Other congenital cataracts
Morphology: Anterior polar, posterior polar, lamellar, total cataract, etc.
Key differences: Location, shape, and color of opacities differ.
Examination: Slit-lamp examination, family history taking.

Cerulean cataract is a hereditary developmental cataract characterized by scattered bluish-white opacities in the lens nucleus and cortex. It is also called “blue dot cataract.” Nuclear opacities are classified by shape into powdery, jellyfish-like, or dendritic types, but cerulean cataract is named based on the color of the opacities.
It follows an autosomal dominant inheritance pattern. Progression is slow, and it often does not affect vision until adulthood. However, some patients experience early visual impairment, and if left untreated, it can lead to amblyopia and nystagmus.
An association with Down syndrome is known. Children with Down syndrome can develop congenital or acquired cataracts, and particularly cerulean cataract (composed of amyloid deposits) has been reported 1).
Children with Down syndrome can develop congenital or acquired cataracts. In particular, cerulean cataract composed of amyloid deposits has been reported 1). Down syndrome is also known to be associated with keratoconus (up to 15% of adults), and cases of coexisting corneal ectasia and cerulean cataract in the same patient have been reported 1).
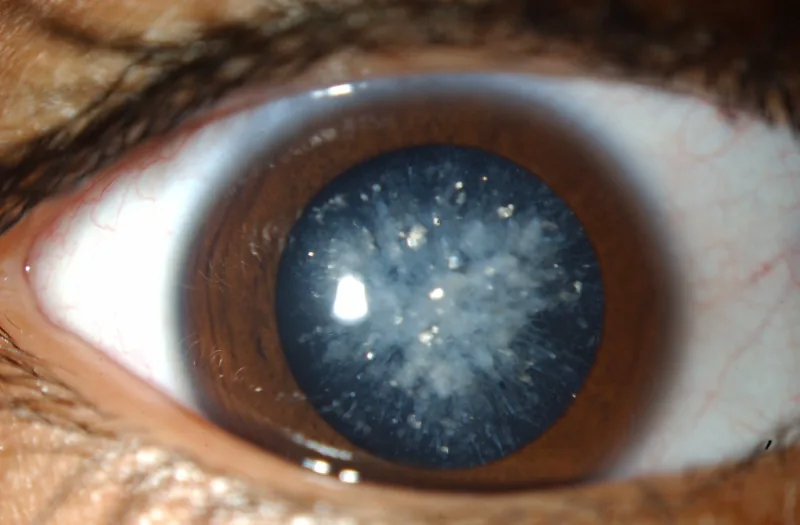
Slit-lamp microscopy reveals tiny blue or white opacities in the superficial layers of the lens nucleus. The opacities begin in the embryonic nucleus and are scattered in the nucleus and cortex. They are usually bilateral.
In cases associated with Down syndrome, keratoconus findings such as Fleischer ring and conical protrusion of the cornea may be observed simultaneously1). Cases complicated by acute corneal hydrops have also been reported1).
In the morphological classification of congenital cataracts, there are capsular, polar, nuclear, lamellar, punctate, sutural, and total cataracts. Blue cataract is classified as a subtype of nuclear cataract.
Blue cataract follows an autosomal dominant inheritance pattern. At least four genetic loci have been identified.
| Locus | Chromosomal Location |
|---|---|
| CCA1 | 17q24 |
| CCA2 | 22q11.2-q12.2 |
| CCA3 | 2q33-q35 |
| CC4 | 16q22-q23 |
Mutations have been identified in the beta-B2-crystallin gene (CRYBB2), gamma-D-crystallin gene (CRYGD), V-MAF avian musculoaponeurotic fibrosarcoma oncogene homolog gene (MAF), and major intrinsic protein of lens fiber gene (MIP) as causative genes.
The most common etiology of congenital cataract overall is idiopathic (30-50%), followed by hereditary (most commonly autosomal dominant), intrauterine infection, metabolic disorders, chromosomal abnormalities, ocular diseases, and systemic diseases/syndromes.
Mutations in CRYBB2 (beta-B2-crystallin), CRYGD (gamma-D-crystallin), MAF, and MIP (major intrinsic protein of lens fiber) have been identified. These encode structural proteins and transcription factors of the lens, and mutations impair lens transparency. Four genetic loci are known: 17q24, 22q11.2-q12.2, 2q33-q35, and 16q22-q23.
This is the basic diagnostic test. Small blue or white opacities are observed on the surface of the lens nucleus. It is detected at birth or during routine eye examinations.
Other congenital cataracts
Morphology: Anterior polar, posterior polar, lamellar, total cataract, etc.
Key differences: Location, shape, and color of opacities differ.
Examination: Slit-lamp examination, family history taking.
Developmental cataract
Morphology: Opacities that progress after birth.
Key differences: Rate of progression and presence of systemic complications.
Examination: Serial slit-lamp examinations.
Traumatic cataract
Morphology: Opacities secondary to trauma.
Key differences: History of trauma.
Examination: History taking, anterior segment examination.
Taking a family history is important for differential diagnosis. Genetic testing may identify the causative mutation.
Currently, there is no treatment to prevent the formation or progression of blue cataract. Regular ophthalmic evaluations are performed to monitor the progression of visual impairment.
When visual acuity decreases and interferes with daily life, cataract surgery (phacoemulsification) and intraocular lens implantation are indicated. Visual function after intraocular lens implantation is good, and surgery is actively performed when it is judged that visual improvement is certain.
For unilateral congenital cataract with severe visual impairment, early surgery is necessary. Intervention at an appropriate time is important from the perspective of amblyopia prevention. In cases with severe ocular complications or central nervous system diseases, visual improvement cannot be expected, so surgical indications are carefully considered.
Cataract surgery is indicated when visual acuity decreases and interferes with daily life or visual development. In many patients, vision is not affected until adulthood, so regular observation is performed until then. In children with early visual impairment, timely surgical intervention is important to prevent amblyopia.
Blue cataract is caused by mutations in crystallin genes. Crystallins are major structural proteins of the lens and are essential for maintaining lens transparency and refractive function.
Mutations in CRYBB2 and CRYGD impair the normal folding of lens proteins. Aggregation of abnormal proteins leads to loss of lens transparency and characteristic blue-white opacification. The MAF gene encodes a transcription factor involved in lens development and differentiation. The MIP gene encodes a major membrane protein of lens fibers and contributes to lens homeostasis.
In blue cataracts associated with Down syndrome, it has been reported that the opacity consists of amyloid deposits 1). Down syndrome is also known to be associated with keratoconus, and it has been pointed out that the complication of hypothyroidism may contribute to the progression of corneal ectasia 1).