of precapillary arterioles. It is visualized as enlargement and irregularity of the foveal avascular zone (FAZ) and expansion of discontinuous avascular areas in the macula on fluorescein angiography (FA) or optical coherence tomography angiography (OCTA).
Historically, Norman Ashton first described in detail the involvement of arteries and capillaries in diabetic retinopathy through postmortem studies using peripheral iris synechiae staining and India ink injection. Pathologically, the process involves hyaline degeneration of terminal arterioles and precapillary vessels → luminal occlusion → loss of arteriolar and capillary beds → formation of venous neovascularization.
Diabetic maculopathy is classified into three types: macular edema, ischemic maculopathy, and retinal pigment epitheliopathy; DMI corresponds to ischemic maculopathy.
Prevalence is closely related to the severity of diabetic retinopathy.
| DMI Severity | Incidence |
|---|---|
| None | 39.7% |
| Suspected | 18.4% |
| Mild | 25.2% |
| Moderate | 11.0% |
| Severe | 5.6% |
DMI occurs in 29.4% of CSME (clinically significant macular edema) cases, of which 19.4% are moderate to severe. DMI is also found in 77.2% of proliferative diabetic retinopathy (PDR) and 59.7% of severe non-proliferative diabetic retinopathy (NPDR) cases.
DMI findings are sometimes characterized as a “featureless retina.” The foveal reflex becomes poor, and hemorrhages, microaneurysms, exudates, soft exudates, and neovascularization disappear or become mild.
Subjective Symptoms
Vision loss: Poor vision even without DME.
Blurred vision/central scotoma: Functional decline due to macular ischemia.
Visual field defect: Localized defect corresponding to ischemic area.
Clinical Findings
Featureless retina: Poor foveal reflex, disappearance or reduction of hemorrhages and exudates.
Ghost vessels: Residual shadows of capillaries that have lost perfusion.
Arteriolar narrowing: In moderate to severe DMI, mean arteriolar diameter is narrowed.
Functional Tests
Microperimetry: Retinal sensitivity is markedly reduced in DCP non-perfusion areas. 1)
AO-OCT: In DCP non-perfusion areas, photoreceptor signal density (IS/OS, COST) is reduced by approximately 40%. 1)
Regarding the correlation between DMI severity and visual acuity, significant visual acuity reduction is observed in moderate to severe DMI. The VA-FAZ correlation has been reported as R² = 0.41–0.51, and papillo-macular ischemia is independently associated with visual acuity reduction.
Datlinger et al. (2021) used AO-OCT and microperimetry to show that in DCP non-perfusion areas, photoreceptor signal density (IS/OS and COST) decreased by approximately 40%, and retinal sensitivity in the same area was also markedly reduced. 1)
Mild DMI may not have a significant impact on visual acuity. In moderate to severe cases, a significant correlation with visual acuity reduction is observed (VA-FAZ correlation R² = 0.41–0.51), and papillo-macular ischemia is also independently associated with visual acuity reduction. If visual acuity is poor despite the absence of DME, evaluation of DMI using tests described in the “Diagnosis and Testing Methods” section is important.
Risk factors for DMI reflect those for diabetic retinopathy in general.
The presence of DMI is associated with increased severity of DME and DR. Medical management (control of blood glucose, blood pressure, and lipids) is effective in slowing the progression of retinopathy.
Notably, spontaneous reperfusion of capillary non-perfusion areas (NPA) has been reported in a case with improved glycemic control (HbA1c from 6.1% to 5.6%). 2)
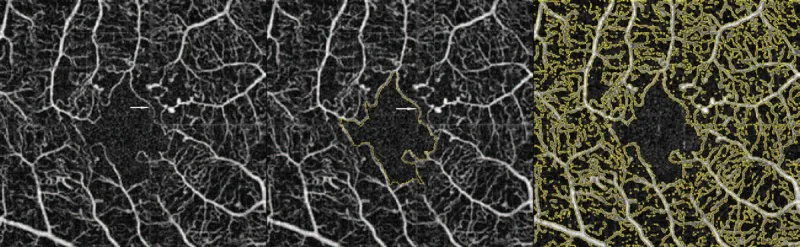
FA is the gold standard for diagnosing DMI.
Disadvantages of FA include invasiveness (intravenous contrast injection), risk of complications (mortality risk about 1 in 200,000), and a procedure time of 20 minutes or more.
OCTA is a non-invasive, high-resolution examination that allows layer-specific analysis of the SCP (superficial capillary plexus), DCP (deep capillary plexus), and CC (choriocapillaris).
A comparison of the characteristics of FA and OCTA is shown below.
| Feature | FA | OCTA |
|---|---|---|
| Invasiveness | Yes | No |
| Layer-specific analysis | Not possible | Possible |
| NPA detection range | Limited | Extensive |
FA
Gold standard: Visualizes FAZ enlargement and capillary dropout.
Invasive: Requires intravenous contrast injection. Mortality risk approximately 1 in 200,000.
Time required: Takes 20 minutes or more.
OCTA
Non-invasive, high resolution: Enables layer-specific analysis of SCP/DCP/CC.
NPA quantification: Correlates with DR severity. Also detects preclinical changes. 3)
Two types of NPA detection: Detection range differs from FA depending on the presence or absence of thinning. 2)
FA is the gold standard but invasive. OCTA is non-invasive and has the advantage of detecting more extensive NPA than FA. 2) In particular, early NPA without retinal thinning (NPA without thinning) can be detected only by OCTA. The characteristics of both are complementary, and it is desirable to use them appropriately depending on the clinical situation.
Currently, there is no established specific treatment for DMI. There is no direct treatment for macular ischemia without DME, and systemic management is fundamental.
Good glycemic control has been reported to potentially contribute to natural reperfusion of NPAs, 2) and medical management (blood glucose, blood pressure, lipids) is the basic strategy for suppressing retinopathy progression.
In stages beyond severe NPDR, consideration of PRP (panretinal photocoagulation) or anti-VEGF therapy is recommended. 3)
When DMI is complicated by DME, the following treatments are performed.
Currently, there is no established specific treatment for DMI. Systemic management (blood glucose, blood pressure, lipids) is fundamental, and there are case reports of natural reperfusion of NPAs with good glycemic control. 2) When DME is present, edema is treated with anti-VEGF drugs, etc., but ischemia itself cannot be directly improved.
Pericyte loss and endothelial cell damage are the earliest signs of vascular changes in DR.
Progressive thickening of the capillary basement membrane due to deposition of type III and IV collagen leads to luminal narrowing. Leukostasis also occurs, and abnormal endothelial cells exacerbate vascular occlusion.
Reduced oxygen and micronutrient supply stimulates VEGF expression, leading to progressive capillary atrophy → widening of intercapillary spaces → prolonged hypoxia → photoreceptor damage. As DR progresses, venous abnormalities, IRMA, severe hemorrhage, and exudation appear. 3)
The macula has three retinal vascular layers (SCP, ICP/MCP, DCP). The DCP contributes 10–15% of oxygen supply to photoreceptors, 1) and its occlusion directly leads to photoreceptor damage.
Müller cells (MC) function as an energy source by supplying lactate to photoreceptors under perfusion impairment. 1) When MC damage occurs, in addition to cone, rod, and MC dysfunction, patchy loss of photoreceptor outer segments and capillary dropout are colocalized. 1)
Ischemic patterns are classified into the following four types (Takashi et al. classification).
| Ischemia type | Frequency |
|---|---|
| Peripheral type | 2.6% |
| Mid-peripheral type | 61.2% |
| Central type | 26.3% |
| Extensive type | 9.9% |
Non-perfusion of the deep capillary plexus (DCP) reduces oxygen supply to photoreceptors (DCP contributes 10–15% of photoreceptor oxygen supply). 1) Müller cell damage also impairs energy supply to photoreceptors. Studies using AO-OCT have confirmed that photoreceptor signal density (IS/OS and COST) decreases by approximately 40% in areas of DCP non-perfusion. 1)
Datlinger et al. (2021) demonstrated that the combination of AO-OCT and OCTA enables DMI assessment at the single cone photoreceptor level. They stated that an integrated research approach combining microperimetry and OCTA contributes to understanding the time course of DMI, and suggested that these advanced imaging parameters may serve as biomarkers in future therapeutic studies. 1)
Conventionally, capillary occlusion was considered irreversible, but cases of spontaneous reperfusion have been reported.
Hou et al. (2022) observed spontaneous reperfusion of NPAs in diabetic patients whose HbA1c improved from 6.1% to 5.6%. They recorded the process of IRMA invading the NPA and forming new capillary networks over time using OCTA. 2)
This report suggests that NPAs may have two distinct characteristics. 2)
In a 1-year OCTA follow-up study, the odds ratio (OR) for baseline nonperfusion and DR progression was 8.73, and the OR for deep nonperfusion and treatment intervention was 3.39, suggesting that OCTA nonperfusion indices may serve as prognostic biomarkers. The baseline FAZ area tends to expand by 5–10% per year in eyes with known DMI. Research on the application of AI to OCTA image analysis is also ongoing. 3)
Although rare, spontaneous reperfusion has been reported. Hou et al. (2022) reported the process of IRMA invading the NPA region and forming new capillary networks. 2) In particular, NPAs without thinning (early changes) tend to reperfuse within 2 months. Good glycemic control may also promote reperfusion.
Datlinger F, Georgi T, Stegmann H, et al. Assessment of detailed photoreceptor structure and retinal sensitivity in diabetic macular ischemia using adaptive optics-OCT and microperimetry. Invest Ophthalmol Vis Sci. 2021;62(13):1.
Hou S, Chen L, Shan K, et al. Spontaneous retinal reperfusion of capillary nonperfusion areas in diabetic retinopathy. Case Rep Ophthalmol. 2022;13:818-824.
American Academy of Ophthalmology Retina/Vitreous Panel. Diabetic Retinopathy Preferred Practice Pattern. Ophthalmology. 2024.