Adie Pupil
Lesion: Ciliary ganglion (peripheral)
Pupil size: Moderate dilation, irregular shape
Laterality: Usually unilateral (about 80%)
Tonic: Present (miosis persists for a long time)
Pilocarpine response: Miosis with low concentration

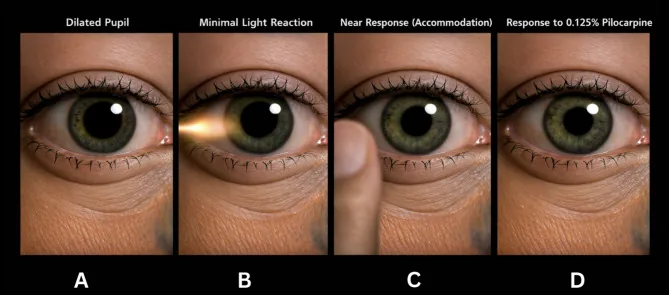
Adie tonic pupil is a condition in which, due to parasympathetic denervation, the light reflex of the affected pupil is lost or diminished, while the near reflex shows a good and tonic constriction.
The disease is named after William John Adie, and was independently reported around the same time in 1931 by Adie, Morgan, Symons, and Holmes. The prevalence is 2 per 1,000 population, with a female-to-male ratio of 2.6:1 (about 70% are female), and the average age of onset is 32 years. About 80% are unilateral, and progression to bilateral occurs at a rate of 4% per year.
Related syndromes include the following two important conditions:
Approximately 80% of cases are unilateral. However, progression to bilateral involvement occurs at a rate of 4% per year, so attention should also be paid to pupillary changes in the contralateral eye during follow-up.
Initially, the affected eye is dilated and anisocoria is marked compared to the contralateral side, but in the chronic course, the affected pupil tends to constrict. This is thought to be due to aberrant regeneration of parasympathetic nerve fibers from the ciliary body innervating the sphincter pupillae muscle.
Most cases are idiopathic (unknown cause), and the responsible lesion is in the peripheral ciliary ganglion and short posterior ciliary nerves.
Identifiable causes include the following:
Regarding the pathogenesis of Ross syndrome, a theory has been proposed that genetic predisposition combined with environmental factors (such as viral infection) contributes. There are reports of Ross syndrome in monozygotic twins and cases following cytomegalovirus infection, suggesting both genetic and infectious factors may be involved3).
Regarding tonic pupil after COVID-19 infection, multiple cases of immune-mediated onset have been reported since the COVID-19 pandemic, and it is increasingly recognized as a neuro-ophthalmic complication after infection1, 2).
Diagnosis is easy when typical symptoms (segmental palsy, vermiform movements, light-near dissociation) are present.
The low-concentration pilocarpine test is most widely used.
Differential diagnosis is needed, focusing on diseases that present with light-near dissociation.
Adie Pupil
Lesion: Ciliary ganglion (peripheral)
Pupil size: Moderate dilation, irregular shape
Laterality: Usually unilateral (about 80%)
Tonic: Present (miosis persists for a long time)
Pilocarpine response: Miosis with low concentration
Argyll Robertson pupil
Lesion: Midbrain pretectal area (syphilis)
Pupil size: Bilateral severe miosis
Laterality: Bilateral
Tonic: None
Pilocarpine response: No response to low concentration
Tectal pupil
Lesion: Dorsal midbrain (e.g., pineal tumor)
Pupil size: Moderate mydriasis
Laterality: Bilateral
Parinaud syndrome: Frequently associated
Tonic: None
Other differential diagnoses include aberrant regeneration of the oculomotor nerve after palsy, orbital trauma/tumor, varicella-zoster infection, Fisher syndrome, Charcot-Marie-Tooth disease, and neurosarcoidosis.
Syphilis serology (RPR), and history of diabetes, trauma, or neurological disease can aid diagnosis. If Ross syndrome is suspected, Minor’s test (iodine-starch test) can detect anhidrotic areas 3).
It cannot be ruled out. In the acute phase, denervation supersensitivity may not yet be established, so the test can be negative. If clinical findings (vermiform movements, segmental palsy) are typical, Adie’s pupil can be diagnosed clinically even with a negative test. Serial testing is also useful.
Adie’s pupil is a benign condition. For most patients, the basic approach is to explain that it is benign and to observe over time.
Prognosis: Accommodative paresis may improve over months to years. However, the pupillary light reflex usually does not recover. When associated with systemic disease, the prognosis is not necessarily favorable, and management of the underlying condition is important.
Treatment of compensatory hyperhidrosis in Ross syndrome: Topical glycopyrrolate, aluminum chloride cream, and botulinum toxin injections are used. For severe cases where surgery is desired, thoracic sympathectomy is an option 3).
Blurred vision and difficulty with near vision due to accommodative paresis may improve over months to years. However, the pupillary light reflex usually does not recover, so normalization of the pupillary response to light cannot be expected. This condition rarely leads to severe visual impairment and is a benign condition that can be safely observed.
Two main mechanisms play a central role in the pathogenesis of Adie’s pupil: “denervation supersensitivity” and “aberrant regeneration.”
Parasympathetic neuroanatomy: 95% of parasympathetic fibers from the Edinger-Westphal (EW) nucleus of the oculomotor nerve innervate the ciliary muscle (involved in accommodation), and 5% innervate the sphincter pupillae (involved in the light reflex). The ratio of neurons in the ciliary ganglion related to the light reflex versus accommodation is 3:97, indicating that fibers for the light reflex are originally few.
The course of onset progresses in the following order:
Neuroanatomical explanation of light-near dissociation: The supranuclear fibers for the near response to the EW nucleus run more ventrally than the midbrain pretectal area and posterior commissure, through which the afferent fibers of the light reflex pass. Therefore, damage to the pretectal area (central) can also cause similar dissociation, but in Adie’s pupil, the lesion is in the ciliary ganglion (peripheral), which is key for differentiation.
COVID-19-related mechanism: In tonic pupils after SARS-CoV-2 infection, three mechanisms have been proposed: direct viral neural invasion, endothelial dysfunction, and neurotoxicity due to excessive inflammation and cytokine release1).
Pathophysiology of Ross syndrome: Dorsal root ganglion neurons and the parasympathetic nervous system are thought to share a common origin from neural crest cells, which is proposed as a hypothesis to explain the loss of tendon reflexes in Ross syndrome3).
Since the COVID-19 pandemic, there have been successive case reports of tonic pupil as a neuro-ophthalmic complication after infection.
Quijano-Nieto et al. (2021) reported a case of a 36-year-old woman who developed bilateral tonic pupil 17 days after COVID-19 infection (PCR positive)1). Brain MRI, blood tests, and cerebrospinal fluid tests were all normal. Strong miosis was confirmed in both eyes with 0.125% pilocarpine eye drops. An immune-mediated mechanism is presumed.
Gopal et al. (2021) reported a case of a 37-year-old woman who developed tonic pupil in the right eye 3 weeks after COVID-19 infection2). Right pupil diameter was 5.5 mm, with vermiform movement and segmental constriction. Miosis was confirmed with 0.1% pilocarpine eye drops. No significant findings on brain and orbital MRI, and post-infection immune response is suspected as the cause.
Ahmad et al. (2022) reported two cases of Ross syndrome diagnosed within one month at the same institution3). Case 1 (24-year-old woman) showed the triad of left tonic pupil, loss of right ankle jerk, and segmental anhidrosis up to T4 level. Case 2 (42-year-old woman) was “Ross syndrome plus” with bilateral tonic pupil, loss of lower limb tendon reflexes, and Horner syndrome.
It has been proposed that Ross syndrome may be a synucleinopathy, like Parkinson’s disease, dementia with Lewy bodies, and multiple system atrophy3). It has also been suggested that Ross syndrome, Holmes-Adie syndrome, and Harlequin syndrome may be different phenotypes of the same disease process, and integration of these disease concepts is expected in the future.
Quijano-Nieto BA, Córdoba-Ortega CM. Tonic pupil after COVID-19 infection. Arch Soc Esp Oftalmol. 2021;96(7):353-355.
Gopal M, Ambika S, Padmalakshmi K. Tonic Pupil Following COVID-19. J Neuroophthalmol. 2021;41:e764-e766.
Ahmad R, Saurabh K. Two Cases of Tonic Pupil: Ross and Ross Syndrome Plus. Cureus. 2022;14(2):e22305.