Band Keratopathy
Incidence: Approximately 32% of cases
Calcium deposition in the central cornea due to chronic inflammation, causing visual impairment. Treated with EDTA chelation or excimer laser.
Juvenile idiopathic arthritis (JIA) is a general term for chronic arthritis of unknown cause that develops in children under 16 years of age. It was formerly called juvenile rheumatoid arthritis. According to the ILAR criteria of the International League of Associations for Rheumatology, JIA is classified into seven subtypes: systemic, oligoarticular, RF-negative polyarticular, RF-positive polyarticular, psoriatic arthritis, enthesitis-related arthritis, and undifferentiated arthritis. Among these, the four subtypes—systemic, oligoarticular, RF-negative polyarticular, and RF-positive polyarticular—correspond to the former juvenile rheumatoid arthritis (JRA) and account for 94% of JIA cases.
Chronic uveitis is the most important ocular complication of JIA and is the most common cause of uveitis in children. It accounts for 41–47% of all pediatric uveitis cases. 1) Uveitis usually appears after the onset of arthritis, but in 3–7% of cases, it precedes arthritis. The median time to onset is 5.5 months, and it often develops within 5–7 years after arthritis onset; in particular, 82–90% of cases occur within the first 4 years. 3)
The rate of uveitis varies greatly depending on the JIA subtype.
| JIA subtype | Uveitis rate | Risk |
|---|---|---|
| Persistent oligoarthritis | 41–46% | High |
| RF-negative polyarthritis | 5–23% | Moderate to high |
| Psoriatic arthritis | 10–36% | Moderate |
| Enthesitis-related arthritis | 7–25% | Moderate |
| Systemic arthritis | 0% | Low |
It is observed in about 20% of oligoarticular type and about 5% of polyarticular type, and is not considered to occur in systemic type. In the Nordic cohort, uveitis was confirmed to complicate 10–22% of all JIA patients. 3)
In the early stage, there are almost no subjective symptoms such as redness, pain, or photophobia, and it progresses quietly, often described as “white uveitis.” Especially in young children, subjective complaints are scarce and examination can be difficult. It is extremely difficult to detect without regular screening using a slit-lamp microscope.
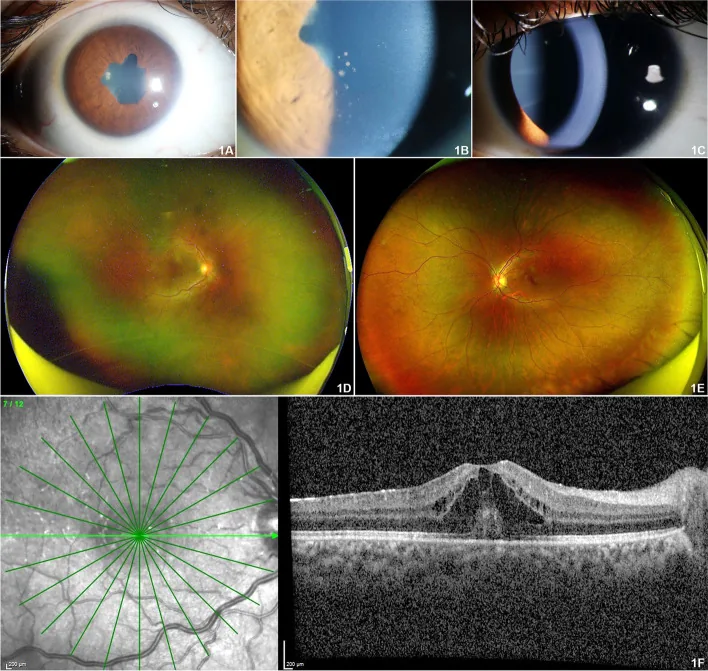
The most prominent feature of JIA-associated uveitis is its initial asymptomatic nature.
97.8% of JIA-associated uveitis presents as anterior uveitis (iridocyclitis), typically bilateral and non-granulomatous. 2) Many patients already have multiple complications at the time of ophthalmologic examination.
Band Keratopathy
Incidence: Approximately 32% of cases
Calcium deposition in the central cornea due to chronic inflammation, causing visual impairment. Treated with EDTA chelation or excimer laser.
Posterior Synechiae
Incidence: Approximately 28% of cases
Adhesion between the iris and lens. Often present at initial diagnosis and can cause pupillary block. Prophylaxis with mydriatics is important.
Complicated Cataract
Cumulative incidence: 0.05 per eye-year
Develops due to chronic inflammation and steroid use (approximately 22%). Cataract surgery is required under controlled active inflammation.
Secondary Glaucoma
Cumulative incidence: 0.03 per eye-year
Present in 15% of cases. More frequent in JIA-associated uveitis than in idiopathic uveitis. A major cause of poor visual prognosis. 2)
Other findings include inflammatory cells in the anterior vitreous, cystoid macular edema (3%), hypotony (9%), optic neuritis, and retinal vasculitis. Large cohort studies report that one-third of patients already have visual impairment at the first visit. Complications such as band keratopathy, posterior capsule opacification, and glaucoma develop in approximately 67% of cases cumulatively.
Leaving it untreated can lead to vision loss. JIA-associated uveitis is a classic example of “painless uveitis” (white uveitis), where inflammation persists without symptoms, causing band keratopathy, cataracts, and glaucoma to progress. Even without symptoms, regular ophthalmology follow-up is the only way to protect vision.
JIA-associated uveitis is a chronic inflammatory disease driven by autoimmune mechanisms, involving interactions between environmental factors and multiple genes. Overproduction of inflammatory cytokines (IL-1, IL-6, TNF-α) plays a central role in pathogenesis. In the joint synovium, granulation tissue called pannus forms, and a similar inflammatory cascade is continuously activated in the uvea.
Risk factors for developing uveitis (Nordic guideline 2023)3):
Protective factors (significantly reduce the risk of developing uveitis)3):
The diagnosis of uveitis in JIA patients is often made during screening examinations. Detection at an asymptomatic stage directly leads to improved visual prognosis.
Risk stratification criteria combine JIA subtype, ANA positivity, age at onset, and disease duration. 3, 4)
| Risk level | Main criteria | Screening frequency |
|---|---|---|
| High risk | Oligoarthritis + ANA positive + onset ≤6 years + disease duration <4 years | Every 3 months |
| Moderate risk | Meets some of the above | Every 6 months |
| Low risk | Systemic type, RF-positive polyarthritis, ERA, onset >6 years | Every 12 months (for 2 years only) |
Diseases that combine arthritis and uveitis include the following: ankylosing spondylitis, Reiter syndrome, psoriatic arthritis, sarcoidosis, inflammatory bowel disease, early-onset sarcoidosis (EOS), Blau syndrome, and TINU syndrome.
It depends on the risk. For high risk (oligoarticular type, ANA positive, onset before age 6, disease duration less than 4 years), screening every 3 months is recommended. 3, 4) For moderate risk, every 6 months; for low risk (systemic type, RF-positive polyarthritis, etc.), observation every 12 months for 2 years is a guideline. It is important to set the interval individually in collaboration with the pediatric rheumatologist and ophthalmologist.
Management of JIA-associated uveitis usually requires co-management by a pediatric rheumatologist and an ophthalmologist. Long-term control of inflammation is fundamental to preventing complications and protecting vision. Uveitis treatment guidelines recommend starting with local treatment and, for refractory cases, stepwise introduction of immunomodulatory therapy and biologic agents. 8)
| Drug | Dosage and Administration | Role |
|---|---|---|
| Methotrexate (MTX) | 10–15 mg/m²/week (subcutaneous or oral) | First-line. Risk of uveitis onset HR 0.14–0.63 |
| Adalimumab | Weight <30 kg: 20 mg/2 weeks, ≥30 kg: 40 mg/2 weeks (subcutaneous) | Second-line for MTX-refractory cases. Efficacy established in SYCAMORE trial |
| Infliximab | 5 mg/kg at weeks 0, 2, 6, then every 8 weeks (intravenous) | Option for refractory cases |
| Tocilizumab (IL-6 inhibitor) | Approved for JIA arthritis in Japan | Evidence accumulating for uveitis |
| Etanercept | — | Ineffective for uveitis (due to TNF receptor inhibition) |
Methotrexate (MTX) is the first-line immunomodulatory therapy for juvenile idiopathic arthritis-associated uveitis. 5) It takes 4 to 12 weeks to take effect. In 27–48% of cases, MTX alone does not adequately control inflammation, and 20% experience side effects (vomiting, liver dysfunction). 2)
Adalimumab is positioned as a second-line option for MTX-refractory cases. The SYCAMORE trial (Ramanan 2017), a double-blind RCT, showed that the treatment failure rate was significantly reduced from 60% in the MTX-alone group to 27% in the MTX+adalimumab combination group (P<0.0001). 6) It is approved for pediatric non-infectious uveitis. As a monoclonal TNF antibody, it has a protective effect on uveitis, unlike the TNF receptor inhibitor etanercept. 3)
The ADJUVITE trial (Quartier 2018) is a double-blind RCT of adalimumab for early chronic JIA-associated anterior uveitis. 7) It is positioned as a study demonstrating the benefits of early introduction.
Before initiating TNF inhibitors, it is essential to perform tuberculosis and HBV screening and infection evaluation. Administration and management in collaboration with an ophthalmologist who is a member of the Ocular Inflammation Society is required. 9)
Not necessarily, but there is strong evidence from the SYCAMORE trial (Ramanan 2017) that the treatment failure rate was significantly reduced from 60% with MTX alone to 27% with combination therapy. 6) The current standard approach is a stepwise strategy: first use MTX, and if adequate inflammation control is not achieved, consider adding adalimumab. To protect the child’s future vision, aggressive treatment is recommended when deemed necessary.
The pathogenesis of JIA-associated uveitis is not fully understood, but immunological mechanisms are thought to be primarily involved.
Cellular immunity: CD4+ T cells (Th17 predominant) infiltrate uveal tissue and produce IL-17, IL-6, and TNF-α. Overproduction of inflammatory cytokines increases vascular endothelial permeability, sustaining edema and inflammatory cell infiltration in the iris and ciliary body. In the joint synovium, pannus formation leads to cartilage and bone destruction, and systemic symptoms such as fever, malaise, anemia, and growth impairment are also induced.
Association with autoantibodies: Antinuclear antibody (ANA) positivity is found in about 80% of patients with uveitis. Associations with HLA-DR5 (DRB1*1101) and HLA-DR11 have been reported in oligoarticular type. Autoreactive T-cell responses and activation of the classical complement pathway are also involved.
Dissociation from arthritis: Uveitis may persist independently even after arthritis symptoms subside. Because it continues through different inflammatory pathways, ongoing ophthalmologic monitoring is necessary even when arthritis is quiescent.
Main tissue changes resulting from chronic inflammation:
Ramanan et al. (2017) conducted a double-blind RCT comparing MTX alone vs. MTX + adalimumab, showing that the adalimumab group reduced treatment failure rates to 27% vs. 60% (P<0.0001). 6) This trial provided the basis for approval of adalimumab for pediatric non-infectious uveitis. Long-term follow-up data are being accumulated with predefined discontinuation criteria.
In a retrospective study from the Bristol area by Cann et al. (2018), among 166 children with non-infectious uveitis, the use of biologics reached 35%, with an incidence of visual loss (logMAR > 0.3) of 0.05/eye-year and severe visual loss (logMAR ≥ 1.0) of 0.01/eye-year. 2) This represents an improvement from 0.10/eye-year before the introduction of biologics, demonstrating the progress of treatment.
The latest recommendations from the Multinational Interdisciplinary Working Group for Uveitis in Childhood (MIWGUC) by Foeldvari et al. (2023) clearly recommend early introduction of adalimumab for MTX-refractory JIA-associated uveitis and advise against selecting etanercept for the treatment of uveitis. 10)
The efficacy of JAK inhibitors such as ruxolitinib and tofacitinib for refractory JIA-associated uveitis has been reported in case reports and small series, but definitive evidence is not yet available at this time.
In the Nordic cohort, new-onset cases were observed even after the transition to adult care after age 16 (12 of 434 cases were new-onset at age 23), highlighting the challenge of seamless collaboration from childhood to adulthood. 3) Since MTX and MMF are teratogenic, adalimumab is considered a relatively safe option for women of childbearing age when selecting medications.