Stage 1 (Mild)
Punctate superficial keratopathy: Irregularity and hyperplasia of the corneal epithelium
Shortened tear breakup time: Decreased tear film stability
Superficial neovascularization: Vascular invasion from the limbus

Corneal neurotization (CN) is a surgical procedure that transfers healthy sensory nerves to the cornea to achieve reinnervation for neurotrophic keratopathy (NK) 1). NK is a refractory disease caused by trigeminal nerve damage, leading to reduced or absent corneal sensation and disruption of epithelial homeostasis, with a prevalence of approximately 5 per 10,000 people.
Conventional NK treatments such as artificial tears and tarsorrhaphy are symptomatic and do not address the underlying nerve damage. CN is the only surgical method that can stabilize the ocular surface and improve long-term prognosis by restoring corneal sensation 1).
Since then, surgical techniques have rapidly diversified, including endoscopic approaches and the introduction of acellular allografts 1).
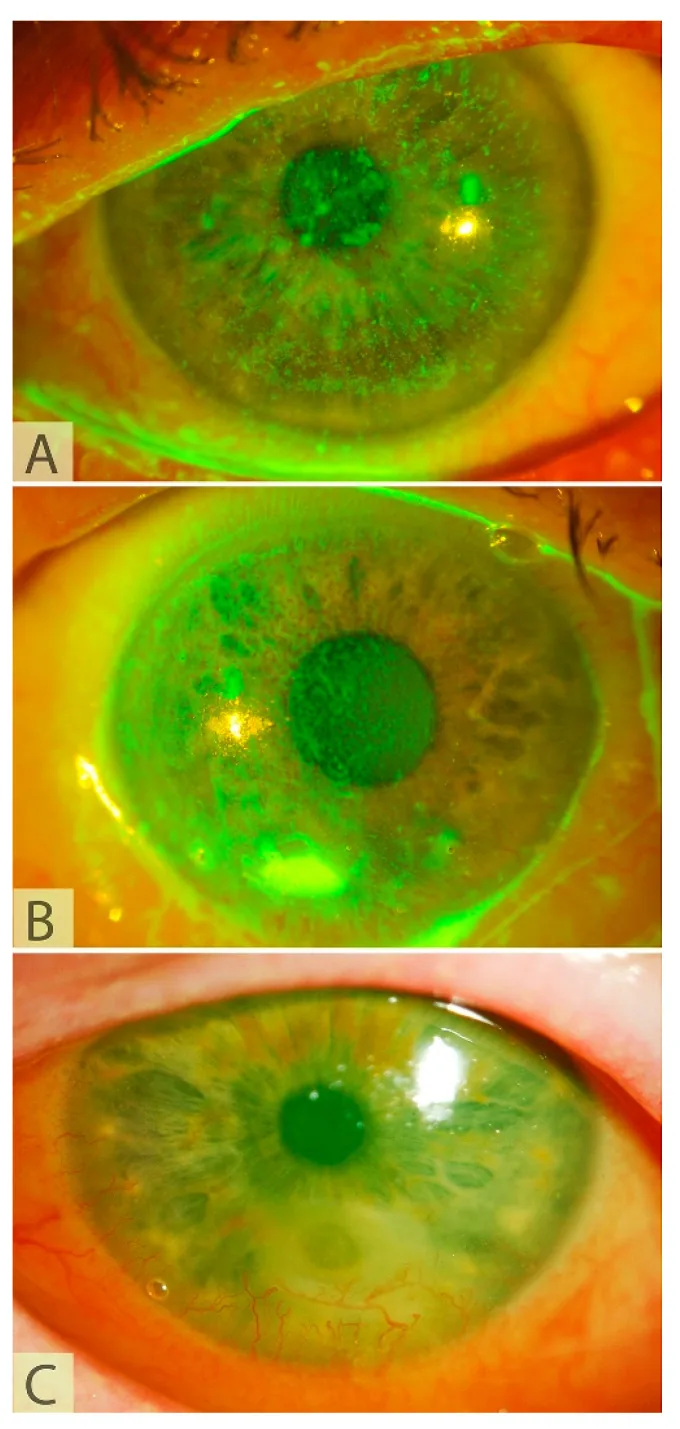
The symptoms and findings of neurotrophic keratopathy that are the target of CN are shown below.
Patients with NK are less aware of symptoms due to decreased corneal sensation.
The severity of NK is assessed using the Mackie classification.
Stage 1 (Mild)
Punctate superficial keratopathy: Irregularity and hyperplasia of the corneal epithelium
Shortened tear breakup time: Decreased tear film stability
Superficial neovascularization: Vascular invasion from the limbus
Stage 2 (Moderate)
Persistent epithelial defect: Epithelial defect with rolled edges
Stromal edema: Accompanied by Descemet’s membrane folds
Anterior chamber inflammation: Mild flare
Stage 3 (Severe)
Corneal melting: Stromal thinning and ulceration
Corneal perforation: Most severe, with progressive melting
Secondary infection: Increased risk of secondary infection
Corneal sensitivity is quantitatively assessed using the Cochet-Bonnet esthesiometer (CB). On a scale from 0 mm (no sensation) to 60 mm (normal), values below 20 mm indicate hypoesthesia1).
NK results from damage at any point along the trigeminal nerve pathway1)3).
Non-invasively observe the density and morphology of the subbasal nerve plexus2). It is useful for preoperative baseline and postoperative nerve regeneration monitoring. Improvement in nerve parameters is confirmed from 3 months postoperatively2)4).
The Cochet-Bonnet esthesiometer (CBA) is the standard quantitative test. A 0.12 mm diameter nylon filament is applied perpendicularly to the cornea starting at 60 mm (maximum length, minimum pressure) and shortened in 10 mm increments until sensation is perceived. Shorter filament length indicates higher pressure and more severe hypoesthesia. Less than 20 mm indicates low sensitivity, and 50 mm or more indicates high sensitivity.
Treatment of NK is based on a stepwise approach according to the Mackie stage.
Patients who do not respond to conservative treatment or have persistent corneal anesthesia for more than one year are candidates for CN 1).
A method of transferring a donor nerve directly to the corneal limbus.
A method of bridging the donor nerve and the cornea using an interposition nerve graft. It is used in 63% of all cases 2).
| Item | Direct Method | Indirect Method |
|---|---|---|
| Speed of sensory recovery | Fast | Somewhat slow |
| Outcomes at 12 months | Equivalent | Equivalent |
| Secondary surgical site | None | Present |
At 12 months, there is no significant difference in sensory recovery between the direct and indirect methods1)2).
Donor nerve selection is based on nerve viability, axon count, and proximity to the cornea5).
| Nerve | Number of myelinated axons | Characteristics |
|---|---|---|
| Supraorbital nerve | Approximately 6,000 | Close to cornea, first choice |
| Supratrochlear nerve | Approximately 2,500 | Anatomical variation possible |
| Great auricular nerve | Approximately 6,530 | Useful for bilateral NK |
Terzis et al. proposed a cutoff of 900 myelinated axons required for effective nerve reconstruction 4). The distal portions of the supraorbital and supratrochlear nerves each contain more than 2,000 myelinated axons, fully meeting this criterion 4)5).
Comparative studies show no significant difference in corneal sensation recovery at 12 months. Direct method does not require a graft and sensation recovery is slightly faster (CB values improve statistically significantly at 3–6 months), but indirect method can be performed through small incisions and is applicable to bilateral NK. The choice of technique depends on donor nerve availability, distance to the cornea, and surgeon experience.
The cornea is one of the most densely innervated tissues in the body, with approximately 7,000 nociceptors per mm². Its sensitivity is 40 times that of dental pulp and 400–600 times that of skin.
Innervation reaches the cornea via the first branch of the trigeminal nerve (ophthalmic nerve) → nasociliary nerve → long ciliary nerves. The nerves penetrate Bowman’s layer and form the subepithelial nerve plexus. 70% of receptors are polymodal (responding to chemical, thermal, and mechanical stimuli), 20% are mechanoreceptors, and 10% are cold receptors.
Corneal nerves contain and release neurotransmitters such as substance P, CGRP, neuropeptide Y, and VIP. These regulate epithelial cell proliferation, migration, and regeneration, and are essential for maintaining corneal homeostasis.
Trophic factors such as NGF, BDNF, GDNF, NT-3, and NT-4/5 are also supplied by corneal nerves. Loss of these factors is the root cause of delayed epithelial healing and homeostatic disruption in NK.
In CN, a healthy donor nerve is repositioned at the corneoscleral junction 1).
In the direct method, the distal end of the donor nerve is directly implanted into the cornea. In the indirect method, an interposition graft bridges the donor nerve and the cornea. In both cases, axonal fibers extend from the donor nerve into the corneal stroma and subepithelial layer, resulting in corneal reinnervation 1).
Axonal regeneration follows the typical pattern of Wallerian degeneration 1). After initial axonal breakdown, macrophages remove degenerated myelin, and Schwann cells differentiate and migrate from the healthy proximal portion of the donor nerve to promote axonal regeneration. Upregulation of NGF and IL-1 plays an important role in promoting axonal sprouting 1).
In a rat model, CN increased corneal nerve density, but only a small proportion of regenerated axons through the graft reached the cornea. It has been suggested that the cornea may selectively allow the growth of only unmyelinated nerve fibers with a specific phenotype 1).
The trophic function of corneal nerves tends to recover earlier than sensory function. Therefore, even if CB value improvement is poor at 12 months, there are cases where corneal nerves are confirmed by IVCM and epithelial defects have healed 1).
A 2025 scoping review (12 studies, 164 eyes) reported a success rate of CN of 60.7–100% (mean 90%). The mean preoperative CCS (CBA) of 2.7 mm significantly improved to 36.0 mm postoperatively (range: 21.1–49.7 mm). Corneal sensation appeared at a mean of 4.1 months and peaked at 12 months 2).
The indirect method (sural nerve graft) was the most common, accounting for 63% of all cases. Acellular allografts showed comparable outcomes to autografts, with advantages of shorter surgical time and avoidance of donor site complications 2).
In a report by Catapano et al. on 19 eyes (mean age 12.5 years), 87% of patients achieved CB 40 mm, and 64% recovered full sensation. The group with fascicular insertion into the corneoscleral tunnel showed earlier sensory recovery 2).
In an 11-year-old boy with Mackie stage 1, objective sensory improvement appeared 6 months after MICN, and central corneal sensation reached 20 mm at 21 months. Schirmer test improved from 15 mm to 30 mm, and visual acuity recovered from 6/18 to 6/5 6).
In two cases of advanced age (62 and 70 years) and long-term denervation (23 years), sustained sensory improvement was not achieved after MICN. Proximal trigeminal nerve injury (e.g., after acoustic neuroma surgery) may have low “functional reserve” and lead to poorer prognosis compared to peripheral causes (e.g., herpetic keratitis) 7).
These findings suggest that advanced age, long-term denervation, and proximal trigeminal nerve injury are poor prognostic factors for CN and should be considered in patient selection and counseling 7).
A 2025 scoping review (164 eyes) reported success rates ranging from 60.7% to 100% (mean 90%). Preoperative corneal sensation (CBA) improved significantly from an average of 2.7 mm to 36.0 mm postoperatively, and at 12 months, over 60% of children achieved normal sensation. However, outcomes may be poorer in elderly patients and those with long-term denervation.
- Hubschman S, Rosenblatt MI, Cortina MS. Corneal neurotization for the treatment of neurotrophic keratopathy. Curr Opin Ophthalmol 2025;36:294-301.
- Samoilă O, Samoilă L, Petrescu L. Corneal Neurotization, Recent Progress, and Future Perspectives. Biomedicines 2025;13:961.
- Dragnea DC, Krolo I, Koppen C, et al. Corneal Neurotization-Indications, Surgical Techniques and Outcomes. J Clin Med 2023;12:2214.
- Saini M, Jain A, Vanathi M, et al. Current perspectives and concerns in corneal neurotization. Indian J Ophthalmol 2024;72:1404-11.
- Serra PL, Giannaccare G, Cuccu A, et al. Insights on the Choice and Preparation of the Donor Nerve in Corneal Neurotization for Neurotrophic Keratopathy: A Narrative Review. J Clin Med 2024;13:2268.
- Lee BWH, Khan MA, Ngo QD, et al. Minimally invasive, indirect corneal neurotization using an ipsilateral sural nerve graft for early neurotrophic keratopathy. Am J Ophthalmol Case Rep 2022;27:101585.
- Ting DSJ, Pradhan SP, Barnes E, et al. Minimally invasive corneal neurotization for neurotrophic keratopathy: The potential effect of age, denervation chronicity and lesion location. Am J Ophthalmol Case Rep 2023;29:101804.